Characterization of Key Molecular Mechanisms and Biological Pathways Involved in Wortmannin Induced Breast Cancer MCF-7 Programmed Cell Death
Rozina Akter1, Michael A. Gealt2, Maurice G. Kleve3, Md. Zakir Hossain4, *
1Department of Applied Biosciences, University of Arkansas at Little Rock, Little Rock, Arkansas, USA
2Central Michigan University, Mount Pleasant, Michigan, USA
3Department of Biology, University of Arkansas at Little Rock, Little Rock, Arkansas, USA
4Department of Pharmaceutical Sciences, Biomanufacturing Research Institute and Technology Enterprise (BRITE), North Carolina Central University, Durham, North Carolina, USA
Abstract
The present study aimed to explore the molecular mechanisms and biological pathways involved in Wortmannin induced breast cancer MCF-7 programmed cell death. The direct cellular and molecular effect of Wtmn was investigated selectively on the MCF-7 cancer cell line. To study morphological effects phase contrast microscopy was used. The mitochondrial membrane potential and detection of caspase activity were investigated using fluorescent microscopy. Our morphological, molecular, and caspases expression indicated the intrinsic apoptosis pathways involved, and confirmed the role of Wtmn in these pathways. Our experimental results demonstrated that Wortmannin (Wtmn), an irreversible and selective PI3-K inhibitor, inhibits the proliferation of MCF-7 breast cancer cells, and facilitates their entry into apoptosis. Our data established the anti-cancer properties of Wtmn on MCF-7 breast cancer cells. These studies directed us towards elucidating the molecular mechanisms and biological pathways mainly involved in this process. This findings offer potential drug screening to search for novel inducers of apoptosis on MCF-7 cells, and could be used to design better drugs.
Keywords
MCF-7, Wortmannin, Caspase, Mitochondrial Membrane Potential, and Apoptosis
Received:May 19, 2016
Accepted: May 28, 2016
Published online: September 10, 2016
@ 2016 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY license. http://creativecommons.org/licenses/by/4.0/
1. Introduction
Breast carcinoma is the most common malignancy as well as high morbidity in women worldwide [1-2]. Although several biological properties of Wtmn have been identified, the precise molecular mechanisms underlying its action on cytotoxicity and cell death appear unknown to date. Novel and advanced alternative therapeutic strategies are needed. Studies reported that Wtmn exhibits anti-tumor effects on human breast cancer cell lines in vivo, but the details mechanism of action of cell death are not with the MCF-7 contexts, specifically in the apoptotic programmed cell death perspective in-vitro [3-7]. Therefore, the goal of this research had a specific aim to elucidate the molecular mechanisms and biological pathways involved in Wtmn induced MCF-7 cancer cell death. In most eukaryotes, mitochondria are not only the power house of cellular energy production but also play crucial role in regulating or initiating apoptotic cell death pathways through numerous mechanisms. For example, mitochondrial membrane potential loss (∆ψm), inner mitochondrial membrane (IMM) pore activation leading to release of apoptogenic factors; proteins and activation of caspases cascade [8-11]. During apoptosis induction, some or all of these changes activated [8-9]. Thus for studying the induction of mitochondria-mediated cell death, mitochondrial functional parameters analysis is important [8]. To induce apoptotic cell death by using chemotherapeutic agent, caspase activation is a common biochemical mechanism and hallmark of apoptosis [12-13]. Therefore, we have explored the possible molecular mechanisms and biological pathways involved in Wtmn induced MCF-7 cancer cell death in-vitro.
2. Experimental
2.1. Materials
The MCF-7 cells, EMEM, bovine insulin, penicillin-streptomycin (P-S), and Trypsin - EDTA (TE) was obtained from ATCC (Rockville, Maryland). The Rho-123, and PBS were purchased from Amresco Inc. (Salon, OH). The Hoechst -33342, Caspase 3 / 7 FAM FLICA, Propidium Iodide (PI) stains were from BioExpress and ICT Inc., USA respectively. The DMSO, FBS, standard tissue culture (75 cm2) flask with filtered cap, 6 well, and 96 well plates were purchased from BioExpress (Kaysville, UT).
2.2. Methods
2.2.1. Cell Culture
The MCF-7 cell culture were performed as previously described [11]. Briefly, cells were grown in EMEM supplemented with 10% FBS and Penicillin / Streptomycin at 37°C with 5% CO2 humidified atmosphere.
