Studies on Adsorption Capacity of Zeolite for Removal of Chemical and Bio-Chemical Oxygen Demands
Milan M. Lakdawala1, Yogesh S. Patel2, *
1Chemistry Department, S. P. T. Arts and Science College, Godhra, Gujarat, India
2Chemistry Department, Government Science College, Gandhinagar, Gujarat, India
Abstract
Pollution of soil and water by hazardous chemicals excreted from industries has become serious environmental concern. From the environmental and aesthetic point of view, removal of pollutants from effluent is extremely essential. Due to the organic loads, the treatment of removal efficiency is rather challenging by conventional methods. Therefore, adsorption process is found to be the most efficient method. Numerous approaches have been studied for the development of low–cost adsorbents. Zeolite is found to be the most effective and economical adsorbent. In the present study, zeolite with specific surface area of 2762.47cm2/gm was used for removal of biochemical oxygen demand from the combined waste water of sugar industry. The combined waste water of sugar industry is treated with different doses (i.e. 5 gm/L, 10 gm/L, 40 gm/L, 80 gm/L, 120 gm/L, 160 gm/L, 200 gm/L) of zeolite. With increase in dosage of zeolite, considerable decrease in biochemical oxygen demand was observed.
Keywords
Zeolite, Adsorption, Freundlich Isotherm, Langmuir Isotherm
Received: April 7, 2015
Accepted: April 19, 2015
Published online: May 28, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Numerous studies have been conducted for the treatment of wastewater using granular activated carbons [1–2], adsorbent [3–4], Fenton’s reagent [5], wet oxidation [6], coagulation–electro oxidation [7] and advanced oxidation with biological oxidation [8]. Application of adsorbents based on zeolite has certain advantages over conventional methods applied for the industrial wastewater treatment. Natural zeolite is highly porous material. It has a natural negative charge which gives it the capacity to adsorb cations. Zeolites have great potential as effective adsorbents in numerous processes of purification of drinking water and wastewater, such as water softening, removal of phosphate [9–12], the removal of ammonia [9, 11–12], the removal of nitrogen [9, 11–13], the removal of dissolved organic matter [9, 13–16] and color [9,17], the removal of heavy metals from industrial wastewater [9, 18–19], the removal of radioactive substances from wastewater [9], desalination seawater [9] and many others.
Langmuir and Freundlich adsorption isotherms were well explained onto natural materials [20–21]. In the present study, we have chosen the southern part of Gujarat state of India, were maximum sugar industries were located. We have collected various samples from the combined waste water of sugar industries. It was aimed to carry out the experiments to remove COD and BOD from the waste water of sugar industry using zeolite with specific surface area and particle size. We have emphasizing on temperature parameter and its effect on adsorption capacity. The change in temperature changes the equilibrium capacity of the adsorbent for a particular adsorbate. Then the combined waste water of sugar industry is treated with different doses (i.e. 5 gm/L, 10 gm/L, 40 gm/L, 80 gm/L, 120 gm/L, 160 gm/L, 200 gm/L) of zeolite. With increase in dosage of zeolite, considerable decrease in biochemical oxygen demand was observed. The adsorption capacity of zeolite was higher then other adsorbents. The results show that, as the adsorbent concentration increases, the amount adsorbed per unit mass (adsorption capacity qeq) of the adsorbent decreases considerably. The decrease in unit adsorption with increase in the dose of adsorbent is basically due to adsorption sites remaining unsaturated during the adsorption reaction. It can be suggested that adsorption capacity of zeolite is higher than other natural materials. The correlation coefficient values of Freundlich isotherm plots were found higher than that of Langmuir isotherm plot. So, Freundlich isotherm is more fitted than Langmuir isotherm, confirming multilayer and heterogeneous surface of adsorbent.
2. Experimental
Zeolite is natural alumino-silicates.
