Biological Activities Importance of Chalcone Derivatives
Jalal Hasan Mohammed*
Faculty of Pharmacy, Department of Pharmaceutical Chemistry, University of Karbala, Karbala, Iraq
Abstract
In this study the chemistry of chalcones has generated intensive scientific interest due to their biological and industrial applications. Chalcones are natural biocides and are well known intermediates in the synthesis of heterocyclic compounds exhibiting various biological activities. Chalcones and their derivatives possess some interesting biological properties such as antibacterial, antifungal, insecticidal, anesthetic, antiinflammatory, analgesic etc. Different methods are available for the preparation of chalcones. The most convenient method is the Claisen-Schimdt condensation of equimolar quantities of arylmethylketone with aryl aldehyde in the presence of alcoholic alkali, In the Claisen-Schmidt reaction, the concentration of alkali used, usually ranges between 10 and 60 %. The reaction is carried out at about 50 C for 12-15 hours or at room temperature for one week.
Keywords
Chalcones, Antibacterial, Antifungal, Anti-Inflammatory
Received: July 2, 2015
Accepted: July 9, 2015
Published online: August 6, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
The chemistry of chalcones has generated intensive scientific studies throughout the world. Especially interest has been focused on the synthesis and biodynamic activities of chalcones. The name "Chalcones" was given by Kostanecki and Tambor. These compounds are also known as benzalacetophenone or benzylidene acetophenone. In chalcones, two aromatic rings are linked by an aliphatic three carbon chain. Chalcone bears a very good synthon so that variety of novel heterocycles with good pharmaceutical profile can be designed. Chalcones are -unsaturated ketone containing the reactive ketoethylenic group –CO-CH=CH-. These are coloured compounds because of the presence of the chromophore -CO-CH=CH-, which depends on the presence of other auxochromes. [1,2] Chalcones are used to synthesize several derivatives like cyanopyridines, pyrazolines isoxazoles and pyrimidines having different heterocyclic ring Systems [3]. As shown in Fig. 1.
The chalcones are associated with different biological activities like Insecticidal, anticancer, anti-inflammatory, bactericidal, fungicidal, Antiviral, antitumor, antimalarial and antiulcer [4]. The presence of -unsaturated carbonyl system of chalcone makes it biologically active145. They have shown antibacterial activity against S. aureus, E. coli, C. albicans, T. utilis, S. sake, W. anomala and some other organisms [5]. Pyrazole is a class of Compounds, which has many applications in different field. One of the methods for the synthesis of such compound is from unsaturated carbonyls (chalcone) by the cyclization with hydrazine and substituted hydrazine. Pyrazole and their derivatives are considered to be important for drugs and agricultural chemicals. Some substituted pyrazoles and their derivatives have been reported to possess several interesting biological activities such as hypnotic properties, antimicrobial, antitumor and antifungal. Many pyrazoles are used for the treatment of thyroid and leukemia. It has incidental antiviral activity against Herpes infections [6].
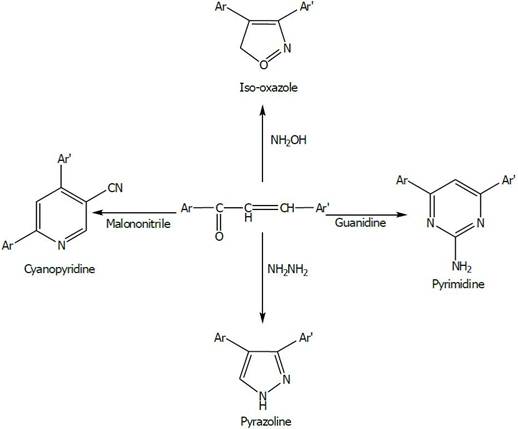
Figure 1. Synthesize several derivatives like cyanopyridines, pyrazolines isoxazoles and pyrimidines [3].
Different methods of nomenclatures for chalcone were suggested at Different times. The following pattern has been adopted by "Chemical Abstracts" published by the American chemical society [7] such as shown in Fig. 2, 3.
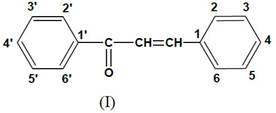
Figure 2. Chemical abstracts published by the American Chemical Society [6].
The British Chemical Abstract and Journal of Chemical Society have followed the following system [7].
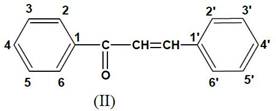
Figure 3. The British Chemical Abstract and Journal of Chemical Society [6].