2.2.2. Phase Contrast Microscopy (PCM)
The MCF-7 cells were cultured in triplicate into 6 well plates at a density of 1 X 106 cells / well and treated with Wtmn for 24 hour period. Images were taken randomly using a Zeiss Axiovert 40 CFL® inverted PCM.
2.2.3. Mitochondrial Membrane Potential Using FM
For FM fluorescence microscopy, Rho-123 and Hoechst 33342 double staining method was applied. Briefly, Wtmn treated cells were pelleted, resuspended in ice-cold PBS, and stained with 1µM Rho-123 for 15 minutes in the dark at room temperature. After washing with PBS cells were resuspended in PBS containing 1 µg / ml Hoechst 33342 and incubate 5 minutes in the dark at room temperature. Then 5 to 6 images for each sample were captured randomly using Nikon 90i fluorescence microscope and analyzed using both FITC green and DAPI blue filter.
2.2.4. Caspases Activity Assay
The MCF-7 cells were allowed to reach about 70% confluency (cell density were adjusted about 1 x 106cells / ml) in six well plate in fresh medium before adding starvation medium and treatment. After that the cells exposed to different concentration of Wtmn (100 nM -10 µM) for specific periods of time (6h - 24h) were collected by centrifugation at 800 x g at 4°C for 12 minutes. Then the cell pellet were resuspended in 290 µl fresh medium followed by the addition of 10 µl FLICA (FAM-DEVD-FMK inhibitors, catalog # 93) reagent and incubated for 1 hour at 37°C and 5 % CO2 in humidified incubator. After collection of the cell pellets were washed two times in working dilution of wash buffer. Then the fresh medium were added and incubated for 1 hour at 37°C humidified incubator. Finally, the medium discarded and the cell pellets were collected and analyzed by FM (fluorescent microscope) according to the manufacturer’s protocol of FAM - FLICA caspase - 3 / - 7 kit (ICT, USA, cat # 93) to detect caspase activity.
2.2.5. Statistical Analysis
Experiments were done in triplicate. Data were expressed as Mean ± Standard Error Mean (SEM). Significance was evaluated by a one way ANOVA and the student T- test.
3. Results
The phase contrast microscopy of Wtmn treated MCF-7 cells have distinct morphological apoptotic characteristics. It includes cell shrinkage with intact membranes as well as condensed cytoplasm along with membrane blebbing (onset of cytoplasmic budding). Morphological observation revealed the typical apoptosis characteristics including loss of adhesion, cell shrinkage, membrane blebbing, reduced cellular volume with condensed contents, body rounded up further demonstrating Wtmn induced apoptosis of MCF-7 cells. Thus, it displays evident to formation of apoptotic bodies.
Mitochondria play a crucial role in the apoptotic process and one of the major mitochondrial events associated with apoptosis is drop of mitochondrial membrane potential (∆ψm). To analyze whether Wtmn induced changes in ∆ψm of MCF-7 cells, the fluorescent dye Rho-123 was used.
The MMP is evaluated by the retention of a specific fluorescent cell-permeant cationic dye, Rho123 which is readily sequestered by active mitochondria after entering into the living cells based on their high transmembrane potential and without cytotoxic effects induction [15-16]. The fluorescent intensity of retained Rho-123 reflects the amount of produced ATP in mitochondria. Since Rho-123 has also several energy independent binding sites [17]. We used it in combination with Hoechst 33342 allowing a clear and elegant distinction between fluorescence microscopic images (Figure 1) of mitochondrial membrane potential (∆ψm) loss induced by Wtmn in MCF-7 cells.
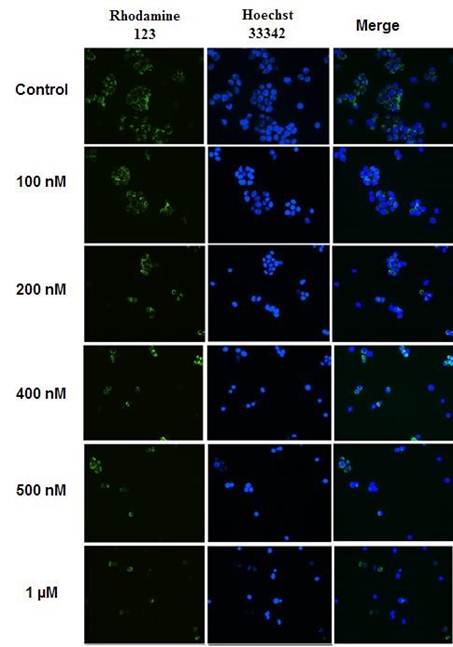
Figure 1. Fluorescent images of mitochondrial membrane potential (∆ψm) loss induced by Wtmn in MCF-7 cells.
