Evaluation Using Fourier Transformed-Infrared Spectroscopy (FT-IR) of Biodegradation by the Strain Trichoderma Viride M5-2 from the Cell Walls of Sugarcane (Saccharum Officinarum Lin) Bagasse Pretreated
Elaine Valiño1,*, A. Elías1, Marlen Rodríguez2, Nereyda Albelo1
1Institute of Animal Science, Physiology Department, Mayabeque, Cuba
2Institute of Materials and Reactive (IMRE), University of Havana, La Habana, Cuba
Abstract
The objective was to prove the molecular change produced in the sugarcane bagasse with pretreated calcium hydroxide and fermented with Trichoderma viride M5-2 strain by means of Fourier Transform Infrared Spectroscopy (FTIR).The fungi fermentation was conducted in a solid state bioreactor. For the spectroscopy analysis by FTIR, the samples were taken from sugarcane bagasse, sugarcane bagasse hydrolyzed with Ca (OH)2 and fermented at a depth of 10 cm in the bioreactor at 96 h, when the highest cellulolytic activity of exo ß1-4 glucanase (2.8 UI/ml) was reached.The spectrum of the intact sugar cane bagasse and the bagasse hydrolyzed with Ca (OH)2 differed in the range 2000-1400±20 cm-1of transmittance. Moreover, differences were found in the sugarcane layer during the fermentation. In the fermented sample spectrum, new bands were visualized in the range 774.30±20 cm-1and 1670-1780±20 cm-1, when obtaining increases in carbonyl, carboxyl and phenolic groups derived from the breaking of the subunits of lignin and structural carbohydrate. The biodegradative effect of the Trichoderma viride M5-2 strain sin the pretreated sugar cane bagasse with Ca (OH)2 was proved by means of the FTIR technique.The production of the endo and exo β 1-4 glucanase enzyme treated with calcium hydroxide bagasse improved. The cellulases produced during the conversion from cellulose to glucose by the fungal mutant strain depend directly on the treatment.
Keywords
Infrared Spectroscopy, Trichodema viride, Sugar Cane Bagasse, Solid State Fermentation
Received: July 11, 2015
Accepted: August 9, 2015
Published online: August 19, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Crop that accumulated lignocellulolytic residues in high scale include rice, corn, wheat, citric, coffee, soybean, sugar cane amongst others. Other authors refer to the chemical composition to emphasize the low percentage in protein and high level in fiber, (1, 2).
Sugarcane byproducts of low biological value like bagasse and bagasse pith filter caked mud where the crop residues were under used in animal feeding due to the following limitations: they are of a lignocellulosic nature, low content of assimilable nitrogen, low digestibility of nitrogen-free extract and its poor palatability. In addition, different methods to increase the nutritional value of fibrous materials are still unknown (3). It should be highlighted that the use of microorganisms is capable of degrading cellulose and lignin in order to obtain the product for industrial use, as well as animal nutrition.
Due to the crystalline capillary structure of the cellulose, the lignocellulosic material to be presented is of a very resistant structure. Similar to the polymerization grade and the lignin wall surrounding it, the pretreated can be used with the objective of breaking the structure and the microorganism that limits the enzyme action.This is classified in physical (5, 6), chemical (7, 8) and biological (9, 10) and can be applied in sequential or simultaneous combinations. This method achieves the following objectives: decrease crystalline of cellulose index and the lignin content, similar to the separation from substrate the lignin complex (11).
The cellulose hydrolysis in monomers of glucose occurs under the action of an enzyme complex acting synergically and represented in endo, exo β 1-4 glucanases, β glucosidases (12) and a great variety to degrade lignin and the hemicelluloses (2). In order to guarantee successful development of enzyme productivity and yield in the lignocellulosic substrate, solid state fermentation was used for its advantages (13, 14). Also, the conventional methods of analyzing the feed’s chemical composition such as fiber fractioning, crude protein, aminoacids, organic matter, ether extract and others were used to determine the degradation degree, and to predict the nutritional and energy value, according to (15).These methods do not detect neither the intrinsic structures of the feed or the components of the biological matrix (16).
The infrared spectroscopy is a not an invasive technique that provides all the information needed for modification at a molecular level in the wall components produced by microbial activity (17).