Zeolite was purchased from Fisher Sci. Comp. USA. Specific surface area of zeolite used is 2762.47cm2/gm. The combined waste water of sugar industry is treated with different doses (i.e. 5 gm/L, 10 gm/L, 40 gm/L, 80 gm/L, 120 gm/L, 160 gm/L, 200 gm/L) of zeolite, stirred well and kept in contact for 24 hours at 25 ̊C temperature then filtered. The glass bottle for sample collection should be thoroughly cleaned by rinsing with 8.0 M HNO3 followed by repeated washing with deionized distilled water. They should be rinsed thrice with the sample water before collection. Samples were stored at temperature below 3˚C to avoid any change in the physicochemical characteristics. The parameters like pH, conductance, alkalinity, COD, BOD, hardness and chloride were measured as per the standard method [22–23]. The pH and electrical conductivity of the samples were measured on the site and the other parameters were analyzed in the laboratory according to the Standard methods for the examination of water and waste water by American Public Health Association, Washington.
3. Results and Discussion
The process parameters like adsorbent dosage, temperature, contact duration, agitator speed are used for color removal, in which total color removal was attained using parameter of temperature. The equilibrium data have been analyzed using Langmuir and Freundlich isotherms, in which Langmuir isotherm is more fitted than Freundlich isotherm, derived from correlation coefficients. This study was carried out in two steps. The first step consisted of the characterization of the combined wastewater samples which is collected from the sugar industries of Bardoli (Gujarat). Samples were collected in sampling bottles and placed in icebox to preserve for analysis. The analyzed parameters were the pH, conductance, alkalinity, COD, BOD, hardness and chloride as per standard method [22]. In the second step physicochemical treatments like adsorption with zeolite applied to combined wastewater in order to reduce COD and BOD. The results for each dose are presented in Table 1, 2 and figure 1, 2.
Table 1 represents the effect of different dosages of Zeolite on various physico-chemical characteristics of combined waste water of Sugar Industry. The pH and alkalinity of the waste water remain unaffected at all dosages. The chloride content increases rapidly with increasing amount of Zeolite i.e. from 112.465mg/L (initial) to 337.346 mg/L. The conductance increases from 1.659 m mho to 2.95 m mho. Hardness increases upto some extent and then decreases with increasing amount of adsorbent. The COD content is found to be 1529.01 mg/L at initial stage and removed upto 1121.27 mg/L at 120gm/L of Zeolite dose and no further removal is found up to 200 gm/L. The initial BOD value is 910 mg/L which is slowly decreases to 630mg/L with 160gm/L of Zeolite and then remain unaffected for higher dose.
Data of Table 1 can be explained as the pH and alkalinity remain unaffected at all dosages. The chloride content increases rapidly with increasing dose; this may be due to the presence of some soluble salts having chloride in Zeolite. The increasing conductance suggests the introduction of ionic species in the system. Hardness increases up to some extent and then decreases with increasing amount of adsorbent, this can be explained considering the ion-exchange capacity of Zeolite. Zeolites are more effective for the removal of all kind of contaminants from the water and waste water than the other alumino-silicates as they have a typical feature of opening of their structure and their ion exchange character. The removal of COD and BOD can be explained on the basis of adsorption phenomena.
Adsorbent: Zeolite; Room temperature: 26±1 ̊C; Specific Surface Area: 2762.47 cm2/gm; Contact duration: 24 hours.
Table 2 represents the data for Freundlich and Langmuir adsorption isotherms along with percent removal of BOD contributing components from Sugar Industry waste water. A considerable decrease in BOD content per gram of adsorbent with increasing concentration of adsorbent was observed. The removal of BOD contributing components is found to decrease from 28 mg/gm to 1.40 mg/gm with increasing amount of Zeolite from 5gm/L to 200gm/L. The percent removal of BOD increases up to 30.77%.
Table 1. The effect of different dosages of Zeolite on various physico-chemical characteristic of the combined Waste Water Sugar Industry.
| Parameter | Untreated | 5 gm/L | 10 gm/L | 40 gm/L | 80 gm/L | 120 gm/L | 160 gm/L | 200 gm/L |
| pH | 6.61 | 8.17 | 8.17 | 8.17 | 8.17 | 8.17 | 8.17 | 8.17 |
| Conductance (m mho) | 1.659 | 2.21 | 2.21 | 2.21 | 2.21 | 2.45 | 2.70 | 2.95 |
| COD (mg/L) | 1529.01 | 1461.05 | 1427.07 | 1291.16 | 1189.23 | 1121.27 | 1121.27 | 1121.27 |
| BOD (mg/L) | 910 | 770 | 744 | 713 | 682 | 669.6 | 630 | 630 |
| Alkalinity (mg/L) | 975 | 975 | 975 | 975 | 975 | 975 | 975 | 975 |
| Hardness (mg/L) | 800 | 925 | 925 | 925 | 825 | 775 | 775 | 775 |
| Chloride (mg/L) | 112.465 | 124.961 | 137.457 | 187.442 | 199.938 | 237.426 | 274.936 | 337.395 |
Table 2. Freundlich and Langmuir adsorption isotherm parameters for BOD contributing components and percent removal of BOD from sugar industry waste water onto Zeolite.
