Antimicrobial Activities of Some Leaves and Fruit Peels Hydrosols
Akwasi Acheampong*, Lawrence S. Borquaye, Samuel Osafo Acquaah, Jonathan Osei-Owusu, Gideon K. Tuani
Department of Chemistry, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
Abstract
The antimicrobial activity of hydrosols of the leaves and fruit peels of some plants have been assessed. The purpose of this study was to determine the antimicrobial potency of the hydrosols of leaves of Cymbopogon nardus, Chromolaena odorata, Ocimum gratissimum, Cymbopogon citratus, and fruit peels of Citrus sinensis and Citrus aurantifolia against Escherichia coli, Staphylococcus aureus, Candida albicans, Bacillus subtilis and Enterococcus faecalis. The hydrosols were obtained by hydrodistillation of the leaves and peels. The agar well-diffusion method was employed for the determination of antimicrobial activity. The results showed that the hydrosol from fresh fruit peels of Citrus aurantifolia exhibited antimicrobial activity against all the tested organisms. However, Hydrosols from dried Cymbopogon nardus leaves, fresh and dried Citrus sinensis fruit peels, dried Chromolaena odorata leaves, and fresh Cymbopogon citratus leaves exhibited no antimicrobial activity. The nature of the leaf and peel, fresh or dried, had impact on the antimicrobial activity. The results suggests that some of the hydrosols of the plants studied could be exploited for their antimicrobial activity against bacteria and fungi.
Keywords
Hydrosols, Essential Oils, Antimicrobial
Received: August 7, 2015
Accepted: August 20, 2015
Published online: August 27, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
The increase in the prevalence of side effects of many synthetic antimicrobial agents and incidence of multidrug resistant bacteria has spurred scientists on the research for plant based antimicrobial of therapeutic potentials (Reische et al., 1998). Essential oils and their by-products have been widely used in folkloric medicine and as such, they have been screened for their potential uses as alternative remedies for the treatment of infectious diseases (Afolayan et al., 2009).
Essential oils, by definition, are volatile oils that have aromatic components and that give plants their peculiar fragrance or flavour. They are often referred to as volatile plant secondary metabolites because they are by-products of plant metabolism. They are usually produced by steam distillation of aromatic plants and are used as fragrances, flavourings and as herbal medicines (Koul, 2008). Some essential oils have been reported to have insecticidal properties (Massebo et al., 2009). Some have also been reported to have antifungal, antiviral, antioxidant and antibacterial properties (Mmbengwa et al., 2009). Essential Oils from Cyclotichium niveum (Bioss) showed a strong antibacterial activity against Klebsiella pneumonia, and Staphylococcus aureus and also has a strong antifungal activity against Candida albicans (Alim et al., 2009). The quantity of essential oils found in most plants are usually 1 to 2%, however, they can have amounts ranging from 0.01 to 10% (Koul, 2008).
Hydrosols, floral water, distillate water, or aromatic water, are the co-products or by-products of steam distillation of plant material. They are quite complex mixtures containing traces of the essential oils and other water-soluble components of the plant. Usually, the plant is being distilled to produce an essential oil; however, some plants are processed only for the hydrosol. During distillation, steam and essential oil components are in intimate contact. On condensation, some essential oil compounds, the more water-soluble oxygenated polar compounds, dissolve in the aqueous phase and are therefore lost to the water distillate or hydrosol (Bohra et al., 1994). Hydrosols are scented, and usually have a pH of 4.5 to 5.5 (Schorr, 2004; Paolini et al., 2008). pH greatly influences the therapeutic effects of the hydrosols (Catty, 2001). Some hydrosols have been shown to have inhibitory effects on some bacteria (Faith et al., 2011; Hussien et al., 2011; Oral et al., 2008; AL-Turki, 2007; Dubey et al., 2011; Tornuk et al., 2011). It is also known that the compositions of hydrosols and their antimicrobial effects depend on plant species and regional conditions (Sağdç and Ozcan, 2003; Oral et al., 2008).