According to (18), these studies show that infrared spectroscopy (IR) is very complex, however the useful information provided about molecular change for the action of the produced fungi offers several advantages among which are the velocity of data collection and samples analysis without solubility treatment and without extraction.
Considering that some conidial fungi, such as Trichoderma viride M5-2, have the ability of partially degrading the lignin and hydrolyzing the cellulose (13), the objective of this study was to prove the molecular change in sugar cane bagasse fermented with the strain of mutant fungus Trichoderma viride M5-2 that were pretreated with calcium hydroxide using Fourier Transformed-infrared spectroscopy (FT-IR).
2. Material and Methods
2.1. Microorganism
A mutant strain of the lignocellulolytic fungus Trichoderma viride M5-2 belonging to the strain bank of the Institute of Animal Science was used. This strain produces phenoloxydases, cellulases enzymes and hydrolytic activity in highly fibrous substrates when assessed through solid state fermentation (13).
2.2. Inoculum
We used the isolated conidial fungus Trichoderma viride M5-2 and identified in the Biotechnology laboratory at the Animal Science Institute in cooperation with the University of Havana’s Mycology Laboratory of the Faculty of Biology. Inoculum spore suspensions were prepared by dragging the microorganism’s previously sown wedges malt agar (Oxoid). This was done using a buffered diluent containing 0.3 g of calcium chloride, 0.75 g proteose peptone, 0.2 ml Tween 80 and 1 mL solution of trace elements according (19). The resulting suspension was filtered through glass wool, with the aim of obtaining a concentration of 5x107spores/ml mycelium free. The final spore concentration was determined in a Neubauer chamber.
2.3. Substrate for Fermentation
The substrate was sugar cane bagasse from "Manuel Martínez Prieto" sugarcane mill in Havana City, was hydrolyzated with Ca (OH)2, to 5% (dry base), and underwent 0,18 MPa of pressure in autoclave for one hour, then washed with distillated water to eliminate alkali excess.
2.4. Chemical and Enzymatic Analysis
Five grams of the fermented solid material were taken at 0, 24, 48, 72, 96, 120 and 168 h, 45 ml of distilled water were added, then agitated in a sieve at 150 rpm for 30 min and filtered to obtain the enzymatic extract, of which the pH was measured and the corresponding enzymatic analyses were conducted.
The enzymes determined were:
Activity endo 1, 4 ß glucanase (CMCase) showing the hydrolytic activity over the carboxymetylcellulose.
Activity exo 1, 4 ß glucanase (PFase) or cellulase paper filters showing hydrolytic activity in the presence of crystalline cellulose.
The activities endo 1, 4 ß- glucanase and exo 1, 4 ß glucanase were determined, calculated and expressed in International Units per dried matter from started substrate (IU/gDM). This activity refers the glucose micromoles released per reaction minute under test conditions of activity (19).
2.5. Bromatological Analysis
The indicators studied were: dry matter (DM), according to (20), lignin (LIG), cellulose (CEL) in agreement with (21) and true protein (TP) by (22). The statistical system INFOSTAD was used for bromatological and enzymatic analysis (23). Differences between means were established, as (24) Duncan (1955).
2.6. Experimental Procedure
The bioreactor was charged at the beginning with 1 Kg of hydrolyzed sugar cane bagasse at 70 % humidity. The aeration being subministrated to reason of 15 L/min.kg of dry solid (11 L/min), was controlled between these values and 22 L/min.kg of dry solid (18 L/min). The complete process was controlled through the regulation of air flow and recirculation of water temperature; 20 g of hydrolyzed sugar cane bagasse (10 cm deep) in solid state fermentation were obtained. The samples were dried in heater at 40ºC and 0.1-0.3 mm size particles obtained in the spectroscopic analysis were utilized for high resolution.
2.7. Spectroscopic Analysis FT-IR
The samples were the following:
Sugar cane bagasse, hydrolyzed sugar cane bagasse with Ca (OH)2 to 5%, hydrolyzed sugar cane bagasse with Ca (OH)2 to 5% and bioreactor fermentation of the strain Trichoderma . viride M5-2 .
For spectroscopic analysis FT-IR to choose bioreactor fermentated samples for 96 hrs at 10cm deep, where the optimum exo ß1-4glucanase enzyme were produced.