Cells were incubated with or without different concentration of Wtmn (100 nM - 1 µM) for 24 hour, stained with Rhodamine123 (Rho-123) and Hoechst 33342 double staining and examined by fluorescence microscopy. The proportions of increased concentration of Wtmn treated cells with gradually decreased Rho-123 fluorescence were observed. After Rho-123 and Hoechst 33342 double staining approximately 300 - 400 cells were analyzed in each experiment with similar result.
Expression of caspase-7 in Wtmn induced MCF-7 cells
Since disrupted MMP associated with apoptotic cell death [18-20]. It is important to investigate whether caspase cascade activation result from perturbation of MMP. The irreversible apoptotic process is directly controlled by caspase cascade activation and the effector caspases (caspase - 3, - 6, - 7) are responsible for many key molecular and structural apoptotic changes. Caspases - 3, - 6 and - 7 are well known biomarkers to trigger apoptotic mechanism induced by different stimuli in different cells [12,21]. To assess this irreversible molecular key event of apoptosis i.e. protease involvement in Wtmn induced MCF-7 cell death, we have examined whether effector caspases activation is associated with apoptotic signaling in MCF-7 cells after exposure to different concentrations of Wtmn (100 nM -1 µM). The commercially available FAM - FLICA caspase - 3 / -7 kits (ICT, USA) was used to detect the presence of caspase - 3 / - 7.
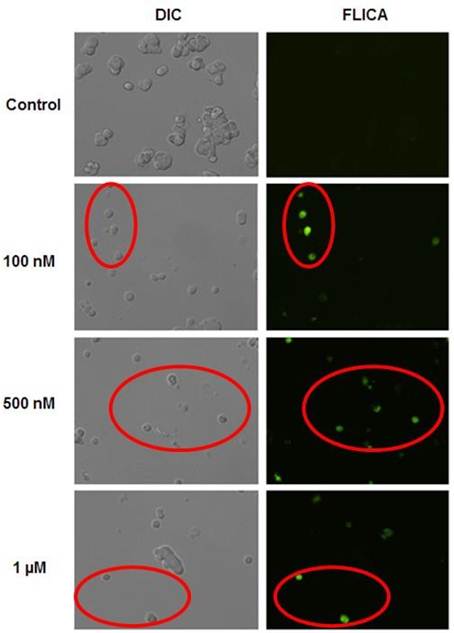
Figure 2. Expression of caspases in MCF-7 cells induced by Wtmn was detected by fluorescence microscopy. Cells were treated without or with Wtmn of different concentration (100nM -1 µM) for 24 hour and labeled with FAM - FLICA caspase - 3 and - 7 as described in materials and methods and viewed under fluorescence microscope. Left panel, the grey DIC image reveals Wtmn untreated (control) and treated (100 nM -1µM) cells in the field of view. Right panel, apoptotic caspase-positive cells fluoresce green (Red circle) with FAM-DEVD-FMK FLICA in the same area of interest. Data are representative of at least 4 - 5 randomly selected fields’ images with similar results.
From our experiments, as shown in Figure 2, the enzyme assay indicated that the Wtmn treated MCF-7 cells were expressing caspases - 7 involved in apoptosis, in a dose dependent manner compared to control in vitro. The caspase - 7 positive (apoptotic) cells fluorescence green with labeled FAM – DEVD - FMK FLICA under FM. However, the DIC image (grey) revealed that, not all of the MCF-7 cells photographed by DIC microscopy were fluorescence green, while those same cells were captured under fluorescence channel (FITC). In contrast, the control group photographed in DIC image exhibited the confluency, but not a significant green fluorescence (caspases negative). Our experimental results revealed that Wtmn treatment in MCF-7 cells expresses caspase-7 in a dose dependent manner compare to control in vitro (red circle). At 1 µM concentration most of the Wtmn treated MCF-7 cells fluoresced green comparing to that of the left panel DIC images. However, caspase-3 did not express in MCF-7 cells is due to their lack of their expression in mRNA level [22].
