Effect of Sodium Citrate Concentrations on Dissolution Performance of ScrapLead Paste
Chunxia Gong1, 2, *, Yanjuan Chen1, Xiaofei Zhang1, Qihui Hu1, Yu Cheng1, Mingxue Fang1, Duyou Wang1, Qun Chen1
1Zhejiang Tianneng battery (Jiangsu) Co., Ltd., Shuyang Jiangsu, China
2School of Chemistry and Chemical Engineering, Southeast University, Nanjing, China
Abstract
In recent years, lead poisoning eventshave occurred frequently. Lead metal impacts body's nervous and blood system significant. the people should appear arrhythmia, renal failure, convulsions, coma and even death if lead levels in the blood of pepole were more than 1000 mg/L. Therefore, it is very important for the survival of human to research the properties of heavy metal lead. In this paper dissolution performance of scrap lead paste with different sodium citrate concentrations has been researched. The photos show the color of solution turning to black from gray with 8.3%-30% sodium citrate solution. pH values of filtrate were all higher than seven. 0.1~ 0.3g sodium citrates were consumed by per gram scrap lead paste. The results show not only the concentrations of Pb2+ in filter liquor but also the dissolution rates of scrap lead paste increase with the increase of concentration of the sodium citrate.
Keywords
Dissolution Performance, Scrap Lead Paste, pH, Concentrations, Sodium Citrate
Received: March 26, 2015
Accepted: April 9, 2015
Published online: April 10, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Our living standard has been continuously improved with the development of society and technology, however, the resources also been consumed unceasingly, and the environmental problem has been increasingly serious. Energy and environmental protection have become two hot issues.
In recent years, lead poisoning events have occurred frequently. Lead metal impacts body's nervous and blood system significant, the people should appear arrhythmia, renal failure, convulsions, coma and even death if lead levels in the blood of pepole were more than 1000 mg/L. Therefore, it is very important for the survival of human to research the properties of heavy metal lead.
At present, lead acid battery for its simple construction, reliable performance, lower price, easy get of raw materials, and many other advantages, has become the one kind of widespread used second batteries1-2. It is mainly used as power source for automobile start and as auxiliary power supply for illumination, minitype electrombile and UPS(Uninterrupted Power Supply)3-5. Lead acid battery as primary consumption product all over the world, at the same time, it also become the major lead renewable resources. Waste lead-acid battery is a hazardous waste, such as to be irresponsibly disposed; the heavy metal by decomposing will bring serious pollution to the environment and extreme harm to human health. Therefore, recycling and reusing of lead from scrap lead acid battery is very important and is the fundamental approach to solve the pollution.
When charging, the cathode material of lead acid battery turns into PbO2 and anode material turns into Pb. While discharge, the cathode and anode electrode active materials all turn into PbSO4. The discharge reactions of materials are as follows,
Cathode reaction, PbO2 + HSO4− + 3H+ + 2e− ![]() PbSO4 + 2H2O
PbSO4 + 2H2O
Anode reaction, Pb + HSO4− ![]() PbSO4 + H+ + 2e−
PbSO4 + H+ + 2e−
Overall reaction, Pb + PbO2 + 2H2SO4 ![]() PbSO4 + 2H2O
PbSO4 + 2H2O
The manufacturing processes of lead acid battery mainly include exploit and transformation of resource, preparation of material for battery and assemble of battery. Among the process of preparation of material and assemble of battery, especially in the process of preparation of electrode, plenty of lead paste will fall off as scrap lead paste. Cyclic utilization of scrap lead paste is a significant measure which would reduce the batteries production fee and environmental pollution of heavy metal lead. The scrap lead paste mainly contains PbSO4 andPbO2; therefore, desulphurization methods for scrap lead paste are important among cyclic utilization of scrap lead paste. The main desulphurization methods are Pyrometallurgy 6-7 and Hydrometallurgy method8. Among Hydrometallurgy method, the follow desulfurization agent will be usually used: Carbonate 9-10 (RSR11-12 and USBM13 technologies use (NH4)2CO3 as desulfurization agent, CX-EW14 technology uses Na2CO3 as desulfurization agent); NaOH 15,16; NaCl-HCl 17; citric acid-sodium citrate (C6H8O7·H2O-Na3C6H5O7·H2O)18-22; sulfate-reducingbacteria (CX-EWS technology)23and so on.
