Seed-Coating Agent Affects on the Physiology and Biochemistry of Astragalus
Xiu-Bo Liu1, 2, Wei-Chao Ren1, Song Yan1, Li-Ying Sun1, Ling Ma2, Wei Ma1, 2, *
1Pharmacy School, Heilongjiang University of Chinese Medicine, Harbin, PR China
2Key Laboratory of Forest Tree Genetic Improvement and Biotechnology, Ministry of Education, Northeast Forestry University, Harbin, PR China
Abstract
Astragalus membranaceus (Fisch.) Bge (AR, Huangqi in Chinese), one of the most important medicinal plants in Asia, was found to exhibit various bioactivities. We developed the appropriate seed coating technology to encounter agronomic and cultivation and carried the seed physiological effects and mechanisms. To better understand the effect and mechanism of seed-coating agent (SCA) on the physiology and biochemistry of AR, the physiological and biochemical effects of SCA on AR were studied under field experiment. The results showed that the seed treated with SCA obviously increased germination rate of AR seed, maintained dramatically higher vigor of root, and chlorophyll content, promoted the area of photosynthesis, significantly improved the dry weight of root, stem, leaf and accumulated more soluble sugar, when compared with the uncoated without the treatment, which provided a guarantee to enhance the hardness resistance of seedlings, and finally to enhance the quality of AR. It can improve photosynthesis and accelerate metabolism, promote the formation and transformation of proteins and carbohydrates, thus providing more material and energy for growth and development of AR. Seed coating can improve astragaloside content, improve the quality of AR, so as to improve medicinal value.
Keywords
Astragalus membranaceus, Seed-Coating Agent, Physiology, Biochemistry
Received: June 8, 2015
Accepted: June 30, 2015
Published online: July 23, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Astragalus membranaceus (Fisch.) Bge (AR, Huangqi in Chinese) is one of the most important medicinal plants in Asia, and has received much of the world attention in recent years, as listed in the 2010 edition of Pharmacopoeia of the People’s Republic of China [1-3]. It was first described in the Chinese book ShenNongBenCaoJing in 200 AD with a wide range of treatment effects and no toxicity. This herb has been extensively studied and various bioactivities, such as hypotensive, inducing vasodilatation, antioxidative, immunostimulating, antiviral, protecting the myocardium in diabetic nephropathy, promoting the motility of human spermatozoa, reducing the capillary hyperpermeability and alleviating the dyskinesia; enhancing cardiovascular function, antiaging, hepatoprotective effect, inhibiting cyclooxygenase-2, sterol biosynthesis, and antibacterial [4-9]. Clinical research showed that AR can improve cardiovascular function, restore and strengthen immune response, and enhance vitality [10].
Since the Germains Seed Company invented the seed-coating agent for rain-fed crops first in the 1930s, the research and development of the seed-coating agent (SCA) have been rapid [11]. At present, seed-coating is widely used on the main crops in developed countries. The seed coating research and extension for the four main crops in China: Corn, wheat, AR, and cotton [12]. Now, the research and extension of seed-coating for rain-fed crops has achieved remarkable results, and the technique of new formulations of seed-coating agents has also achieved significant progress [13-15]. However, all researches of seed coating in the past were limited to accelerating growth and defending diseases and pests; the research for effect and mechanism of seed-coating has not been reported on the plant quality, so far at home and abroad. As a case study, it is expected that, by means of seed coating, the AS seed quality can be realized, and at the same time, the AR production can also be enhanced. Using AR seed treated with SCA as materials, this study focused on explore the effect of SCA on the physiological and biochemical activities of AR, to provide a new technical means and theoretical basis for us to enhance the quality of AR.
2. Material and methods
2.1. Materials
Seed samples of AR were collected from the Medicinal Botanical Garden of Heilongjiang University of Chinese Medicine. Seed-coating agent (Fig S1) for examination was prepared by our Laboratory. Formula composition of SCA consists of wetting agent CL-3 (0.05%), 602 dispersing agent (2.0%), a thickening agent xanthan gum (2.0%) and magnesium aluminum silicate (0.05%), a film forming agent sodium carboxymethyl cellulose (1.0 %), antifreeze agent ethylene glycol (5.0 %).