4. Discussions
It was determined from our experiments that the Wtmn treated MCF-7 cancer cells significantly exhibit apoptogenic morphological, biochemical and molecular characteristics. It is evident by membrane blebbing, cytoplasmic volume reduction, followed by cell shrinkage, disruption of MMP indicators of early apoptotic event and effector caspase - 7 activation. Those properties demonstrated that the cells undergoing apoptotic cell death. Several previous studies reported that disruption of MMP leading to apoptotic cell death [18,20,23,26]. Evidences suggested that proper maintenance of mitochondrial membrane potential is essential for cell survival through ATP generation and oxidative phosphorylation [27-28]. Since mitochondria is considered the power house of cells, irrespective of eukaryotes (higher or lower), mitochondrial dysfunction or loss of MMP leads to PCD [27,29]. Thus, mitochondria are the major stress induced cellular target of injury in apoptotic cell death [27].
We have analyzed the downstream effect of irreversible inhibitor (Wtmn) of PI3-K signaling to understand Wtmn induced apoptosis mechanism. In MCF-7 cells, Wtmn provoked a significant loss of MMP. These research analyzed intracellular compartment such as nuclear, mitochondrial and cytosolic changes associated with Wtmn induced apoptosis that caused the loss of MMP leads to activation of key effector protease. This appearance is positively correlated with the Wtmn concentration and apoptosis induction despite the absence of normal caspase-3 in the MCF-7 cells. Thus, these results support the mechanism that Wtmn could induce apoptosis in MCF-7 cells leads to disruption of MMP and expression of the caspase - 7 cascades. Based on published reports [14,30], and the findings presented here we proposed critical cellular and molecular events necessary for Wtmn treated MCF-7 cells apoptosis induction (Figure 3).
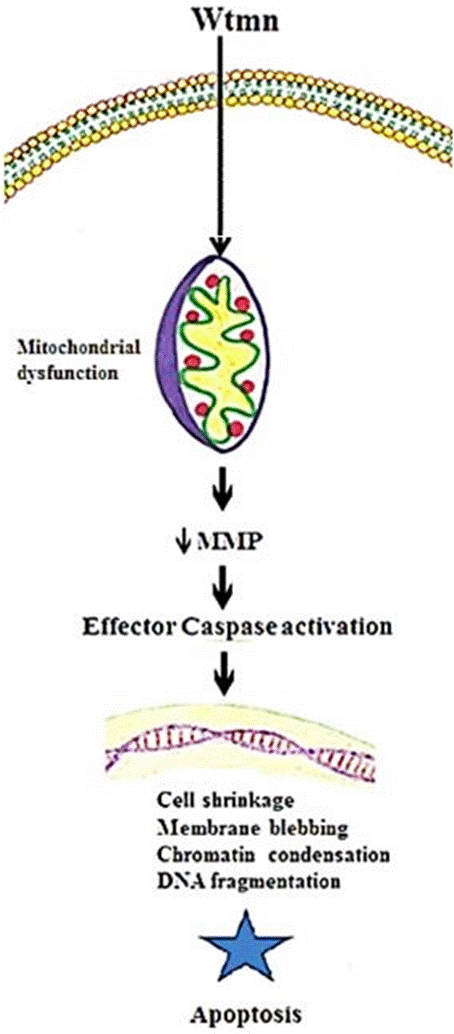
Figure 3. Key molecular mechanisms of Wtmn treated MCF-7 breast cancer cell death.
Our results suggesting that caspase-dependent intrinsic apoptotic pathways were involved in Wtmn induced MCF-7 cell death. These results are consistent with the previous studies that Wtmn induces programmed cell death in different cells [13,30,33]. Our results indicate that the disruption of MMP as well as activation of effector caspase - 7, all are indication of apoptosis signaling mechanism involvement.
5. Conclusions
Taken together, these results demonstrated for the first time insight into the mitochondrial dependent intrinsic cell death pathway induced by Wtmn in MCF-7 cells through morphological and biochemical alterations of mitochondrial and cellular functions. It follows the apoptotic phases such as cell shrinkage, loss of MMP, chromatin condensation, membrane blebbing, apoptotic body formation and activation of caspase cascade. Therefore, all these results suggested the intrinsic apoptotic cell death pathways are involved in Wtmn induced MCF-7 breast cancer cells. These results suggest a novel biomolecular target pathway-apoptosis induction by Wtmn. This understanding could be used to improve the potential anti-cancer properties of the breast cancer therapeutics.
