Novel Catalytic Synthesis of Xanthene and 1,8-Dioxooctahydroxanthene Derivatives Using ZnO Nanoparticles Catalyst Under Green Conditions
Ali Gharib1, 2, *, Leila Vojdani Fard3, Nader Noroozi Pesyan4, Mina Roshani1
1Department of Chemistry, Islamic Azad University, Mashhad, Iran
2Department of Chemistry, Agricultural Researches and Services Center, Mashhad, Iran
3Education Ministry, Education Organization of Razavi Khorasan, Mashhad, Iran
4Department of Chemistry, Faculty of Science, Urmia University, Urmia, Iran
Abstract
These catalytic condensation reactions represent green chemical processes and ZnO nanoparticles catalyst is cost-effective, easy to handle, and easily removed from the reaction mixtures and also, ZnO nanoparticles catalyst was found to be highly efficient, eco-friendly and recyclable heterogeneous catalyst for the multicomponent reaction of dimedone, aromatic aldehydes, and a nitrogen source (ammonium acetate or aromatic amines) under solvent-free conditions, giving rise to 1,8-dioxodecahydroacridines in high yields.
Keywords
Xanthene, β-naphthol, Condensation, Aromatic Aldehydes, Nanoparticles, Catalyst
Received: March 21, 2015
Accepted: April 6, 2015
Published online: April 19, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Xanthenes and benzoxanthenes are important class of compounds that are found in numerous biologically active molecules. This structural motif has also been investigated for a wide range of activities such as bactericidal [1], anti-inflammatory [2], antiviral [3], as well as photodynamic therapy [4], In particular, the xanthenedione structure is present in a number of natural products [5], and has been a component of dyes [6], fluorescent materials for visualization of biomolecules and in laser technologies due to their spectroscopic properties [7‐9]. Consequently, several methods have been developed for the synthesis of xanthene derivatives, which in general can be obtained by the condensation of appropriate active methylene derivatives with aldehydes catalyzed by mineral acids [10]. Xanthenes were also prepared by the cyclization of polycyclic aryltriflate esters [11], catalyzed by palladium or the reaction of aryl magnesium halides with triethylorthoformate [12,13]. 14-Substituted-14H-dibenzo[a,j]xanthene derivatives have been found to possess a broad spectrum of biological activities, including antibacterial [14], anti-inflammatory [15], and antiviral activities [16]. Furthermore, these compounds have been used in photodynamic therapy [17] and as antagonists for paralyzing the action of zoxazolamine [18,19]. These compounds have also been used as dyes [20,21], in laser technology [22], and in fluorescent materials for visualization of biomolecules [23] and as pH sensitive fluorescent materials for the visualization of biomolecules [24]. Classically, xanthene derivatives were synthesized either by the reaction of aryloxymagnesium halides with triethylorthoformate [25], the trapping of benzynes with phenols [26], or by the reaction of β-naphthol with 2-naphthol-1-methanol [27], formamide [28], carbon monoxide [29], and aldehydes or acetals [30,31]. Unfortunately, many of these existing methodologies suffer from one or more disadvantages such as prolonged reaction times, low yields, use of harmful organic solvents, requirement for an excess of catalyst/reagents, and harsh reaction conditions. 1,8-dioxooctahydroxanthenes are important class of oxygen heterocycle in which a phenyl substituted pyran ring is fused on either side with two cyclohexenone rings. Presence of conjugated bis-dienone functionality makes these compounds sensitive to attack by nucleophiles and light energy. In the recent past, there is a renewed interest in the synthesis of this class of compounds as number of its applications increased, both in the field of medicinal chemistry and material science. 