Fig. S1. Pattern of SCA.
2.2. Treatment Methods
2.2.1. Laboratory Experiment
The AR seed was treatment at the variable temperature for 12h (constant temperature soak at 41 °C for 4 h, then keep moisturizing at 25 °C for 8 h, frequently changing the water). After the surface to dry suction with absorbent paper, dried at room temperature for 30 min, and then AR seed coated with SCA (1:50). AR seed with no coating was the uncoated group. The randomly selected 100 seeds were placed into a dish that has paved double wet filter paper, spread out and covered with a layer of moist filter paper, repeated three times, and placed in an incubator at 25 °C. The fourth days, we investigate the germination, the seventh days, we calculated the germination rate. The Germination rate of AR seedlings (%) = Numbers of Germination seed /Total seed investigated x 100.
2.2.2. Field Experiment
The method of seed treatment was the same as in the laboratory experiment. After cleaning, the AR seeds for examination were coated with the SCA mentioned above in the proportion of 1:50 (w/w), and then dried by airing to prepare for use. The treated seeds were evenly sown in the plots of the experimental field in Heilongjiang University of Chinese Medicine, China, on 25 March, 2011, with an area of 50 cm x 50 cm. The treatments were divided into two groups, the treated with SCA and the untreated with SCA (Fig S2), which were arranged in a randomized block design with three replications. The AR Germination rate was surveyed and the physiological and biochemical parameters were determined with three replications. At 30th day, 60th day, 90th day, 120th day and 150th day after sampling, and its underground, aboveground related indicators were measured; in each test of 6 plants were treated with three replications.

Fig. S2. The seeds form after astragalus seed coated. The coated seed (A) and uncoated seed (A).
2.3. Measurement Methods
Length indicators: plant height, root diameter, stem diameter were measured using the direct determination method; Quality index: root dry weight, stem dry weight and leaf dry weight was using drying (105 ºC fixing, 80 ºC drying) weighing method; Stem plumpness: the ratio was the stem weight/height; Seedling index: (stem width / height) × aboveground dry weight; Leaf area: select 20 fresh leaves were punch with a diameter of 4 mm punch, after drying weighed to calculate the leaf dry weight and further to investigative the leaf area.
2.4. Physiological Indicators of AR
2.4.1. Determination of Root Vigor
The root system of seedlings was washed apart from the soil with water, and the entire root was cut from the seedlings, transferred to filter paper to absorb the water, and then weighed. The root vigor was determined according to the alphanaphthylamine oxidation method described by Zhang (1992). The experiment was repeated three times.
2.4.2. Determination of Chlorophyll Content
The fully-expanded leaves (accurately 0.2 g) were sampled to determine the chlorophyll content after they were removed ends and veins and cut into pieces. The experiment was carried out as described by Zhang (1992) and repeated three times.
2.4.3. Determination of Nitrate Content
The nitrate content was determined using 0.5 g leaves for each treatment in accordance with the approach outlined by Zhang (1992) and repeated three times.
2.4.4. Measurement of Soluble Sugar Content
The soluble sugar content was measured according to the method described by Bai (1995). 0.5 g leaves were homogenized by grindmg with quartz sand. The homogenized leaf capacity was increased to 50 mL. It was put aside for 1 h at room temperature, and then filtered. The extract of 0.5 mL was sampled for the measurement. The experiment was repeated three times.
2.4.5. Determination of Protein Content
The protein content was determined using 0.5 g leaves for each treatment in accordance with the approach outlined by Zhang (1992) and repeated three times.
2.5. Measurement of Astragaloside of 1 Year Old AR
Astragaloside is the major active ingredients of AR, which has been shown to be clinically effective for treating chronic diseases. Diones U-3000 liquid chromatography system (Diones, USA) was consisting of a quaternary pump, an on-line degasser, an autosampler, and a diode-array UV/Vis multi-wavelength detector (DAD). The separations were performed on a Diamonsil-C18 column (4.6 mm×250 mm, 5 μm) and the column temperature was set at 30 °C. The mobile phase was composed of acetonitrile-water (32:68). The solvents were filtered and degassed prior to use. The solvent flow rate was 0.8 mL/min and the injection volume was 20 μL. The detection wavelength for content determination was set at the maximal UV absorption peak of the compound, it was 202 nm for astragaloside. The absorption spectra were recorded within 200–400 nm. The established method was successfully validated by acceptable linearity, limits of detection and quantitation, precision, repeatability, stability and accuracy.