At this stage, many researchers have used sodium citrate hydrometallurgy18-22 to dope with scrap lead paste; therefore, dissolution performance of scrap lead paste with different sodium citrate concentrations is very important for dope with scrap lead paste.
2. Experimental
The sodium citrate solutions (8.3%, 15%, 20%, 25%) were prepared as follows: each of 13.8 ml, 25 ml, 33 ml or 41.7ml of Na3C6H5O7×2H2O (30%) was constant volume to 50 ml with deionized water.
Experimental process as follows: 5 g of scrap lead paste was added to 50 ml Na3C6H5O7×2H2O solution under vigorous stirring. After the slurry had been stirred for 5 min at 20°C in air atmosphere, it was filtered, washed with distilled water for three times, and then dried at 60°C. The volume and pH value of filtrates and the masses of filter cakes were wrote down. The filtrates were diluted 2000 times and then tested the concentrates of Pb2+ in filter liquor using atomic absorption spectroscopy (AAS). The dissolution rate of scrap lead paste was calculated by follow pattern.
![]()
r, dissolution rate; c, concentrates of Pb2+ infiltrates; V, volume of filtrates; m, mass of scrap lead paste.
3. Results and Discussion
3.1. The Color of Slurries


Fig. 1. The color of filter liquor in different sodium citrate concentrations.
The photos of slurries in different sodium citrate concentrations were shown in fig. 1. The colors of slurries turn to black from gray with 8.3%-30% sodium citrate solution. It can be seen that higher the concentrations of sodium citrate, the more color changes of scrap lead paste.
3.2. pH Value of Filtrates
The pH curve of filtrates in different sodium citrate concentrations was shown in fig 2. It can be seen from the curve that pH value of filtrates were all higher than 7 but lower than 7.5 after solved by different concentrates of Na3C6H5O7×2H2O. Therefore, filtrates are neutral after solved by sodium citrate of different concentrations.
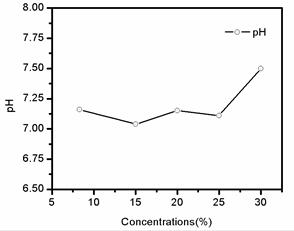
Fig. 2. pH curve of the filtrates in different sodium citrate concentrations.
3.3. Mass of Sodium Citrate Consumed by Scrap Lead Paste per Gram
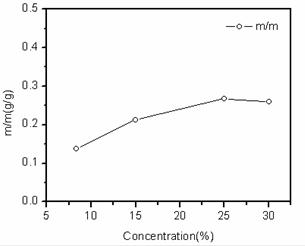
Fig. 3. Mass of sodium citrate waste by lead paste per gram with different sodium citrate concentrations.
Fig. 3 shows the mass of sodium citrate consumed by scrap lead paste per gram. The mass of sodium citrate consumed by scrap lead paste per gram was 0.1~0.3g, higher the concentrations of sodium citrate, more the mass of sodium citrate was consumed. Therefore, dissolution rates of scrap lead paste increased by increasing of the sodium citrate concentration.
3.4. Percentages of Solid Remains in Sodium Citrate
Fig. 4 shows the percentages of solid remains in sodium citrate with different concentrations. The percentages of solid remains with different concentrations of sodium citrate were from 5% to 25%. It shows a decrease of the percentages of solid remains with higher the concentrations of sodium citrate. Therefore, dissolution rates of scrap lead paste increased by increasing of the sodium citrate concentration. This result was consistent with the result of fig 3.