Antimicrobial properties of the essential oils from the leaves and fruit peels of the plants under study have been reported by other scientists (Onawunmi et al., 1984; Nakahara et al., 2003; Sharma and Triphathi, 2006; Fisher and Carol, 2008; Sukanya et al., 2009; Natheer et al., 2012). However, not much is reported of the antimicrobial properties of the hydrosols of the leaves and fruit peels of these plants. The objective of this study was, therefore, to evaluate the antimicrobial activity of the hydrosols of the fresh and dried leaves of Cymbopogon nardus (Citronella), Chromolaena odorata (Siam Weed), Ocimum gratissimum (Basil), Cymbopogon citratus (Lemon grass), and the fresh and dried fruit peels of Citrus sinensis (Orange) and Citrus aurantifolia (Lime) against E. coli, S. aureus, C. albicans, B. subtilis, E. faecalis.
2. Materials and Methods
2.1. Plant Samples
Plants for the work were obtained from the herbarium of the Department of Pharmacognosy and the Botanic gardens all of Kwame Nkrumah University of Science and Technology, Kumasi in Ghana. The samples were collected between the months of May and August 2011 for the distillation. The common and scientific names of the plants used in this study are shown in Table 1.
2.2. Preparation of Hydrosols
Between 80 and 450g of plant samples, fresh or air dried, were hydrodistilled using the Clevenger type apparatus for over 3 hours. Leaves from Cymbopogon nardus, Chromolaena odorata, Ocimum gratissimum and Cymbopogon citratus were used for the preparation, whilst peels from Citrus sinensis and Citrus aurantifolia were used.
2.3. Conductivity and PH of the Hydrosols
A Pc 300 cyber scan conductivity pH meter was used to determine the conductivity and pH of the hydrosols at room temperature (25°C). The instrument was calibrated using a buffer of pH 4, pH 7 and pH 11 before the determination. The results of the pH and conductivity of the hydrosols are shown in Table 2.
Table 1. Plants from which hydrosols were obtained.
| Scientific Name | Family | Common Name | Plant part used |
| Cymbopogon nardus | Poaceae | Citronella | Leaves |
| Chromolaena odorata | Asteraceae | Siam Weed | Leaves |
| Citrus sinensis | Rutaceae | Orange | Fruit Peels |
| Citrus aurantifolia | Rutaceae | Lime | Fruit Peels |
| Ocimum gratissimum | Lamiaceae | Basil | Leaves |
| Cymbopogon citratus | Poaceae | Lemon grass | Leaves |
Table 2. PH and conductivity of the plant hydrosols.
| Fresh | Dried | |||
| Plant Hydrosols | pH | Conductivity (µS) | pH | Conductivity (µS) |
| C. aurantifolia | 2.34 | 132.8 | 2.63 | 119.5 |
| O. gratissimum | 2.74 | 160.0 | 7.38 | 614.0 |
| C. citratus | 2.54 | 233.0 | 2.41 | 730.0 |
| C. nardus | 3.53 | 25.2 | 5.35 | 173.9 |
| C. sinensis | 3.69 | 27.8 | 3.24 | 62.1 |
| C. odorata | 2.46 | 106.1 | 2.19 | 80.6 |
2.4. Microbial Strains
The microbial strains which were used for the study were Candida albicans ATCC #10239, Bacillus subtilis NTCC #10073, Eschericia coli ATCC #25922, Enterococcus faecalis ATCC #29212 and Staphylococus aureus ATCC #25923. The bacterial strains, Bacillus subtilis, Eschericia coli, Enterococcus faecalis, and Staphylococus aureus were cultured overnight at 37°C in Mueller Hinton agar. The fungus was cultured for 72 hours.
2.5. Antimicrobial Screening of the Hydrosols
The agar well-diffusion method was employed. 20 ml of Nutrient Agar was melted on a water bath. The molten agar was then stabilized at 45°C for 15 minutes in a thermostatic water bath. The agar containing tubes were then inoculated separately with 0.1 ml of the cultured test organisms. The tubes were then rolled in between the palms to ensure thorough mixing. The contents were then poured into separate sterilized Petri dishes. The dishes were then allowed to set on a laminar horizontal flow chamber after which holes were punched in each dish using 10 mm diameter sterilized cork borer. The hydrosols were then poured into the labeled holes made in the dishes. The dishes were then allowed to pre-incubate for one hour before incubating them in a bacteriological incubator at a temperature of 37°C for a period of 24 hours. The inhibition zones were determined in triplicates and the antimicrobial activity was expressed as the mean of the inhibition zone (mm) produced by the hydrosols. The results of the antimicrobial screening of the hydrosols are shown in Table 3.