3. Results and Discussion
3.1. Effect of SCA on the Germination Rate of AR Seed
The investigation for germination rate of AR seed showed that the rate of the uncoated seed was considerably lower. However, after treatment with SCA, the average germination rate of seedlings was rather high, for example, the rate of AR seed treated by SCA was 20.66 % and 16.54 %, respectively (Table 1). It indicated that this SCA can obviously promote germination rate of AR seed.
Table 1. The germination rate of Astragalus seeds.
| Treatment | The average germination (%) | The average germination rate (%) |
| Control | 52.67 | 72.13 |
| Seed-coating agent | 73.33 | 88.67 |
3.2. Effect of SCA on the Morphological Indicators of AR
AR seeds treated by SCA, with the increasing sowing time, plant height showed an increasing trend at the 30 d, 60 d, 120 d, 150 d, compared with the uncoated group, it decreased by 9.59%, 32.80%, 1.94% and 5.85%, respectively (Fig 1a). The test result showed that SCA treatment reduced the plant height in the early seedling growth. Astragalus root length at 30 ~ 90 d in coated group were shorter than the uncoated group, only 5.94% higher than the uncoated group at 120th d, indicating that the influence of SCA on the root length of AR is not obvious (Fig 1b). Under the SCA condition in field, root diameter increased by 29.12, and 18.67 %, respectively, over the uncoated group (Fig 1c). As can be seen from Fig 1d, after seed coating, stem diameter gradually increased by 29.84% from 60 d when compared with the uncoated group.
Dynamic change of dry weight of root (Fig 2a), stem (Fig 2b), leaf (Fig 2c), petiole (Fig 2d) in coated group was consist with the uncoated group 30 ~ 60 d after sowing. After sowing 60 d, coated and uncoated groups began to clearly separate, and the dry weight significantly increased. After 150th day sowing, the dry weight of root, stem, leaf, petiole increased by 49.31%, 15.21%, 63.07% and 19.44%, respectively, over the uncoated group. Stems plumpness mainly used to reflect the quality of unit length of the stem, plumpness is the higher and the mechanical strength is the better and the lodging capacity is stronger.
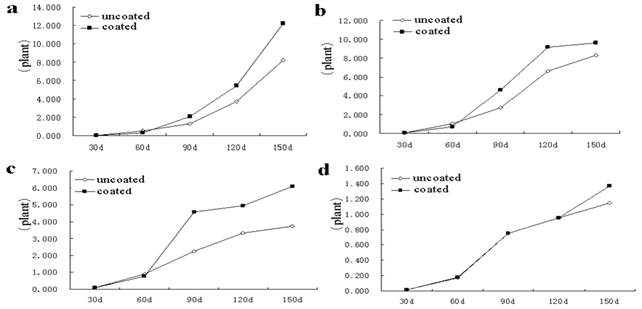
Fig. 1. Effect of seed-coating agent on the morphological indicators of Astragalus Dynamic change of plant height (a), of root length (b), root diameter (c), stem diameter (d).
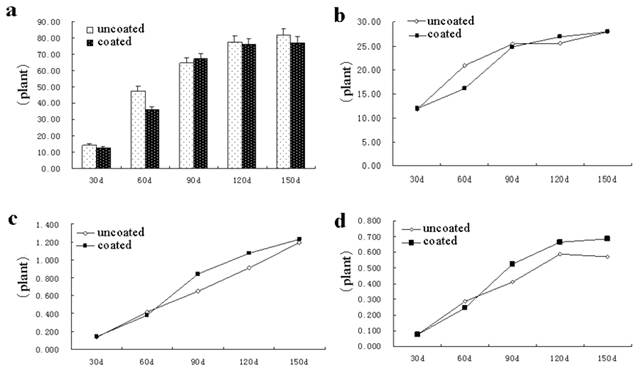
Fig. 2. Effect of seed-coating agents on the Quality indicators of Astragalus Dynamic change of dry weight of root (a), stem (b), leaf (c), petiole (d).