Analysis by Fourier Transformed-infrared spectroscopy (FT-IR): the solid fermented samples were selected for the FT-IR analysis, reaching maximum cellulolytic activity. The equipment used was an ATI Mattson Genesis Series FT-IR spectrometer, with a special device for solid samples, adjusted to total attenuated reflectance in pieces of potassium bromide (KBr), which allowed measuring the IR spectrum in the solid samples without previous preparation.The spectra were obtained in transmittance form, from a wave longitude range of 4000-600 cm-1averaging 100 impulses above particles (ʎ/cm).
3. Results and Discussion
Figure 1 shows the corresponding spectrum of the bagasse sample and figure 2 shows the hydrolyzed bagasse; the signal assignment was obtained from literature dates (Table 1) according to (25) from the spectroscopic FT-IR analysis of biodegradation by the Trichoderma viride M5- 2 strain in the pretreated bagasse sugar cane cell wall. Qualitative information of molecular level in the different functional chemical groups was obtained using the total attenuated reflectance technique.
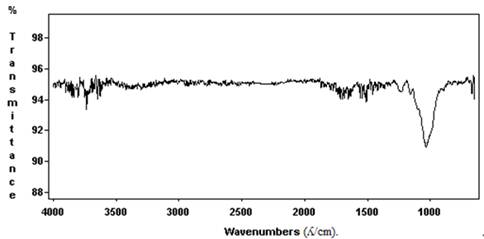
Figure 1. Infrared spectroscopy (FT-IR) of sugar cane bagasse, averaging 100 impulses on the particles (l /cm).
Table 1. Signal assignment FT-IR of the spectrum bands of woods according to Gilardi et al., (1995).
| Bands position (cm-1) | Funtional group |
| 1670- 1650 | C=C Alkene |
| 1265- 1235 | O-H Phenolics |
| 1600- 1500 | C=C Aromatics |
| 2960- 2850 | O-H Methyl and Methylene groups |
| 1780- 1670 | C=O Carbonyl |
| 3600- 3200 | O-H Alcohol |
| 1200- 800 | O-H Alcohol (primary and secondary) and aliphatic ether. |
| 910 | C=C Alkene |
Due to the complex structure of bagasse, overlaying bands appear in the cellular walls, even when the treatment for the lignin from hydrolysis fiber bagasse with Ca (OH)2 was performed. The results were not drastic, but the difference is evident between bagasse cane and the hydrolysis with Ca (OH)2, (figure 2) in the range of 2000-1400 cm-1, in this last case the bands were more open. The kinetic study carried out with fermented bagasse for 96 h to indicate endo and exo β 1-4 glucanase activity of 47.3 and 26.91 UI/gDM (figure 4). The soluble protein showed an increase (P˂0.001) concerning the hydrolysis bagasse control without fermentation of 6.5 mg/gDM and the true protein was 10.53% which represents 84% of the crude protein (P˂0.001).
Many authors have used this pre-treatment as a step previous to the enzymatic hydrolysis (26), for obtaining an effective action of endo and exo β 1,4glucanases enzyme, (27) in bagasse treatment with NaOH resulting in sustainable production of endo β 1,4glucanases of 28.84U/g with Aspergillus nidulans strain and confirmed structural changes in the bagasse using different techniques like FT-IR, which strongly depended on the pretreatment.
When Jayapal et al. (28) combined the sodium hydroxide and potassium hydroxide for recovering xylooligossacaride in sugar cane, the analysis of enzymatic activity showed thissugar with a polymerization grade variation. However, when Alves et al. (29) performed the pretreatment with sulphuric acid, in the FT-IR spectrum, it did not reveal significant changes in the chemical structure of the cellulose, thus an effective treatment should be stronger. However the topochemical characterization of sugar cane pretreated with alkaline sulphite for (30), indicated that surface lignin was dissolved during the pretreatment while surface hemicelluloses were exposed or relocated.

Figure 2. Infrared spectroscopy (FT-IR) of sugar cane bagasse pretreated with Ca (OH)2, averaging 100 impulses on the particles (l /cm).

Figure 3. Infrared spectroscopy (FT-IR) of sugar cane bagasse pretreated with Ca (OH)2 and fermentated by Trichoderma viride M5-2 strain at 96 hr, averaging 100 impulses on the particles (l /cm).