Table 3. Antimicrobial effect of the leaves and fruit peels hydrosols against E. coli, S. aureus, C. albicans, B. subtilis and E. faecalis (inhibition zone in mm).
| Hydrosol Source | E. Coli | S. aureus | C. albicans | B. subtilis | E. faecalis |
| C. nardus (dried leaves) | - | - | - | - | - |
| C. nardus (fresh leaves) | - | - | - | - | - |
| C. sinensis (dried peels) | - | - | - | - | - |
| C. sinensis (fresh peels) | - | - | - | - | - |
| C. aurantifolia (dried peels) | - | 1 | - | - | - |
| C. aurantifolia (fresh peels) | 4 | 5 | 2 | 4 | 1 |
| C. odorata (dried leaves) | - | - | - | - | - |
| C. odorata (fresh leaves) | - | 1 | - | 3 | - |
| C. citratus (dried leaves) | 3 | 3 | 2 | 6 | - |
| C. citratus (fresh leaves) | - | - | - | - | - |
| O. gratissimum (dried leaves) | 2 | 1 | 1 | - | 1 |
| O. gratissimum (fresh leaves) | 2 | 2 | 1.5 | 2 | - |
(-) no inhibition zone
3. Results and Discussions
3.1. pH and Conductivity of Hydrosols
In general, the hydrosols of the dried samples had greater conductivities than those of the fresh samples with the exception of C. odorata. There is no particular trend seen for the pH values of the hydrosols of the fresh and dried samples. As the hydrosols of the dried samples of C. aurantifolia, O. gratissimum, and C. nardus had higher pH values than the hydrosols of the fresh samples, the opposite is the case for C. citratus, C. sinensis, and C. odorata.
Hydrosols are either neutral or slightly acidic and the pH greatly influences the therapeutic effects. The pH values obtained for the plant hydrosols indicated that the hydrosols were acidic which are in agreement with Catty, 2001. There was an observable difference in pH and conductivity between the fresh plant hydrosol and that of the dried hydrosol of the same plant species which is indicative of difference in therapeutic effects. There was, however, no correlation between the pH and conductivity of the plants hydrosols as the R2 values for the hydrosols of the fresh and dried samples were respectively 0.6105 and 0.1135.
3.2. Antimicrobial Activity
The hydrosols from the plants studied exerted varying levels of antimicrobial effect against the five tested organisms. The results are presented in table 3.
It can be observed that, the hydrosols from dried and fresh leaves of C. nardus, dried and fresh peels of C. sinensis, dried leaves of C. odorata and fresh leaves of C. citratus were not able to inhibit any of the tested organisms. The hydrosol from dried leaves of C. aurantifolia was able to inhibit only the S. aureus. The hydrosol from fresh leaves of C. odorata was also able to inhibit S. aureus and B. subtilis but not the rest. The hydrosol from fresh fruit peels of C. aurantifolia was the only hydrosol able to inhibit all the tested organisms. The hydrosol from dried leaves of C. citratus was able to inhibit all the tested organisms except E. faecalis. The hydrosol from dried leaves of O. gratissimum was able to inhibit all the tested organisms except B. subtilis. Also, with the exception of E. faecalis, the hydrosol from fresh leaves of O. gratissimum was able to inhibit all the tested organisms.
It can also be seen that there are differences in antimicrobial activity between hydrosols of fresh and dried samples of the same plant. For instance the hydrosol from fresh peels of C. aurantifolia was able to inhibit the entire tested organisms compared to the dried C. aurantifolia which was able to inhibit only S. aureus. The non-volatile chemical group, primarily hydrophilic acidic constituents, reported to occur in hydrosols could contribute to the inhibitory effect and could possibly be in synergy with the trace essential oil components.