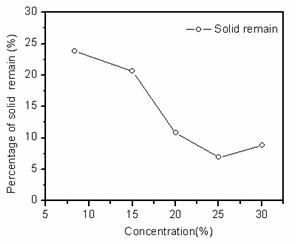
Fig. 4. Percentage of solid remains in sodium citrate with different sodium citrate concentrations.
3.5. Concentrations of Pb2+ in Filtrates
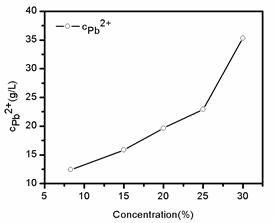
Fig. 5. Concentrations of Pb2+ in filter liquor with different sodium citrate concentrations.
Fig. 5 shows the concentrations of Pb2+ in filtrates with different concentrations of sodium citrate. The concentrations of Pb2+ in filtrates with different concentrations of sodium citrate were from 12.42 to 35.35 g·L-1. The concentration of Pb2+ in filtrate was increased by increasing the concentration of sodium citrate. Therefore, dissolution rates of scrap lead paste increased by increasingof the sodium citrate concentration. This result was also consistent with the results of fig 3 and fig 4.
3.6. Dissolution Rates of Scrap Lead Paste
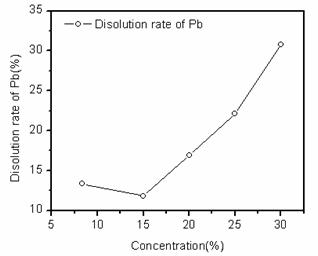
Fig. 6. Dissolution rates of scrap lead paste in different concentrations of sodium citrate.
Fig. 6 shows the dissolution rates of scrap lead paste in different concentrations of sodium citrate. The dissolution rates of scrap lead paste in different concentrations of sodium citrate were from 11.8% to 30.8%. Higher the concentrations of sodium citrate, the dissolution rates of scrap lead paste in filtrates were also higher. Therefore, dissolution rates of scrap lead paste increased by increasing of the sodium citrate concentration. This result was same as the results above all.
4. Conclusions
The colors of slurries turn to black from gray with 8.3%-30% sodium citrate solutions. Filtrates are neutral after solved by sodium citrate of different concentrations. There were consumed 0.1~0.3g sodium citrate by scrap lead paste per gram. Concentration of Pb2+ (12.42 ~35.35 g·L-1) in filter liquor was increased by increasing of the sodium citrate concentration. The dissolution rates of scrap lead paste in different concentrations of sodium citrate were from 11.8% to 30.8%. Higher the concentrations of sodium citrate, the dissolution rates of scrap lead paste in sodium citrate water solution were also higher. It is very important for the survival of human to research the properties of heavy metal lead.
Acknowledgements
We would like to thank the help of Hui Zhang, Bin Wang, Lifang Zhang in Zhejiang Tianneng battery (Jiangsu) Co., Ltd. and Jinxi Chen in School of Chemistry and Chemical Engineering, Southeast University.
References
- S.M. Tabaatabaai, M.S. Rahmanifar and S.A. Mousavi, "Lead-acid batteries with foam grids,"[J]. Journal of Power Sources, vol.158, pp.879-884, 2006.
- Y. Chen, B.Z. Chen and X.C. Shi, "Preparation and electrochemical properties of pitch-based carbon foam as current collectors for lead acid batteries,"[J]. Electrochimica Acta,vol.53,pp. 2245-2249, 2008.
- M.L. Soria, J. Valenciano and A. Ojeda, "Development of ultra high power, valve-regulated lead-acid batteries for industrial applications,"[J]. Journal of Power Sources,vol.136, pp.376-382, 2004.
- D. Pavlov, G. Petkova, M. Dimitrov, "Influence of fast charge on the life cycle of positive lead-acid battery plates,"[J].Journal of Power Sources, vol.87, pp.39-56, 2000.
- C.H. Yeh, C.C. Wan and J.S. Chen, "Physical and electrochemical characterization of PbO2 electrode prepared at different H2SO4/H2O/PbO ratios,"[J]. Journal of Power Sources, vol.101, pp.219-225, 2001.