1,8-dioxo-octahydroxanthenes shows useful biological activities such as anti-inflammatory, antibacterial, antiviral activities [32], finds use in biodegradable agrochemicals [33,34], cosmetics and pigments [35], fluorescent materials [36], photodynamic therapy [37], luminescent sensors [38], and in laser technologies [39]. The formation of intermediate, 2,2’-arylmethylenebis(3-hydroxy-5,5-dimethyl-2-cyclohexene-1-one), in the synthesis of 1,8-dioxooctahydroxanthenes can take place even without a catalyst [40]. However, there is always need for a catalyst in the cyclization step to get the cyclized compound [41-43]. ZnO nanoparticles catalyst was found to be highly efficient, eco-friendly and recyclable heterogeneous catalyst for the multicomponent reaction of dimedone, aromatic aldehydes, and a nitrogen source (ammonium acetate or aromatic amines) under solvent-free conditions, giving rise to 1,8-dioxodecahydroacridines in high yields [44]. To react two molecules of dimedone (5,5-dimethyl-1,3-cyclohexane dione) with various aromatic aldehydes [45], by using of different Lewis acid catalysts such as triethylbenzyl ammonium chloride [46], p-dodecyl benzenesulfonic acid [47], diammonium hydrogen phosphate under various conditions [48], sulfonic acid under ultrasonic irradiation [49], ionic liquids [50], Amberlyst-15 [51], NaHSO4-SiO2 or silica chloride [52], phosphomolybdic acid supported on silica gel [53], nanosized MCM-41-SO3H under ultrasonic irradiation [54], sulfonic acid on silica gel [55], Dowex-50W ion exchange resin under solvent-free conditions [56], HClO4-SiO2 [57], ZnO and ZnO-acetyl chloride [58] and heteropoly acid supported MCM-41[59].
2. Experimental Section
2.1. Instrumentation
All reactions were carried out without any special precautions in an atmosphere of air. Chemicals were purchased from Fluka and S. D. Fine Chemicals and directly used for the synthesis. Thin‐layer chromatography (TLC): precoated silica gel plates (60 F254, 0.2 mm layer; E. Merck). 1H NMR and 13C NMR (Innova 400 MHz and Brucker Gemini 100 MHz) spectra were recorded in CDCl3 using TMS as internal standard. IR and MS were recorded on a Thermo Nicolet Nexus 670 FT‐IR spectrometer and Finnegan MAT 1020 mass spectrometer operating at 70 eV.
2.2. General Procedure for the Synthesis of 1,8-Dioxooctahydroxanthenes Derivatives in Water (Scheme 1)
To a solution of aromatic aldehyde (2 mmol) and dimedone or 1,3‐diketone (2 mmol) in water (10 mL) was added ZnO nanoparticles catalyst (1 mmol). Then The mixture was refluxed for the appropriate reaction time. After completion of the reaction (the progress of the reaction was monitored by TLC using n-hexan: ethylacetate as eluent), the mixture was cooled and the solid residue was separated and dissolved in dichloromethane. The solution was filtered and ZnO nanoparticle catalyst was isolated and could be reused. The organic phase was evaporated under reduced pressure and the reaction mixture was recrystallized in ethanol to give pure product (Scheme 1, Table 1). All the products are known compounds and the spectral properties and melting points of them matched well with those reported earlier [60-62]. Spectroscopic and physical data of some representative compounds are given below:
3,3,6,6-Tetramethyl-9-phenyl-3,4,6,7-tetrahydro-2H-xanthene-1,8(5H,9H)dione (4a):
M.p.: 203‐205 oC; 1H NMR (400 MHz, CDCl3): δH 1.11 (s, 6H, 2´CH3), 1.25 (s, 6H, 2´CH3), 2.47‐2.25 (m, 8H, 4´CH2), 5.47 (s, 1H, CH), 7.02‐7.06 (d, J = 7.93 Hz, 2H, ArH), 7.09‐7.15 (m, 1H, ArH), 7.24‐7.19 (m, 2H, ArH). 13C NMR (100 MHz, CDCl3): δC 27.4, 29.6, 31.3, 32.6, 46.4, 46.9, 115.5, 125.7, 126.8, 128.2, 137.9, 189.3, 190.3. FT‐IR (KBr, cm‐1): ν 2962, 2928, 1592, 1372, 1159, 1041, 842, 774, 692. Anal Calcd for C23H26O3: C 78.83, H 7.48. Found: C 78.53, H 7.20. HRMS (EI) Calcd. for C23H26O3 [M]+, 350.2002, Found 680.1008.