Figure 4. Cellulase production kinetics in the of sugar cane bagasse pretreated with Ca (OH)2 (5%) and fermentation by Trichoderma viride M5-2 in solid state fermentation bioreactor.
When these spectrums are compared (figure 1 y 2) with the solid state fermented bagasse in a bioreactor in (figure 3), less intensity in the vibration bands of benzene g (C=CH) is observed in a range of 670±20 cm-1and a new intensity band of 774.30±20 cm-1 appeared where the mono, di and tri aromatic rings were found replaced by the OH group or methyl groups, derivate from benzene which supposes a hydrolytic action of the fungi over the independent fiber of the chemical hydrolysis.
The bands corresponding to the carbohydrates and aliphatic ethers appear in all the spectrums, nevertheless, in the fermented samples they are wider and of higher intensity, which could be related to hydrolytic action of the fungi over the bagasse, when decreasing 6.05 percent unit of cellulose (p˂0.001) (Figure 3).
According to Valiño et al. (13) the chemical analysis of the fermentation to the product showed a fiber decrease of around 5%, while the neutral fiber decreased 14.9 percent per unit in correspondence with the amount of hemicelluloses used (p˂0.001).The result is of importance in determining the nutritive value of animal feed in order to increase nutrient bioavailability by acting on feed components prior to or after consumption (31).
The decrease of acid detergent fiber did not allow detection of variations in the lignin percentage as it is observed in bands of 1670-1780±20 cm-1without much intensity, which corresponds to the carbonyls groups as a result of carbohydrate degradation in the lignin (Figure 3). However, higher fiber digestibility is obtained by 20.98 percent per unit with regard to intact bagasse (P˂0.001).
Qin et al.(32) used tree decay white fungi in bagasse cane and found more effective the degradation in the lignin than in the cellulose and hemicelluloses (85-93%), as well as when using the FT-IR technique for the decay brown (33), although exo β glucanase of 130.45 UI/ml values are considered in this fungi. This could be a result of the high time of fermentation used to perform the analysis, as well as the use of Basidiomycetes, which metabolize lignin more efficiently than other fungi (2,34) as is the case of the results obtained with this fungus conidial.
The FT-IR bagasse data correspond with the chemical analysis of the fermented samples for T. viride M5-2 when a 3% decrease of lignin in fermented bagasse was observed and a 4% decrease in the cellulose of dry matter (Figure 5). This information can be useful in the design of different enzymes mixtures to be used.
The obtained results in the bioreactor indicated that varying changes in the lignin degradation with conidial fungi may be obtained, according to differences in experimental conditions. This may be possible due to many factors like lignolytic nitrogen concentration due to the dry matter of the fibrous substrate during the fermentation (35). Additionally, the decrease of other cellular constituents that make possible improved digestibility could increase or decrease lignin concentration (36,37).
Many authors consider that the method for determining the insoluble acid fiber may not be appropriate for biotransformed substrate by microorganism (16, 38) considering 72% of the lignin’s solubility in sulphuric acid.
The FT-IR technique demonstrated that during the fermentation with the strain of conidial fungi in sugar cane bagasse, oxidations of the aromatic structures of lignin occur, leading to an increase in phenolic groups and breaking the formation of carboxylic, carbonyl and alkenes groups, as well as alcoholic, carbonyl and carboxylic groups derived from the biodegradation of carbohydrates that cannot be detected via this process.

Figure 5. Lignin and cellulose bagasse sugar cane fresh, hydrolyzed with Ca (OH) 2 (5%) and fermented with the mutant fungus Trichoderma viride M5-2.
4. Conclusions
The biodegradative effect of the Trichoderma viride M5-2 strains in the sugar cane bagasse pretreated with Ca (OH)2 by means of the Fourier Transformed-infrared spectroscopy technique was proven. The production of the endo and exo β 1-4 glucanase enzyme treated with calcium hydroxide improved the bagasse. The cellulases produced during the conversion from cellulose to glucose by the fungus mutant strain depend directly on the treatment.
References
- G.D. Singh, H.O. Singh, S. Kaur, S.L. Bansal, S.B. Kaur Value-addition of agricultural wastes for augmented cellulase and xylanase production through solid-state tray fermentation employing mixed-culture of fungi, Industrial Crops and Products, 2011, 34(1): 1160-1167.