2. Experimental Section Biological Activities of Chalcones Derivatives
Zaianb Ngani et.al [8] Synthesis and antibacterial study of Aspirin-Chalcone derivatives. In this study, new aspirin-chalcone derivatives have been successfully synthesized and characterized via FTIR, 1H and 13C NMR spectroscopy. The antibacterial activities of synthesized compounds were investigated towards Escherichia coli ATCC 8739 via turbidimetric kinetic method. The newly synthesized aspirin-chalcone derivatives, however, showed poor antibacterial activity against E. coli ATCC 8739 at the concentration of 50, 80 and 100 ppm. The effect of the molecular structure of the synthesized compounds on the antibacterial activity is discussed. As shown in Fig. 4.
Navin Kumar Tailor [9] synthesis and antifungal activity of certain chalcones and their reduction, antifungal activity was evaluated by using agar diffusion. Chalcones were more potential than corresponding tetrahydrochalcones. One step reduction by using sodium Formate and 5% Pd/C is a very efficient method. Α, β- unsaturated carbonyl functionality imparts very significant role in antifungal activity such as Fig. 5.
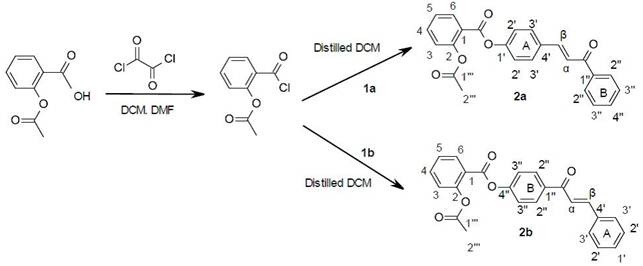
Figure 4. Aspirin-Chalcone derivatives [8].
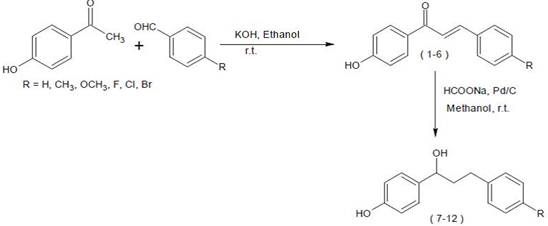
Figure 5. Synthesis and antifungal activity of certain chalcones [9].
Thanh-Dao Tran et. al [10] synthesis and antibacterial activity of some heterocyclic chalcone analogues alone and in combination with antibiotics, a series of simple heterocyclic chalcone analogues have been synthesized by Claisen Schmidt condensation reactions between substituted benzaldehydes and heteroaryl methyl ketones and evaluated for their antibacterial activity. The structures of the synthesized chalcones were established by IR and 1H-NMR analysis, such as in the Fig. 6.
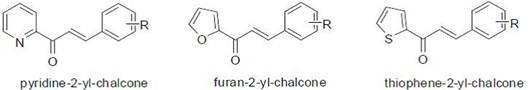
Figure 6. Some heterocyclic chalcone analogues [10].
N. RamalaKshmi et.al [11] synthesis, characterization and biological screening of some novel 1, 3, 5 Trisbstituted 2-Pyrazolines, pyrazolines of nicotinic acid derivatives were synthesized. Nicotinic acid was converted to nicotinic acid hydrazide. 10 different chalcones were synthesized by reacting different aromatic aldehydes with paracetamol. These chalcones on reaction with nicotinic acid hydrazide yielded 10 different 2-pyrazolines. The structure of synthesized compounds was confirmed by spectral data. They were screened for anti-microbial activities Table 1 in such as in the Fig. 7.
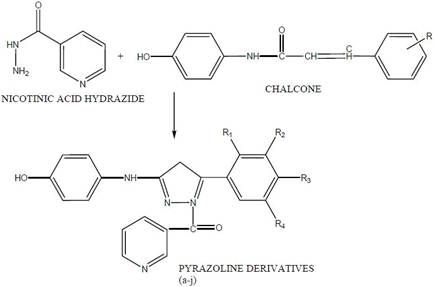
Figure 7. SOME NOVEL 1, 3, 5 TRISUBSTITUTED 2-PYRAZOLINES, pyrazolines of nicotinic acid [11].