Hydrosol of fresh peels of C. aurantifolia showed the highest antimicrobial activity against four of the tested organisms, i.e. E. coli, S. aureus, C. albicans and E. faecalis. This high antimicrobial activity could partly be due to higher content of phenols and flavonoids in this hydrosol. It was, however, the hydrosol of dried leaves of C. citratus that showed the highest antimicrobial activity against B. subtilis. The most susceptible of all the microbes was S. aureus as six of the hydrosols were able to inhibit its growth to various extents. However, E. faecalis was the least susceptible as only two of the hydrosols were able to inhibit it to a minimal extent.
The results also revealed that hydrosols that showed activity against the Gram negative E. coli were able to show antimicrobial activity against the fungus (C. albicans) and most of the gram positive strains (B. subtilis, S. aureus and E. faecalis). However, not all the hydrosols that inhibited the gram positive strains were able to inhibit the gram negative E. coli.
AL-Turki (2007) in his work indicated that hydrosols of thyme, peppermint, black pepper and garlic exhibit antibacterial activity against B. subtilis and S. enteritidis. Also Nascimento et al. (2000) investigated inhibitory effect of plant extracts on antibiotic resistant bacteria and reported that thyme (Thymus vulgaris), clove (Caryophyllus aromaticus) and basil (Ocimum basilicum) were effective against P. aeruginosa but they did not affect E. coli. It has been demonstrated that the antimicrobial effects of the essential oils act by causing structural and functional damages to the bacterial cell membrane (Goni et al., 2009)
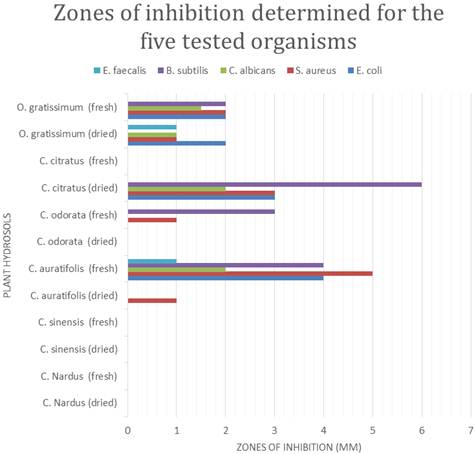
Figure 1. Zones of inhibition for the five selected organisms determined for the hydrosols.
4. Conclusion
The plant hydrosols of the leaves and fruit peels exhibited varying degrees of antimicrobial activity against the five tested microbes, E. faecalis, B. subtilis, C. albicans, S. aureus and E. coli. Hydrosol from fresh leaves of C. aurantifolia was the only one which inhibited the entire tested organisms. Fresh and dried leaves hydrosols of the same plant exhibited different antimicrobial activity against the tested microbes. Also the fresh and dried fruit peels hydrosols of the same plant exhibited different antimicrobial activities against the tested microbes. Hydrosols of both fresh and dried leaves and peels of some of the plants exhibited no antimicrobial activity. Hence not all hydrosols of leaves and fruit peels of plants may exhibit antimicrobial activity. The hydrosols, which are usually in higher quantity than essential oils after hydrodistillation, can be exploited for their antimicrobial activity against some Gram negative and Gram positive bacteria as well as some fungi.
References
- Afolayan J, and Ashafa A O T (2009). Chemical composition and antimicrobial activity of the essential oil from Chrysocoma ciliata L. leaves. Journal of Medicinal Plants Research 3(5): 390-394.
- Alim A, Goze I, Cetin A, Atas A D, Vural N and Donmez E (2009). Antimicrobial activity of the essential oil of Cyclotrichium niveum (Bioss) Manden. Et Scheng. African Journal of Microbiology Research 3(8): 422-425.
- AL-Turki A I (2007). Antibacterial effect of thyme, peppermint, sage, black pepper and garlic hydrosols against Bacillus subtilis and Salmonella enteritidis. Journal of Food, Agriculture & Environment 5 (2): 92-94.
- Bohra P, Vaze A S, Pangarker V G et al. (1994). Adsorptive recovery of water soluble essential oil components. J Chem Technol Biotechnol. 60(1): 97-102
- Catty S (2001). Hydrosols: The next aromatherapy. Vermont: Healing Arts Press
- Dubey M, Sharma S, Sengar R, Bhadauria S, Gautam R K (2011). In vitro antibacterial activity of Lantana camara leaves hydrosol. Journal of Pharmacy Research 4(11): 3972.