The stems plumpness of AR disposed with SCA increased by 61.90, 42.35, and 22.55%, respectively, over the uncoated group (Fig 3a). These results indicated that the SCA could significantly improve the stem plumpness of AR under SCA conditions, and could improve seedling resistance to a variety of biological adversity. The seedling index in the coated group and the uncoated group continued to increase from 90 d after sowing, significantly increased by 64.52% compared with the uncoated group, indicating that SCA can significantly improve seedling index, help shape the AR seed, and improve the disease resistance of AR (Fig 3b). Fig 3c shows that leaf area of AR in coated group significantly increased at 60th d after sowing than that of the uncoated group, up to 99.73 %, indicating that SCA can significantly improve the area of photosynthesis in order to enhance the quality of AR
3.3. Effect of SCA on the Chlorophyll Content of AR
Under SCA condition in field, the SCA could improve the chlorophyll content of AR seedlings. For example, the total chlorophyll content of seedlings for AR disposed with SCA decreased at 90thday when compared with those at uncoated group (Fig 4a); whereas the chlorophyll a content of the uncoated increased when compared with the uncoated group (Fig 4b), the chlorophyll b content of AR treated with the seed-coating agent increased at 60thday and decreased at 120thday, respectively, over the uncoated (Fig 4c). These results confirmed that the SCA could not only significantly improve the chlorophyll content of seedlings but could also maintain the chlorophyll content at a high level.
3.4. Effects of SCA on the Nitrate Content of AR
For the treatments coated with the four SCA, the nitrate content was significantly higher than the uncoateds, increased by 69.94 %, when compared with those at normal condition (Fig 4d). Under SCA condition in field, SCA increased the nitrate content of AR. The accumulation for the treatments treated with SCA was significantly higher than that of the uncoated group.
3.5. Influence of SCA on the Protein and Soluble Sugar Content of AR
For the treatments coated with the four SCA, the protein content was significantly higher than the uncoateds, increased by 27.46 % at September, when over the uncoated (Fig 4e). SCA can increase the protein content of AR. The protein accumulation for the AR treated with SCA was significantly higher than that of the uncoated group. As seen in Fig 4f, SCA could increase the soluble sugar content of seedlings. More than 40 constituents in AR saponins have been identified from the AR root, of which astragaloside is a major active ingredient [16]. Astragaloside has various pharmacological activities and is used as a quality-uncoated marker component of AR in the Chinese Pharmacopoeia (2010 version). It has been reported that astragaloside has an antioxidant effect and the accepted underlying mechanisms include modulation of energy metabolism and Ca2+ homeostasis [17]. Moreover, astragaloside shows neuroprotective effects on promoting axonal regeneration and reconstruction of neuronal synapses [18-20]. The root in uncoated and coated AR contained the astragaloside were 0.139% and 0.162%, respectively, determined by HPLC method, increased 0.023% compared to the uncoated group. The results showed that after treatment by SCA can improve astragaloside content.
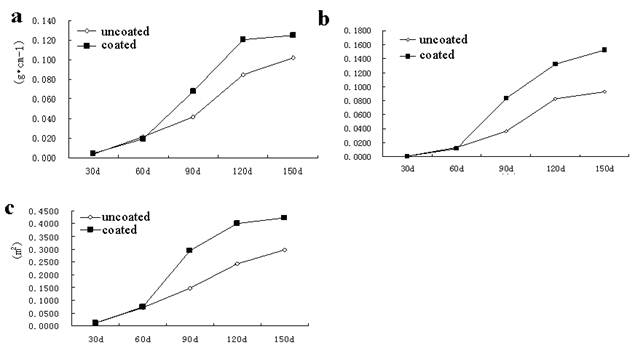
Fig. 3. Dynamic change of the stem plumpness (a), seedling index (b), leaf area (c).