9-(3-chlorophenyl)-3,3,6,6-tetramethyl-3,4,5,6,7,9-hexahydro-1H-xanthene-1,8(2H)-dione (4c):
M.p.: 182-164 oC; 1H NMR (400 MHz, CDCl3): δH 1.01 (s, 6H, 2´CH3); 1.14 (s, 6H, 2´CH3); 2.32 (dd, J= 1.7, J = 3.4 Hz, 4H, 2´CH2); 2.48 (s, 4H, 2´CH2); 4.77(s, 1H, CH); 4.66; 7.15-7.27 (m, 4H, Ar); 13C NMR (100 MHz, CDCl3): δC 196.1, 162.3, 159.2, 145.6, 128.9, 120.7, 115.5, 114.2, 111.8, 55.1, 50.6, 40.8, 32.0, 31.6, 29.1, 27.4; 13C NMR (100 MHz, CDCl3): δC 36.1, 117.2, 118.4, 123.5, 123.9, 125.0, 127.6, 128.7, 129.0, 129.5, 130.3, 131.1, 131.2, 144.8, 148.5. FT-IR (KBr, cm-1): ν 3032, 2976, 1680, 1662, 1622, 1494, 1476, 1364, 1200, 1174, 1136, 1097, 1000, 855, 840; Anal. Calcd for C23H25ClO3: C 71.77; H, 6.55; Found: C 71.76, H, 6.53. HRMS (EI) Calcd. for C23H25ClO3 [M]+, 384.1001, Found 384.1004.
9-(4-Bromophenyl)-3,3,6,6-tetramethyl-3,4,5,6,7,9-hexahydro-2H-xanthene-1,8-dione (4d):
M.p.: 241-242 oC;1H NMR (400 MHz, CDCl3): δH 0.97 (s, 6H, C(CH3)2; 1.12 (s, 6H, C(CH3)2); 2.12-2.21 (q, J= 16.60 Hz, 4H, 2´CH2); 2.40 (s, 4H, 2´CH2); 4.63 (s, 1H, CH); 7.10(d, J= 8.30 Hz, 2H, Ar) 7.27 (d, J= 8.30 Hz, 2H, Ar); 13C NMR (100 MHz, CDCl3): δC 196.1, 162.4, 143.1, 131.1, 130.2, 120.3, 115.4, 50.8, 40.5, 31.3, 29.1, 27.3 (4´CH3); FT‐IR (KBr, cm‐1): ν 3440, 2931, 1662, 1584, 1360, 1272, 1200, 1135, 1046, 695, 570. Anal Calcd for C23H25BrO3: C 64.34; H 5.87; Found: C 64.31, H 5.85. HRMS (EI) Calcd. for C23H25BrO3 [M]+, 428.1002, Found 428.1005.
14-(4-Chlorophenyl)-14H-dibenzo[a,j]xanthene (4i):
M.p.: 291-292 °C; 1H NMR (400 MHz, CDCl3): δH 6.74 (s, 1H), 7.16 (d, J = 6.8 Hz, 2H), 7.45-7.62 (m, 10H), 8.90 (d, J = 7.79 Hz, 2H), 8.68 (d, J = 7.6 Hz, 2H). 13C NMR (100 MHz, CDCl3): δC 36.2, 117.3, 118.1, 123.5, 123.8, 125.0, 127.4, 128.8, 129.1, 129.6, 130.1, 131.1, 131.2, 144.8, 148.5. FT‐IR (KBr, cm–1): ν 3066, 2922, 1622, 1590, 1514, 1456, 1397, 1245, 1237, 1209, 1140. Anal Calcd for C27H17ClO: C 82.54, H 4.36. Found: C 82.32, H 4.13. HRMS (EI) Calcd. for C27H17ClO [M]+, 392.2001, Found 392.2009.