Table 1. Anti-microbial activity [11].
| Compd | Anti-bacterial (25μg/disc) | Anti-fungal (25μg/disc) | |||||||
| Zone of inhibition in mm | Zone of inhibition in mm | ||||||||
| SA | SE | KP | EC | CA | AN | ||||
| a | 12 | 11 | 14 | 13 | 17 | 15 | |||
| b | 18 | 17 | 15 | 17 | 18 | 14 | |||
| c | 14 | 19 | 17 | 14 | 21 | 14 | |||
| d | 19 | 18 | 22 | 21 | 12 | 17 | |||
| e | 19 | 13 | 16 | 14 | 15 | 16 | |||
| f | 16 | 15 | 15 | 14 | 18 | 16 | |||
| g | 11 | 13 | 18 | 17 | 20 | 18 | |||
| h | 20 | 20 | 23 | 18 | 21 | 19 | |||
| i | 15 | 13 | 14 | 16 | 21 | 21 | |||
| j | 17 | 18 | 19 | 20 | 18 | 15 | |||
| STD | 24 | 22 | 28 | 27 | 26 | 22 | |||
Samer A. Hasan et.al [12] Synthesis of new diclofenac derivatives by coupling with chalcone derivatives as possible mutual produgs. The structure of the synthesized derivatives has been characterized by elemental Microanalysis (CHN), FTIR spectroscopy, and other physicochemical properties. The anti-inflammatory activity of the synthesized fluorinated chalcone derivative was performed using the cotton pellet-induced granuloma in rats as a model, and found to be comparable to dexamethasone in this regard such as in the Fig. 8.
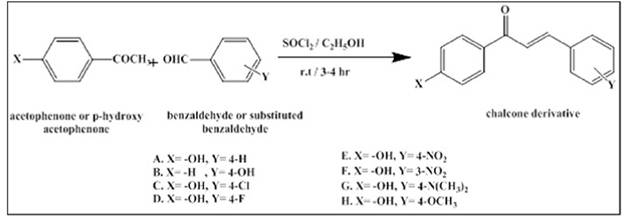
Figure 8. Diclofenac derivatives by coupling with chalcone derivatives [12].
Hocine Aichaoui et.al [13] synthesis and pharmacological evaluation of antioxidant chalcone derivatives of 2(3H)-benzoxazolones, so chalcones featuring an analgesic/anti-inflammatory pharmacophore, i.e., The 2 (3H) -benzoxazolone heterocycle such as in the Fig. 9.
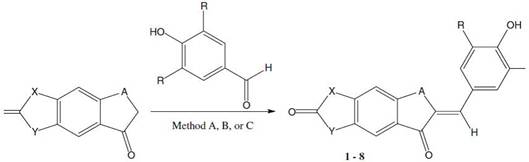
Figure 9. Synthesis and pharmacological evaluation of antioxidant chalcone derivatives of 2 (3H) –benzoxazolones [13].
Suvitha Syam et. al [14] synthesis of chalcones with anticancer activities, several chalcones were synthesized and their in vitro cytotoxicity against various human cell lines, including human breast adenocarcinoma cell line MCF-7, human lung adenocarcinoma cell line A549, human prostate cancer cell line PC3, human adenocarcinoma cell line HT-29 (colorectal cancer) such as in the Fig. 10.
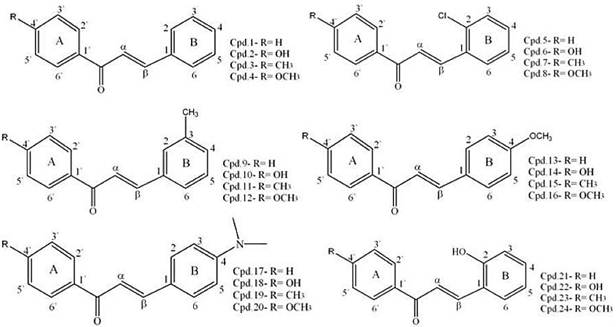
Figure 10. Synthesis of chalcones with anticancer activities [14].
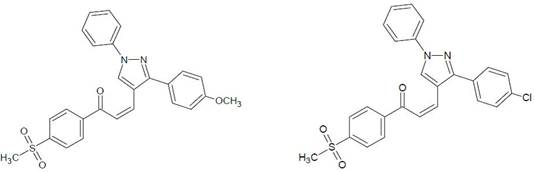
Figure 11. Synthesis and antifungal activity of some novel chalcones containing pyrazole moiety [15].