- J.S. Van Dyk and B.I. Pletschke, A review of lignocelluloses bioconversion using enzymatic hydrolysis and synergistic cooperation between enzymes factor affecting enzymes, conversion and synergy, Biotechnology Advances, 2012, 30:1458-1480.
- R.Z. Salimi and H. Estefahani, Evaluation of the effect of particle size, aeration rate and harvest time on the production of cellulase by Trichoderma reesei QM9414 using response surface methodology. Food and Bioproducts Processing, 2010, 88(1):61-66.
- N. Bansal, R. Tewari, R. Soni and S. Kumar, Production of cellulases from Aspergillus niger NS-2 in solid state fermentation on agricultural and kitchen waste residues, Waste Management, 2012, 32(7):1341-1346.
- R. Singh, A.J. Varma, R.L Seeta and M. Rao, Hydrolysis of cellulose derived from steam exploded bagasse by Penicillium cellulases: Comparison with commercial cellulase. Bioresource Technology, 2009, 100 (24): 6679-6681.
- L.W.Yuan, Y.W. Pei and F Lu, Screening and Identification of Cellulase-Producing Strain of Fusarium Oxysporum, Procedia Environmental Sciences, 2012, 12 (Part B): 1213-1219.
- A. Hideno, H. Inoue, K. Tsukahara, S. Yano, X. Fang, T. Endo and S. Sawayama Production and characterization of cellulases and hemicellulases by Acremonium cellulolyticus using rice straw subjected to various pretreatments as the carbon source, Enzyme and Microbial Technology, 2011, 48 (2, 8): 162-168.
- L.H.M. da Silva, S.C. Rabelo, A. da C. Carvalho, Effects of the pretreatment method on high solids enzymatic hydrolysis and ethanol fermentation of the cellulosic fraction of sugarcane bagasse, Bioresource Technology, 2015, 191, 312-321.
- S.J.A. van Kuijk, A.S.M. Sonnenberg, J.J.P. Baars, W.H. Hendriks and J.W. Cone, Fungal treated lignocellulosic biomass as ruminant feed ingredient: A review, Biotechnology Advances, 2015, 33(1):191-202.
- D. Arpan, P. Tanmay, K.H. Suman, J. Arijit, M. Chiranjit, K. Pradeep, M. Das, R.P. Bikas and C.M. Keshab, Production of cellulolytic enzymes by Aspergillus fumigatus, ABK9 in wheat bran-rice straw mixed substrate and use of cocktail enzymes for deinking of waste office paper pulp, Bioresource Technology, 2013, 128: 290-296.
- R. Singh, A. Shukla, S. Tiwari and M. Srivastava, Review on delignification of lignocellulosic biomass for enhancement of ethanol production potential, Renewable and Sustainable Energy Reviews, 2014, 32: 713-728.
- D.B. Wilson Microbial diversity of cellulose hydrolysis, Current Opinion Biotechnology, 2011, 14:259-263.
- E.C. Valiño, E.A. Elías, V. Torres, T. Carrasco and N. Albelo, Improvement of sugarcane bagasse composition by the strain Trichoderma viride M5-2 in a solid-state fermentation bioreactor, Cuban Journal of Agricultural Science, 2004, 38 (2): 145.
- R.S. Rani, R. K. Sukumaran, A.P.L Kumar, C. Larroche and A. Pandey, Advancement and comparative profiles in the production technologies using solid-state and submerged fermentation for microbial cellulases, Enzyme and Microbial Technology, 2010, 46 (7,7): 541-549.
- N.R.C., Nutrient requirement of beef cattle, 7th ed., National Research Council, Washington D.C., National Academy Press, 2001.
- P. YU, An emerging method for rapid characterization of feed structures and feed component matrix at a cellular level and relation to feed quality and nutritive value, Archives of Animal Nutrition, 2006, 60(3):229 – 244.
- D.T. Chadwick, K.P. McDonnell, L.P. Brennan, C.C. Fagan and C.D. Everard, Evaluation of infrared techniques for the assessment of biomass and biofuel quality parameters and conversion technology processes: A review, Renewable and Sustainable Energy Reviews, 2014, 30: 672-681.
- M. Mandels and R.E. Andreotti, Measurement of sacarifying cellulose, Biotechnology Bioengineering Symp, 1976, 6: 2133.