- Fisher K and Carol P (2008). Potential antimicrobial uses of essential oils in food: is citrus the answer. Trends in Food Science and Technology 19(3): 156-164.
- Goni P, Lopez P, Sanchez C, Gomez-Lus R, Becerril R, and Nerin C (2009). Antimicrobial activity in the vapour phase of a combination of cinnamon clove essential oils. Food Chemistry 116: 982-989.
- Hussien J., Teshale C. and Mohammed J (2011). Assessment of the Antimicrobial Effects of Some Ethiopian Aromatic Spice and Herb Hydrosols. International Journal of Pharmacology, 7: 635-640.
- Koul O (2008). Phytochemicals and insect control: Antifeedant approach. Crit. Rev. Plant Sci. 27: 1-24.
- Massebo F, Tadesse M, Bekele T, M and Gebre-Michael T (2009).Evaluation on larvicidal effects of essential oils of some local plants against Anopheles arabiensis Patton and Aedes aegypti Linnaeus (Diptera, Culicidae) in Ethiopia. African Journal of Biotechology 8 (17): 4183-4188.
- Mmbengwa V, Samie A, Gundidza M, Matikiti V, Ramalivhana N J, Magwa M L (2009). Biological Activity And Phytoconstituents Of Essential Oil From Fresh Leaves Of Eriosema englerianum. African Journal of Biotechnology 8 (3):361-364.
- Nakahara K, Alzoreky N S, Yoshihashi T, Nguyen H T T, Trakoontivakorn G (2003). Chemical composition and antifungal activity of essential oils from Cymbopogon nardus. Japan Agricultural Research Quarterly 37(4): 249 – 252.
- Nascimento G G F, Locatelli J, Feitas P C, Silva G L (2000). Antibacterial activity of plant extracts and phytochemicals on antibiotic-resistant bacteria. Braz J. Microbiol. 31: 247-256.
- Natheer S E, Sekar C, Amutharaj P, Rahman M S A, & Khan K F (2012). Evaluation of antibacterial activity of Morinda citrifolia, Vitex trifolia and Chromolaena odorata. African Journal of Pharmacy and Pharmacology 6(11): 783-788.
- Onawunmi G O, Yisak W A, Ogunlana E O (1984). Antibacterial constituents in the essential oil of Cymbopogon citratus. Journal of Ethnopharmacology 12(3): 279-286.
- Oral N, Gülmez M, Vatansever L, Güven A (2008). Application of antimicrobial ice for extending shelf life of fish. J. Food Prot. 71 (1): 218-222.
- Paolini J, Leandri C, Desjobert J-M, Barboni T, Costa J (2008). Comparison of liquid-liquid extraction with headspace methods for the characterization of volatile fractions of commercial hydrolats from typically Mediterranean species. J. Chromatogr. A. 1193: 37-49.
- Reische D W, Lillard D A, Eitenmiller R R (1998). Antioxidants in food lipids. In Ahoh C C and Min D B eds., Chemistry, Nutrition and Biotechnology. New York, Marcel Dekker, pp. 423-448.
- Sağdç O, Özcan M (2003). Antibacterial activity of Turkish spice hydrosols. Food Cont. 14: 141-143.
- Schorr S M (2004). Bioresonance and phytotherapeutic hydrosols in healing. Bioponic Phytoceuticals P.O.Box 2120, Kihei, HI 96753. http://www.bioponic.com/pdfs/Bioresonance.pdf. pp. 1-20. (27/06/2011).
- Sharma N, Triphathy A (2006). Fungitoxicity of the essential oil of Citrus sinensis on post-harvest pathogens. World Journal of Microbiology and Biotechnology 22(6): 587-593.
- Sukanya S L, Sudisha J, Hariprasad P, Niranjana, S R, Prakash H S, & Fathima S K (2009). Antimicrobial activity of leaf extracts of Indian medicinal plants against clinical and phytopathogenic bacteria. African Journal of Biotechnology, 8(23): 6677-6682.
- Tornuk F, Cankurt H, Ozturk I and Sagdic O (2011). Efficacy of various plant hydrosols as natural food sanitizers in reducing Escherichia coli and Salmonella typhimurium on fresh cut carrots and apples. International Journal of Food Microbiology 148 (1): 30-35.