9-(3-Methoxyphenyl)-3,3,6,6-tetramethyl-3,4,5,6,7,9-hexahydro-2H-xanthene-1,8-dione (4u):
M.p.: 159-161 oC; 1H NMR (400 MHz, CDCl3): δH 1.03 (s, 6H, C(CH3)2); 1.13 (s, 6H, C(CH3)2); 2.10-2.21 (q, J= 15.86 Hz, 4H, 2´CH2); 2.45 (s, 4H, 2´CH2); 3.39 (s, 3H, OCH3); 4.69 (s, 1H, CH); 6.56-7.08 (m, 4H, Ar); 13C NMR (100 MHz, CDCl3): δC 196.1, 162.3, 159.2, 145.6, 128.9, 120.7, 115.5, 114.2, 111.8, 55.1, 50.6, 40.8, 32.0, 31.6, 29.1, 27.4; FT-IR (KBr, cm-1): ν 3440, 2931, 1660, 1587, 1360, 1276, 1200, 1136, 1046, 695, 574; Anal. Calcd for C24H28O4: C, 75.76; H, 7.42; Found: C, 75.75, H, 7.40. HRMS (EI) Calcd. for C24H28O4 [M]+, 380.2001, Found 380.2006.
2.3. General Procedure for the Preparation of 14-aryl-14H-dibenzo[a,j]xanthenes (Scheme 2, Table 5)
To a solution of aromatic aldehyde (2 mmol) and β-naphthol (2 mmol) in water (10 mL) was added ZnO nanoparticles catalyst (1 mmol). The mixture was refluxed for the appropriate reaction time. Upon completion of the reaction, as indicated by TLC, the organic phase was washed with 1 mol/L aque ous NaOH solution (1 mL). The organic phase was then collected and the solvent removed in vacuo to give the crude product, which was purified by recrystallization from hot ethanol to afford pure product.
2.4. Catalyst Synthesis Procedure
Zinc oxide nanoparticle used in the experiments was produced in according to the procedure [49] with modification in following manner. Zinc powder 0.019 g (0.3 mmol) in dust form was sonicated for 2 h with mL of n-butanol. To the above solution, 1.2 mL of triethanolamine (TEA) was added slowly. It was then sonicated for ten more minutes. Finally, the mixture was irradiated in a closed vessel mono-mode microwave reactor at 140 °C and 10.9 bar for 6 min. The obtained white solid suspension was centrifuged, washed several times with distilled water and vacuumn dried. It was then calcined at 900 °C for 1 h and can be stored for extended period of time.
2.5. Characterization of Catalyst
The obtained particles were characterized by X-ray powder diffraction (XRD). The XRD pattern of the newly prepared zinc oxide nanoparticles showed the presence of peaks, which corresponds to hexagonal wurtzite structure [63-65]. The size of the particles has been computed from the width of first peak using Debye Scherrer formula:
D = K λ/β COS θ
here K is constant, λ is the wavelength of X-rays employed radiation (1.54056 Å), β is corrected full width at half maximum and θ is Bragg angle. The 2θ value is 19.0104. The crystallite size of the powder particles is calculated as about 30 nm [64,65].