- J. M. Jiang, "Situation and development trend of lead smelting technology at home and abroad,"[J].Energy Saving of Non-Ferrous Metallurgy,vol.3, pp.4-8,2013.(in Chinese)
- S. K. Guo, A. P. He, A. Q. Liu, et al. "New pyrometallurgi-cal processes and equipment for smelting lead," [J]. Shanghai Nonferrous Metals,vol.23, pp.20-23, 2002.(in Chinese)
- X.J. Sun, J.K. Yang, W. Zhang, et al. "Lead acetate trihydrate precursor route to synthesize novel ultrafine lead oxide from spent lead acid battery pastes," [J]. Journal of Power Sources, vol.269, pp. 565-576, 2014.
- X. Zhu, J. Yang, L. Gao, et al. "Preparation of lead carbonate from spent lead paste via chemical conversion,"[J]. Hydrometallurgy, vol.134-135, pp.47-53, 2013.
- X. Zhu, L. Li, X. Sun, et al."Preparation of basic lead oxide from spent lead acid battery paste via chemical conversion,"[J]. Hydrometallurgy, vol.117-118, pp. 24-31, 2012.
- H.D. McDonald, "Method of recovering lead values from battery sludge,"[P]. United States Patent, 4229271. 1980-10-21
- R.D. Prengaman. "Recovering lead from batteries,"[J]. Jom-Journal of the Minerals Metals& Materials Society, vol.47, pp.31-33, 1995.
- E.R. Cole, A.Y. Lee and D.L. Paulson, "Update on recovering lead from scrap batteries,"[J]. Journal of Metals, vol.37, pp. 79-83, 1985.
- M. Olper, P. Fracchia, "Hydrometallurgica1 process for an overall recovery of the components of exhausted lead-acid batteries,"[P].United States Patent, 4769116. 1988-9-6
- W.P. Chen, J.S. Gong and Q.Z. Li, "Researeh on Proeess for RedueingPbO:Contained in Lead Baffery Serapwith Fe2+,"[J]. Journal of Hunan University, vol.22, pp.53-58, 1995. (in Chinese)
- W.P. Chen, "New Technology of Hydrometallurgical Recovering Lead from Waste Battery Slurry,"[J]. Journal of Hunan University, vol.23, pp.111-115, 1996 (in Chinese)
- D. Andrews, A. Raychaudhuri, "Frias C. Environmentally sound technologies for recycling secondary lead,"[J]. Journal of Power Sources, vol.88, pp.124-129, 2000.
- M. S. Sonmez, R. V. Kumar, "Leaching of waste battery paste components: Part 1. Lead citrate synthesis from PbO and PbO2," [J]. Hydrometallurgy, vol.95, pp.53-60, 2009.
- J. K. Yang, X. F. Zhu, W. C. Liu, et al. "Review of recovery technology for lead paste from spent lead acid battery," [J]. Mod Chem Ind, vol.29, pp.32-36, 2009.
- M.S. Sonmez and R.V. Kumar, "Leaching of waste battery paste components. Part 2: Leaching and desulphurisation of PbSO4 by citric acid and sodium citrate solution,"[J]. Hydrometallurgy, vol.95, pp.82-86, 2009.
- X. Zhu, X. He, J. Yang, et al. "Leaching of spent lead acid battery paste components by sodium citrate and acetic acid,"[J]. Journal of Hazardous Materials, vol.250-251, pp.387-396, 2013.
- D.N. Yang, J.W. Liu, Q. Wang, et al. "A novel ultrafine leady oxide prepared from spent lead pastes for application as cathode of lead acid battery,"[J]. Journal of Power Sources, vol.257, pp.27-36, 2014.
- M. Olper, M. Maccagni and C.J.N. Buisman, "Electrowinning of lead battery paste with the production of lead and elemental sulphur using bioprocess technologies,"[C]. proceedings of the LEAD~ZINC 2000 symposium, Pittsburgh Pennsylvania USA, Minerals Met&Mat Soc, pp.209-210, 2000