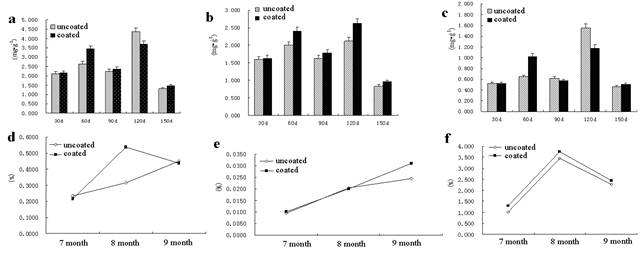
Fig. 4. Effect of seed-coating agents on the physiological indicators of AstragalusDynamic change of the content of total chlorophyll (a), chlorophyll a (b), chlorophyll b (c), NO3-_N (d), protein (e), soluble sugar (f).
Several researches were done about seed-coating agents, but few researches on the seed-coating agent affects on the physiology and biochemistry of AR. In this study, using the prepared SCA, the germination rate of AR seedlings was significantly improved by the seed coating treatments. The germination rate of seedlings is the ultimate outward manifestation of various physiological processes, which can directly reflect the strength of AR. It indicated that this seed-coating agent can obviously promote germination rate of AR seed, significantly improve the dry weight of root, stem, leaf, petiole of AR, stems plumpness and the chlorophyll content, the area of photosynthesis in order to enhance the quality of Astragalus. The root vigor and the chlorophyll content reflect the abilities of nutrient absorption and photosynthetic assimilation of seedlings, which are the important physiological indices measuring the quality of seedlings. The results showed that SCA were able to maintain a high root vigor and high chlorophyll content for seedlings, so as to provide a guarantee for maintaining the normal nutrient absorption and growth, normal photosynthetic capability, and for strengthening the hardness tolerance of seedlings. Sugar accumulation is able to increase the concentration of cell sap, lower its freezing point, alleviate the over-dehydration of cytoplasm, and protect cytoplasmic colloid from cold coagulation, thereby enhancing the cold tolerance of plants. In comparison with the conventional application method, this technique can change the field operation into an indoor operation, , and thus attained the aim of simple operation, saving resources, and being environment friendly, and provided a new technical way to broaden the application fields of seed-coating agents and reduce the damage to AR.
4. Conclusion
In this study, the SCA maintained a higher root vigor and higher chlorophyll content of seedlings, and kept normal nutrient absorption and photosynthesis of AR seedlings, which provided a guarantee to enhance the hardness resistance of seedlings. The SCA also increased the content of nitrate, protein and soluble sugar, which improved the tolerance of the cell itself, and thereby reduced the injury rate of seedlings, thus demonstrating a significant effect of environmental tolerance. Based on the comprehensive evaluation of these physiological and biochemical indices tested above, our study provided an initial description of SCA could improve seedling resistance to a variety of biological adversity, and may help us to enhance the quality of AR.
Acknowledgments
This work was supported by grants from the Forestry Public fund(Grant No. 201004079).
References
- Liu XB, Ma L, Zhang AH, Zhang YH, Jiang J, Ma W, Zhang LM, Ren WC, Kong XJ.High-throughput analysis and characterization of Astragalus membranaceus transcriptome using 454 GS FLX.PLoS One. 2014;9(5):e95831.
- Fu J, Huang L, Zhang H, Yang S, Chen S.Structural features of a polysaccharide from Astragalus membranaceus (Fisch.) Bge. var. mongholicus (Bge.) Hsiao.J Asian Nat Prod Res. 2013;15(6):687-92.
- Li Z, Zhang L, He W, Zhu C, Yang J, Sheng M.Astragalus membranaceus Inhibits Peritoneal Fibrosis via Monocyte Chemoattractant Protein (MCP)-1 and the Transforming Growth Factor-β1 (TGF-β1) Pathway in Rats Submitted to Peritoneal Dialysis.Int J Mol Sci. 2014;15(7):12959-71.
- Chen W, Xia Y, Zhao X, Wang H, Chen W.The critical role of Astragalus polysaccharides for the improvement of PPARα [correction of PPRAα]-mediated lipotoxicity in diabetic cardiomyopathy.PLoS One. 2012;7(10):e45541.
- Lee DY, Noh HJ, Choi J, Lee KH, Lee MH.Anti-inflammatory cycloartane-type saponins of Astragalus membranaceus.Molecules 18(4):3725-32.