J. J. Vora et.al [15] Synthesis and antifungal activity of some novel chalcones containing pyrazole moiety, various chalcone derivatives of 1-(4-methyl sulfonylphenyl)-3-(1, 3-diphenyl-1H-pyrazol-4-yl)-2-propen-1-one were Synthesized by condensation of 1, 3-diphenyl-1H-pyrazole-4-carbaldehyde and 4-methyl sulfonylphenyl acetophenone. Chalcone derivatives were characterized by FT-IR, 1H-NMR, Mass spectral analysis and elemental analysis. All the synthesized compounds have been screened for their antifungal activities by using cup-plate method such as in the Fig. 11.
3. Results and Discussion
In this work the focus was on Chalcones are also key precursors in the synthesis of many biologically important heterocycles such as benzothiazepine, pyrazolines, 1, 4-diketones, and flavones. Thus the synthesis of chalcones has generated vast interest to organic as well as for medicinal chemists.
4. Conclusion
In summary, we have synthesized some chalcones and identified them from their spectral data. We have most of the compounds were very active biological agents, chalcones and pyrazole derivatives were synthesized. Compounds with electron releasing groups such as methoxy and hydroxyl showed better antibacterial activity than other not having such groups structures of the synthesized substituted chalcones were confirmed from their respective IR, 1H- NMR Studies. From the antimicrobial screening it was observed that all the compounds exhibited activity against all the organisms employed.
Acknowledgments
The author would like to acknowledge the Faculty of Pharmacy, University of Karbala in carrying out this research work.
References
- S. V. Kostanecki and Tambor, J. Chem Ber., 32, 1921.
- H. Rupe and D. Wasserzug, J. Chem Ber., 34, 3527, 1901.
- M. A. El. Hashah; M. El-Kady; M. A. Saiyed and A. A. Elaswy, Egypt. J. Chem., 27, 715, 1985.
- M. Larsen, H. Kromann, A. Kharazmi and S. F. Nielsen, Bioorg. Med. Chem. Lett. 15, 4858, 2005.
- S. Ishida, A. Matsuda and A. Kawamura, Chemotherapy, 8, 146, 1960.
- P Prasanna raja, M.S. Riyazulah, V. Siva kumar, International Journal of ChemTech Research, Synthesis and Biological Evaluation of some Chalcone Derivatives, Vol.2, No.4, pp. 1998-2004.
- P. S. Utale, P. B. Raghuvanshi and A. G. Doshi, Asian J. Chem., 10, 597, 1998.
- ZAINAB NGAINI et. al, Borneo Journal of Resource Science and Technology, Synthesis and Antibacterial Study of Aspirin-Chalcone Derivatives, 2013, 3(1): 52-57.
- Navin Kumar Tailor, Indo Global Journal of Pharmaceutical Sciences, Synthesis & Antifungal Activity of Certain Chalcones & Their Reduction, 2014; 4(1): 25-28.
- Thanh-Dao Tran et.al, Molecules, Synthesis and Antibacterial Activity of Some Heterocyclic Chalcone Analogues Alone and in Combination with Antibiotics, 2012, 17, 6684-6696.
- N. Ramalakshmi, S. Deepa, K. Sumanth Srinivas, A. Puratchikodyand S. Arunkumar, RJC, SYNTHESIS, CHARACTERIZATION AND BIOLOGICAL SCREENING OF SOME NOVEL 1,3,5 TRISUBSTITUTED 2-PYRAZOLINES, Vol.2, No.2 (2009), 393-396.
- SAMER A. HASAN AND AMER N. ELIAS, International Journal of Pharmacy and Pharmaceutical Sciences, SYNTHESIS OF NEW DICLOFENAC DERIVATIVES BY COUPLING WITH CHALCONE DERIVATIVES AS POSSIBLE MUTUAL PRODRUGS, Vol 6, Issue 1, 2014.
- Hocine Aichaoui Faouzi Guenadil, Med Chem Res, Synthesis and pharmacological evaluation of antioxidant chalcone derivatives of 2(3H)-benzoxazolones, 2009, 18:467–476.
- Suvitha Syam, Siddig Ibrahim Abdelwahab, Mohammed Ali Al-Mamary and Syam Mohan, Molecules, Synthesis of Chalcones with Anticancer Activities, 2012, 17, 6179-6195.
- J. J. Vora#, Dharmesh M. Patel, Ketan C. Parmar, Bhavesh M. Patel and Sunil. B. Vasava, Scholars Research Library, Synthesis and antifungal activity of some novel chalcones containing pyrazole moiety, 2014, 6(5):166-170.