References
- Li, Y., Zhang L., Li, K., Li, J., Xiang, R., Zhang, J., Li, H., Xu, Y., Wei, Y., Gao J., and Lin, P., and Wei, Y. 2015. ZNF32 inhibits autophagy through the mTOR pathway and protects MCF-7 cells from stimulus-induced cell death. Scientific Reports 5, Article number: 9288 (2015), doi: 10.1038/srep09288.
- Jia, T., Zhang, L., Duan, Y., Zhang, M., Wang, G., Zhang, J. and Zhao, Z., 2014. The differential susceptibilities of MCF-7 and MDA-MB-231 cells to the cytotoxic effects of curcumin are associated with the PI3K/Akt-SKP2-Cip/Kips pathway. Cancer Cell International. 14:126.DOI: 10.1186/s12935-014-0126-4.
- Wymann MP, Bulgarelli-Leva G, Zvelebil MJ. 1996. Wortmannin inactivates phosphoinositide 3-kinase by covalent modification of Lys-802, a residue involved in the phosphate transfer reaction. Mol Cell Biol.; 16(4):1722-33.
- Schultz RM., Merriman RL. Andis SL. 1995. In vitro and in vivo antitumor activity of the phosphatidylinositol-3-kinase inhibitor, wortmannin. Anticancer Res; 15(4):1135-9.
- Davol PA, Bizuneh A, Frackelton AR Jr. 1999. Wortmannin, a phosphoinositide 3kinase inhibitor, selectively enhances cytotoxicity of receptor-directed-toxin chimeras in vitro and in vivo. Anticancer Res.; 19(3A): 1705-13.
- Price BD Price BD, Youmell MB. 1996. The phosphatidylinositol 3-kinase inhibitor wortmannin sensitizes murine fibroblasts and human tumor cells to radiation and blocks induction of p53 following DNA damage. Cancer Res; 56(2):246-50.
- Lemke LE, Paine-Murrieta GD, Taylor CW, Powis G. 1999. Wortmannin inhibits the growth of mammary tumors despite the existence of a novel wortmannin-insensitive phosphatidylinositol-3-kinase. Cancer Chemo ther Pharmacol; 44 (6): 491-7.
- Mattiasson G. 2004. Flow cytometric analysis of isolated liver mitochondria to detect changes relevant to cell death. Cytometry A; 60(2):145-54.
- Hossain, M. Z, Khudhayer, W.J., Akter, R., Karabacak, T., and Kleve, M.G. 2016. Cytotoxic and anti-cancer effects of nickel nanowires against pancreatic cancer cells. Materials Research Society Proceedings Volume 1416-2012.
- Punj, V. and Chakrabarty, A. M. 2003. Redox proteins in mammalian cell death: an evolutionarily conserved function in mitochondria and prokaryotes. Cellular Microbiology; 5: 225–231.
- Akter R, Hossain MZ, Kleve MG, and Gealt MA. 2012. Wortmannin induces MCF-7 breast cancer cell death via the apoptotic pathway, involving chromatin condensation, generation of reactive oxygen species, and membrane blebbing. Breast Cancer: Targets and Therapy. 4: 103–113.
- Muhamad S., Lope Pihie A. H., Latif J., Rha C, and Sambandan T.G. 2011. Induction of apoptosis in MCF-7 via the Caspase pathway by longilactone from Eurycoma longifolia Jack. Research in Pharmaceutical Biotechnology; 3(1): 1–1.
- Waxman DJ, Schwartz PS. 2003. Harnessing apoptosis for improve anticancer gene therapy. Cancer Res; 63: 8563-8572.
- Jeong SY, Seol DW. 2008. The role of mitochondria in apoptosis. BMB Rep;41(1):1122.
- Abu Faddan N, Sayed D, Ghaleb F. 2011. T lymphocytes apoptosis and mitochondrial membrane potential in Down's syndrome. Fetal Pediatr Pathol. Egypt J Pediatr Allergy Immunol. 8(1):35-40.
- Sun L, Chen T, Wang X, Chen Y, Wei X. 2011. Bufalin Induces Reactive Oxygen Species Dependent Bax Translocation and Apoptosis in ASTC-a-1 Cells. Evid Based Complement Alternat Med; 249-090.
- Lopez-Mediavillaa C., Orfaob A., Gonzaleza M., Medinaa JM. 1989. Identification by flowcytometry of two distinct rhodamine-123-stained mitochondrial populations in rat liver FEBS letter;254 (1–2):115–120.