- Du HW, Zhao XL, Zhang AH, Yao L, Zhang YY (2014)Rapid Separation, Identification and Analysis of Astragalus membranaceus Fisch Using Liquid Chromatography-Tandem Mass Spectrometry.J Chromatogr Sci 52(3):226-31.
- Hong MJ, Ko EB, Park SK, Chang MS.Inhibitory effect of Astragalus membranaceus root on matrix metalloproteinase-1 collagenase expression and procollagen destruction in ultraviolet B-irradiated human dermal fibroblasts by suppressing nuclear factor kappa-B activity.J Pharm Pharmacol 2013, 65(1):142-8.
- Xu X, Li F, Zhang X, Li P, Zhang X, Wu Z, Li D.In vitro synergistic antioxidant activity and identification of antioxidant components from Astragalus membranaceus and Paeonia lactiflora.PLoS One. 2014;9(5):e96780.
- Tong X, Xiao D, Yao F, Huang T.Astragalus membranaceus as a cause of increased CA19-9 and liver and kidney cysts: a case report.J Clin Pharm Ther. 2014. doi: 10.1111/jcpt.12173.
- Yao M, Qi Y, Bi K, Wang X, Luo X, et al.(2000) A precolumn derivatization high-performance liquid chromatographic method with improved sensitivity and specificity for the determination of astragaloside IV in Radix Astragali. Journal of Chromatographic Science 38:325-328.
- Brooker NL, Lagalle CD, Zlatanic A, Javni I, Petrovic Z.Soy polyol formulations as novel seed treatments for the management of soil-borne diseases of soybean.Commun Agric Appl Biol Sci. 2007;72(2):35-43.
- Venne J, Beed F, Avocanh A, Watson A.Integrating Fusarium oxysporum f. sp. strigae into cereal cropping systems in Africa.Pest Manag Sci. 2009;65(5):572-80.
- Hannan MA, Hasan MM, Hossain I, Rahman SM, Ismail AM, Oh DH.Integrated management of foot rot of lentil using biocontrol agents under field condition.J Microbiol Biotechnol. 2012 Jul;22(7):883-8.
- Dong HZ.Underlying mechanisms and related techniques of stand establishment of cotton on coastal saline-alkali soil.Ying Yong Sheng Tai Xue Bao. 2012;23(2):566-72.
- Wang K, Yan PS, Cao LX, Ding QL, Shao C, Zhao TF.Potential of chitinolytic Serratia marcescens strain JPP1 for biological control of Aspergillus parasiticus and aflatoxin.Biomed Res Int. 2013;2013:397142.
- Lv L,Wu SY,Wang GF,Zhang JJ,Pang JX,Liu ZQ,Xu W,Wu SG,Rao JJ. Effect of astragaloside IV on hepatic glucose-regulating enzymes in diabetic mice induced by a high-fat diet and streptozotocin.Phytother Res.2010;24(2):219-24.
- Tu L, Pan CS, Wei XH, Yan L, Liu YY, Fan JY, Mu HN, Li Q, Li L, Zhang Y, He K, Mao XW, Sun K, Wang CS, Yin CC, Han JY.Astragaloside IV protects heart from ischemia and reperfusion injury via energy regulation mechanisms.Microcirculation. 2013;20(8):736-47.
- Luo Y, Qin Z, Hong Z, Zhang X, Ding D, Fu JH, Zhang WD, Chen J.Astragaloside IV protects against ischemic brain injury in a murine model of transient focal ischemia.Neurosci Lett. 2004;363(3):218-23.
- Zhu J, Lee S, Ho MK, Hu Y, Pang H, Ip FC, Chin AC, Harley CB, Ip NY, Wong YH.In vitro intestinal absorption and first-pass intestinal and hepatic metabolism of cycloastragenol, a potent small molecule telomerase activator.Drug Metab Pharmacokinet. 2010;25(5):477-86.
- He Y, Xi J, Zheng H, Zhang Y, Jin Y, Xu Z.Astragaloside IV inhibits oxidative stress-induced mitochondrial permeability transition pore opening by inactivating GSK-3β via nitric oxide in H9c2 cardiac cells.Oxid Med Cell Longev. 2012;2012:935738.