3. Results and Discussions
Aqueous phase organic synthesis has attracted the attention of chemists as it overcomes the harmful effects associated with the organic solvents and is environmentally benign. In view of the above, the development of a generally applicable and environmentally benign methodology for the synthesis of xanthenes derivatives is highly desirable. we report herein an aqueous phase synthesis of 1,8‐dioxooctahydroxanthenes from 1,3‐cyclohexanedione and aromatic aldehydes in the presence of ZnO nanoparticles catalyst. Multicomponent reactions (MCRs) constitute an especially attractive synthetic strategy for rapid and efficient library generation because the products are formed in a single step and diversity can be achieved simply by varying the reaction components. Thus, new routes utilizing a MCR protocol for the synthesis of these molecules can attract considerable attention in the search for rapid-entry methods to these heterocycles. We wish to report for the first time a facile and efficient synthetic strategy for the preparation of 1,8-dioxo-octahydroxanthenes by the reaction of dimedone with aromatic aldehydes at reflux and using ZnO nanoparticles as a heterogeneous catalyst under green solvent (water) conditions (Scheme 1).
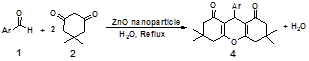
Figure 1. Synthesis of 1,8-dioxooctahydroxanthene using ZnO nanoparticles catalyst.
First, to determine the optimum quantity of ZnO nanoparticles catalyst, the reaction of dimedone and 2,5-dimethoxybenzaldehyde was carried out under reflux and water as green solvent conditions, using different quantities of ZnO nanoparticles as green catalyst (Table 1). As can be seen from Table 1, 0.08 g of ZnO nanoparticles gave an excellent yield in 20 min. Blank experiments in the absence of the catalyst showed that the reaction did not give corresponding 3,3,6,6-tetramethyl-9-phenyl-3,4,5,6,7,9-hexahydro-1H-xanthene-1,8(2H)-dione (Table 1, Entry 6). Thus, the catalyst had a catalytic effect on the mentioned reaction. The efficiency of the reaction is affected mainly by the amount of ZnO nanoparticles (Table 1). No product was obtained in the absence of the catalyst even after 200 min (Table 1, entry 6) indicating that the catalyst is necessary for the reaction. Increasing the amount of the catalyst increased the yield of the product 4a. The optimal amount of ZnO nanoparticles was 0.08 g (Table 1, entry 3); increasing the amount of the catalyst beyond this value did not increase the yield noticeably (Table 1, entries 4, 5).
Table 1. Optimization of the quantity of ZnO nanoparticles catalyst in the reaction of dimedone and 2,5-dimethoxybenzaldehyde under reflux conditions and water as green solvent.
| Entry | Catalyst (g) | Time (min) | aYield (%) |
| 1 | 0.01 | 15 | 70 |
| 2 | 0.03 | 15 | 85 |
| 3 | 0.08 | 20 | 94 |
| 4 | 0.1 | 20 | 94 |
| 5 | 0.3 | 40 | 95 |
| 6 | Free | 200 | - |
aYields refer to the isolated pure products.
Furthermore, the reaction was carried out in different solvents and under solvent-free conditions. As shown in Table 2, the yields of the reaction under solvent-free conditions were greater and the reaction times were generally shorter than the conventional methods. The best result was obtained under reflux conditions for 20 min and in the presence of water as green solvent. Increasing the reaction time did not improve the yield in Table 2.
Table 2. Effect of the reaction time on the synthesis of 3,3,6,6-tetramethyl-9-phenyl-3,4,5,6,7,9-hexahydro-1H-xanthene-1,8(2H)-dione using ZnO nanoparticles catalyst under reflux conditions and water as green solvent.
| Entry | Solvent | Temperature (°C) | Time (min) | aYield (%) |
| 1 | H2O | Reflux | 20 | 94 |
| 2 | Ethyl acetate | Reflux | 47.5 | 74 |
| 3 | Acetonitrile | Reflux | 48 | 71 |
| 4 | Methanol | Reflux | 44 | 88 |
| 5 | Ethanol | Reflux | 42 | 90 |
| 6 | DMF | Reflux | 95 | 62 |
| 7 | DMSO | Reflux | 105 | 63.5 |
| 8 | CHCl3 | Reflux | 91 | 65 |
| 9 | CH2Cl2 | Reflux | 92.5 | 60 |
| 10 | CCl4 | Reflux | 155 | 59.5 |
| 11 | Free | Reflux | 300 | - |
aYields refer to the isolated pure products.