- Shim HY, Park JH, Paik HD, Nah SY, Kim DS, Han YS. 2007. Acacetin-induced apoptosis of human breast cancer MCF-7 cells involves caspase cascade, mitochondriamediated death signaling and SAPK/JNK1/2-c-Jun activation. Mol Cells;24(1):95-104.
- Ding, W. X., Shen, H. M., and Ong, C. N. 2000. Critical role of reactive oxygen species and mitochondrial permeability transition in microcystin-induced rapid apoptosis in rat hepatocytes. Hepatology 32, 547−555.
- Schrivastava A., Tiwari M., Sinha RA., Kumar A., Balapure AK.. 2006. Molecular iodine induces caspase independent apoptosis in human breast carcinoma cells involving mitochondria-mediated pathway. J. Biol. Chem; 281:19762−19771.
- Chan WH, Wu HJ, Shiao NH. 2007. Apoptotic signaling in methylglyoxal-treated human osteoblasts involves oxidative stress, c-Jun N-terminal kinase, caspase-3, and p21activated kinase 2. J Cell Biochem.;100(4):1056-69.
- Jänicke RU. 2009. MCF-7 breast carcinoma cells do not express caspase-3. Breast Cancer Research and Treatment; 117 (1): 219–221.
- Chua YL, Zhang D, Boelsterli U, Moore PK, Whiteman M, Armstrong JS. 2005. Oltipraz-induced phase 2 enzyme response conserved in cells lacking mitochondrial DNA. Biochem Biophys Res Commun.337(1):375-81.
- Armstrong JS, Steinauer KK, Hornung B, Irish JM, Lecane P, Birrell GW, Peehl DM, Knox SJ. 2002. Role of glutathione depletion and reactive oxygen species generation in apoptotic signaling in a human B lymphoma cell line. Cell Death Differ. 9(3):252-63.
- Grivicich, I., Regner, A., da Rocha, A. B., Grass, L. B., Alves, P. A. 2005. Irinotecan/5fluorouracil combination induces alterations in mitochondrial membrane potential and caspases on colon cancer cell lines. Oncol. Res; 15, 385−392.
- Kallio, A., Zheng, A., Dahllund, J., Heiskanen, K. M., and Harkonen, P. 2005. Role of mitochondria in tamoxifen-induced rapid death of MCF-7 breast cancer cells. Apoptosis 10; 1395−1410.
- Roy A., Ganguly A, Dasgupta B., Das, Pal C, Jaisankar P. and Majumder HK. 2008. Mitochondria-Dependent Reactive Oxygen Species-Mediated Programmed Cell Death Induced by 3,3′-Diindolylmethane through Inhibition of F0F1-ATP Synthase in Unicellular Protozoan Parasite Leishmania donovani.Molecular Pharmacology 74(5): 1292-1307.
- Gottlieb RA. 2001. Mitochondria and apoptosis. Biol Signals Recept 10: 147-161.
- Mehta A and Shaha C. 2004. Apoptotic death in Leishmania donovani promastigotes in response to respiratory chain inhibition: complex II inhibition results in increased pentamidine cytotoxicity. J Biol Chem 279: 11798-11813.
- Hossain M. Z. and Kleve M. G. 2011. Nickel nanowires induced and reactive oxygen species mediated apoptosis in human pancreatic adenocarcinoma cells. International Journal of Nanomedicine, 6: 1475–1485.
- Shi Y-Q., Blattmann.H., Crompton NE. 2000 Wortmannin selectively enhances radiation-induced apoptosis in proliferative but not quiescent cells. International Journal of Radiation Oncology; 49 (2): 421-425.
- Wang X., Wu Q., Zhang L., Wu Y., Shu Y. 2010. Wortmannin induced apoptosis of leukemia cells by reducing PI3K/Akt. The Chinese-German Journal of Clinical Oncology; 9(12): 734–738.
- Seol JW, Lee YJ, Kang HS, Kim IS, Kim NS, Kwak YG, Kim TH, Seol DW and Park SY. 2005. Wortmannin elevates tumor necrosis factor-related apoptosis-inducing ligand sensitivity in LNCaP cells through down-regulation of IAP-2 protein. Exp Oncol; 27(2):120-4.
- Wu Q, Chen Y, Guohui Cui, Cheng Y. 2009. Wortmannin inhibits K562 leukemic cells by regulating PI3k/Akt channel in vitro. Journal of Huazhong University of Science and Technology - Medical Sciences; 29: 451–456.