| No | Adsorbent conc. m (gm/L) | Eq.Conc. Ceq (mg/L) | Removal x =C0-Ceq (mg/L) | qe= x/m (mg/gm) | Removal % | log Ceq | logx/m | 1/Ceqx103 | 1/qex102 |
| 1 | 0 | 910 | --- | --- | --- | 2.9590 | --- | 1.0989 | --- |
| 2 | 5 | 770 | 140 | 28 | 15.38 | 2.8864 | 1.4471 | 1.2987 | 3.5714 |
| 3 | 10 | 744 | 166 | 16.6 | 18.24 | 2.8716 | 1.2201 | 1.3441 | 6.0241 |
| 4 | 40 | 713 | 197 | 4.925 | 21.64 | 2.8531 | 0.6924 | 1.4025 | 20.3045 |
| 5 | 80 | 682 | 228 | 2.85 | 25.05 | 2.8338 | 0.4548 | 1.4662 | 35.0877 |
| 6 | 120 | 669.6 | 240.4 | 2.003 | 26.42 | 2.8258 | 0.3017 | 1.4934 | 49.9251 |
| 7 | 160 | 630 | 280 | 1.75 | 30.77 | 2.7993 | 0.2430 | 1.5873 | 57.1428 |
| 8 | 200 | 630 | 280 | 1.40 | 30.77 | 2.7993 | 0.1461 | 1.5873 | 71.4285 |
Adsorbent: Zeolite; Room temperature: 26±10C; Specific Surface Area: 2762.47 Cm2/ gm; Contact duration: 24 Hours.
The logarithmic values of equilibrium concentration (Ceq) and removal per unit weight (x/m) were given in table which were used for the explanation of Freundlich adsorption isotherm model and plot whereas the inverse values needed for Langmuir isotherm model.
Table 2 represents the data for Freundlich and Langmuir adsorption isotherms along with percent removal for BOD onto Zeolite. This information is used to prove the adsorption isotherm model and from that the Adsorption intensity, Adsorption energy and Adsorption capacity can be calculated. The percent removal of BOD seems to be increased with increase in dose of adsorbent. The logarithmic and inverse values of Ceq and x/m are used for plot of isotherm.
The logarithmic value of equilibrium concentration and removal per unit weight gives the linear plot for BOD by Zeolite confirms the applicability of Freundlich adsorption isotherm. It is the most widely used mathematical description of adsorption in aqueous systems. The equation is an empirical expression that covers the heterogeneity of the surface and exponential distribution of sites and their energies. With the purpose of linearization the equation is represented in logarithmic form as
log x/m = log K + 1/n log Ceq
The plot of log Ceq versus log x/m gives straight line with a slope of 1/n and log K is the intercept of log x/m at log Ceq = 0 which indicates that Freundlich adsorption isotherm model is applicable.
The same table shows the Langmuir adsorption isotherm for BOD by Zeolite. Langmuir isotherm is a plot of the amount of impurity adsorbed by Zeolite against the amount of impurity that remains in solution. It is a preliminary test to check the efficiency of particular material.
This mode of action can be explained on the basis of Langmuir’s model, i.e. ‘Ideal localized monolayer model’ according to which:
1. The molecules are adsorbed at definite sites on the surface of the adsorbent.
2. Each site can accommodate only one molecule (monolayer).
3. The area of each site is a fixed quantity determine solely by the geometry of the surface.
4. The adsorption energy is the same at all the sites.
Such behavior on the basis of kinetic consideration, presuming that the adsorbed molecules cannot migrate across the surface interacts with other neighboring molecules can be mathematically expressed as under:
1/qe = 1/Ө0b x 1/Ceq + 1/Ө0
Where,
qe = amount of solute adsorbed per unit weight of adsorbent(mg/gm)
= x/m i.e. x is amount of adsorbate adsorbed (mg/L) m=weight of adsorbent (gm/L)
Ceq = equilibrium concentration of the solute (mg/L)
Ө0 = Langmuir constant related to adsorption capacity (mg/gm)
b = Langmuir constant related to adsorption energy (L/mg)
Figure 1 represents the plot of log Ceq Vs log x/m (BOD) for zeolite. The straight line nature of the plot corresponds to slope 1/n and intercept K. 1/n is related to adsorption intensity whose value is 4.7642 for COD while intercept K on Y-axis related to adsorption capacity is found to be 0.068
Plot of log Ceq versus log x/m is a straight line in nature, presented in figure 1 suggests the applicability of this isotherm and indicate monolayer coverage of the adsorbate on the outer surface of the adsorbent. The steep slope indicates high adsorptive intensity at high equilibrium concentration that rapidly diminished at lower equilibrium concentration covered by the isotherm. As Freundlich equation indicates the adsorptive capacity x/m is a function of the equilibrium concentration of the solute. Therefore, higher capacity is obtained at higher equilibrium concentrations.
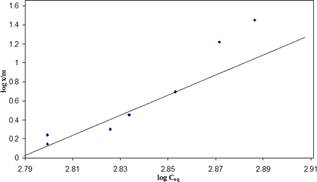
Figure 1. Freundlich adsorption isotherm for BOD contributing component onto Zeolite.
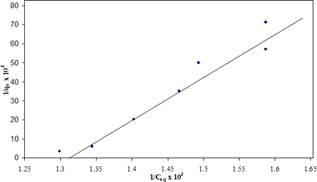
Figure 2. Langmuir adsorption isotherm for BOD contributing component on to Zeolite.
Fig. 2 represents the plot of Langmuir parameters viz, 1/Ceq x 103 and 1/qe x 103. The nature of the curve for BOD onto zeolite from sugar industry is linear however the intercept on X-axis related to adsorption energy (L/mg) i.e. b x 103 is 1.31 L/mg for BOD exerting components. These values can be used to calculate the adsorption capacity Ө0 i.e 39.048 (mg/gm).
The plot of Langmuir adsorption isotherm for BOD contributing components onto zeolite presented in fig. 2 is a straight line in nature which confirms the applicability of the Langmuir model and also the monolayer coverage. The Langmuir constant Ө0 in mg/gm related to adsorption capacity indicate availability of more surface active region onto adsorbent site and b x 103 L/mg related to adsorption energy in terms of x/m is a characteristic of the system.
4. Conclusion
In the present study we have successfully used zeolite with specific surface area of 2762.47cm2/gm for removal of biochemical oxygen demand from the combined waste water of sugar industry. Following results are obtained. (1)
i The maximum BOD removal is found at 160gm/L of zeolite concentration i.e. 30.77% from the combined waste water (2)
ii With increasing amount of Zeolite there found dramatic increase in chloride content (3)
iii At room temperature Zeolite works as an adsorbent and follow Freundlich and Langmuir isotherm models. The results give straight line which confirms the applicability of isotherm
iv The Freundlich constant K an intercept on X axis is related to adsorption capacity is found to be 0.068 while the slope 1/n is related to adsorption intensity is found to be 4.7642.
The straight line of the Langmuir plot gives intercept on Y axis called b x 103 L/mg i.e. adsorption energy is 1.31 L/mg and the calculated adsorption capacity Ө0 mg/gm is 39.048.
References
- Bansoda RR, Losso JN, Marshall WE, Rao, RM, Portier RJ (2004) Pecan shell-based granular activated carbon for treatment of chemical oxygen demand (COD) in municipal wastewater. J. Bioresour. Tech. 94:129–135.
- Ahmedna M, Marshall WE, Rao RM (2000) Production of granular activated carbons from selected agricultural by-products and evaluation of their physical, chemical and adsorption properties. J. Bioresour. Tech. 71:113–123.
- Pala A, Tokat E (2000) Color removal from cotton textile industry wastewater in an activated sludge system with various additives. J Water Res 36:2920–2925.
- Guohua C (2004) Electrochemical technologies in wastewater treatment. Sep Purif Tech 38(1):11–41.