- G. Gilardi, L. Abis and Cass, A. Carbon-13 Cp/MAS solid-state NMR and FT-IR spectroscopy of wood cell wall biodegradation, Enzyme and Microbial Technology, 1995, 17: 268-275.
- AOAC. In: Official methods of analysis of A.O.A.C. 16 TH Ed Association of Official Agricultural Chemist Washington, 2005.
- P. Van Soest, J.B. Robertson and BA Lewis, Methods for dietary fiber, neutral detergent fiber, and non-starch polysaccharides in relation to animal nutrition, Journal of Dairy Science, 1991, 74:3583-3597.
- H. Meir, Laborpraktikure Tierernahurungund, futtermittelm für Tierproduzenten, Verlag, Berlin, 1986.
- J.A. Di Rienzo, F. Casanoves, M.G. Balzarini, L. Gonzalez, M. Tablada and C.W. Robledo, InfoStatversión 2012, Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina, URL, 2012.
- D. B Duncan. Multiple ranges and multiple F test, Biometrics, 1955, 11:1.
- G. Gilardi, L. Abis and A. Cass, Carbon-13 Cp/ MAS solid-state NMR and FT-IR spectroscopy of wood cell wall biodegradation. Enzyme and Microbial Technology, 1995, 17: 268-275.
- R. Sindhu, A. Pandey and P. Binod, Chapter 4 - Alkaline Treatment Pretreatment of Biomass, 2015, 51-60.
- A. Jabasingh and C.N. Valli, Utilization of pretreated bagasse for the sustainable bioproduction of cellulase by Aspergillus nidulans MTCC 344 using response surface methodology, Industrial Crops and Products, 2011. 34 (3):1564-1571.
- N. Jayapal, A.K. Samanta, A.P. Kolte, S. Senani, M.l. Sridhar, K.P. Suresh and K.T. Sampath, Value addition to sugarcane bagasse: Xylan extraction and its process optimization for xylooligosaccharides production, Industrial Crops and Products, 2013, 42:14-24.
- B.M. Alves, S. Romo-Sánchez, J. Úbeda-Iranzo and A.I.F. Briones-Pérez, Fungi isolated from olive ecosystems and screening of their potential biotechnological use, New Biotechnology, 2012, 29(3,15): 451-456.
- F.M. Mendes, E. Heikkilä, M.B. Fonseca, A.M.F Milagres, A. Ferraz and P. Fardi, Topochemical characterization of sugar cane pretreated with alkaline sulfite, Industrial Crops and Products, 2015, 69: 60-67.
- M.L.W Pariza and M. Cook, Determining the safety of enzymes used in animal feed Regulatory Toxicology and Pharmacology, 2013, 56 (3): 332-342.
- X. D. Qin, J.Y. Shui, N. Zhu, E.W. Tao and H.Y. Li, Sugarcane bagasse degradation and characterization of three white-rot fungi, Bioresource Technology, 2013)131: 443-45.
- Z. Wang, H.O. Xin and A Geng, Cellulase production and oil palm empty fruit bunch saccharification by a new isolate of Trichoderma koningii D-64, Process Biochemistry, 2012, 47(11):1564-1571.
- S. Kalia, K. Thakur, A. Kumar and A. Celli, Laccase- assisted surface functionalization of lignocellulosics. Journal of Molecular Catalysis B: Enzymatic, (2014) 102: 48-58.
- M. Camassola and A.J.P. Dillon, Biological pretreatment of sugar cane bagasse for the production of cellulases and xylanases by Penicillium echinulatum, Industrial Crops and Products, 2009, 29 (2–3):642-647.
- S. Manju and B.C. Singh: Chapter 9, Production of Hemicellulolytic enzymes for Hydrolysis of Lignocellulosic, Biomass Biofuels, 2011, 203-228.
- L.A.B. Ribas, Q. Han, H. Jameel, H. Chang, J.L. Colodette and F.J.G. Borges, Production of fermentable sugars from sugarcane bagasse by enzymatic hydrolysis after autohydrolysis and mechanical refining. Bioresource Technology, 2015, 180: 97-105.
- T. Foyle, L. Jennings and P. Mulkahy, Compositional analysis of lignocellulosic material: evaluation of methods used for sugar analysis of waste paper and straw, Bioresourse Technology, 2007, 98:3026-36.