- Martinez NS, Fernandez JF, Segura XF, Ferrer AS (2003) Pre-oxidation of an extremely polluted industrial wastewater by the Fenton’s reagent. J. Hazard. Mater. B101:315–322.
- Hung CM, Lou JC, Lin CH (2003) Removal of ammonia solutions used in catalytic wet oxidation processes. J. Chemosphere. 52:989–995.
- Xiong Y, Strunk P, Xia H, Zhu X, Karlsson H (2000) Treatment of dye wastewater contain acidic orange II using a cell with three-phase three dimensional electrodes. J. Water Res. 35(17):4226–4230.
- Gogate PR, Pandit AB (2004) A review of imperative technologies for wastewater treatment 11: hybrid methods. J. Adv. Environ. Res. 8(3/4):501–551.
- Kolaković S, Stefanović D, Milićević D, Trajković S, Milenković S, Kolaković SS., Anđelković L (2013) Effects of reactive filters based on modified zeolite in dairy industry wastewater treatment process. Chemical Industry & Chemical Engineering Quarterly 19(4):583−592.
- Zhang M, Zhang H, Xu D, Han L, Zhang J, Zhang L, Wu W, Tian B(2011)Removal of Phosphate from Aqueous Solution Using Zeolite Synthesized from Fly Ash by Alkaline Fusion Followed by Hydrothermal Treatment.Sep. Sci. Technol. 46(14):2260-2274.
- Zhang BH, Wu D, Wang C, He S, Zhang ZJ, Kong HN (2007)Simultaneous removal of ammonium and phosphate by zeolite synthesized from coal fly ash as influenced by acid treatment. J. Environ. Sci. 19(5):540-545.
- Lin L, Zhongfang L, Wang L, Liu X, Zhang Y, Wan C, Lee DJ, Tay JH (2013) Adsorption mechanisms of high-levels of ammonium onto natural and NaCl-modified zeolites. Separation and Purification Technology 103:15–20.
- Stefanakis AI, Akratos CS, Gikas GD, Tsihrintzis VA (2009) Effluent quality improvement of two pilot-scale, horizontal subsurface flow constructed wetlands using natural zeolite (clinoptilolite). Microporous Mesoporous Mater 124:131–143.
- Tian WH, Wen XH, Qian Y (2004) Using a zeolite medium biofilter to remove organic pollutant and ammonia simultaneously. J. Environ. Sci. 16(1):90-93.
- Qiu L, Zhang S, Wang G, Du M (2010) Performances and nitrification properties of biological aerated filters with zeolite, ceramic particle and carbonate media. Bioresour. Technol. 101(19):7245–7251.
- Ji G, Tong J, Tan Y (2011) Wastewater treatment efficiency of a multi-media biological aerated filter (MBAF) containing clinoptilolite and bioceramsite in a brick-wall embedded design. Bioresour. Technol. 102(2):550–557.
- Syafalni S, Abustan I, Dahlan I, Wah CK, Umar G (2012) Treatment of dye wastewater using granular activated carbon and zeolite filter. Mod. Appl. Sci. 6(2):37–51.
- Erdem E, Karapinar N, Donat R (2004) The removal of heavy metal cations by natural zeolites. J. Colloid Interface Sci. 280:309–314
- Hui KS, Chao CYH, Kot SC (2005) Removal of mixed heavy metal ions in wastewater by zeolite 4A and residual products from recycled coal fly ash. J. Hazard. Mater. 127:89–101.
- Balasubramaniam N, Raja ER, Lalith K, Prabha S (1998) Adsorption Dynamics: Comparison of Adsorption Potentials of Fibrous Keratinous Materials, viz., Human Black and White Hairs. Asian J of Chemistry 10(1):136–145.
- Venkateswarlu P, Venkata Ratnam M, Subba Rao D, Venkateswara Rao M (2007) Removal of chromium from an aqueous solution using Azadirachta indica (neem) leaf powder as an adsorbent. Int. J. Phys. Sci. 2 (8):188–195.
- APHA (American Public Health Association), 1995. Standard methods for the examination of water and wastewater [S]. 19th ed. Washington, D. C.: APHA
- Rand MC, Greenberg AE, Taras MJ (1976) Standard methods for the examination of water and waste water – APHA 14th ed., 42.