Also, the effect of various solvents on the rate of the reaction was studied (Table 2). We also examined the effect of solvents, as can be seen, ethanol and water were favorable solvents for this synthesis and whereas much lower yields were observed with other solvents, including ethyl acetate and methanol. But water was chosen, because it is acceptable solvent for green chemistry and environment.
Table 3. Effect of various solvents on the synthesis of 3,3,6,6-tetramethyl-9-phenyl-3,4,5,6,7,9-hexahydro-1H-xanthene-1,8(2H)-dione using ZnO nanoparticles catalyst.
| Entry | Time (min) | aYield (%) |
| 1 | 20 | 94 |
| 2 | 25 | 94 |
| 3 | 35 | 94 |
| 4 | 55 | 94.5 |
| 5 | 65 | 94.5 |
| 6 | 85 | 94.5 |
| 7 | 100 | 94.5 |
| 8 | 120 | 95 |
| 9 | 240 | 95 |
| 10 | 300 | 95 |
aYields refer to the isolated pure products.
By using this nanocatalyst, we observed that a wide range of aldehydes having both electron-donationg and electron-withdrawing groups are equally facile for the reaction, resulting in the formation of xanthenes derivatives in very good yields; although, electron-withdrawing groups were slightly better.
We also, herein report a new, convenient, mild, and efficient procedure for the synthesis of 14-aryl-14H-dibenzo[a,j]xanthene derivatives catalyzed by ZnO nanoparticles as an effective and novel nanocatalyst requiring only mild reaction conditions (Scheme 2).
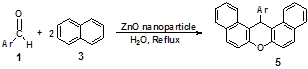
Figure 2. 14-aryl-14H-dibenzo[a,j]xanthene in the presence of ZnO nanoparticles catalyst under reflux conditions.
Using these optimized reaction conditions, the scope and efficiency of this approach was explored for the synthesis of a wide variety of substituted 14-aryl-14H-dibenzo[a,j]xanthenes and the results are summarized in (Table 4, entries 10, 24, 25). The reactions of variety of structurally diverse aldehydes with β-naphthol were investigated using the optimized reaction conditions (Scheme 2). It was found that all the reactions proceeded to give the corresponding benzoxanthenes (Table 4, entries 10, 24 and 25) in high yields. This protocol tolerates aromatic aldehydes containing both electron-donating and electron-withdrawing substituents well. The electronic effects and the nature of the substituents on the aldehydes showed some clear trends in terms of yields and reaction times. Aromatic aldehyde substrates with electron-withdrawing groups reacted well at faster reaction rates than aromatic aldehyde substrates with electron-donating groups. Ortho-substituted aromatic aldehydes, however, did not react as smoothly, likely because of steric hindrance, and longer reaction times were required to get the corresponding products in high yields (Table 4, entries 24, 25).
Table 4. One-pot synthesis of 1,8-dioxooctahydroxanthenes using ZnO nanoparticles as catalyst.
| Entry | Product | Time (min) | aYield(%) |
| 1 |
| 20 | 94 |
| 2 |
| 170 | 91 |
| 3 |
| 143 | 95 |
| 4 |
| 145 | 91 |
| 5 |
| 150 | 92 |
| 6 |
| 150 | 97 |
| 7 |
| 100 | 93 |
| 8 |
| 125 | 95 |
| 9 |
| 126 | 93 |
| 10 |
| 145 | 96.5 |
| 11 |
| 150 | 95 |
| 12 |
| 214 | 87 |
| 13 |
| 128 | 96 |
| 14 |
| 133 | 95.5 |
| 15 |
| 170 | 92 |
| 16 |
| 184 | 89.5 |
| 17 |
| 153 | 92.5 |
| 18 |
| 88 | 92 |
| 19 |
| 128 | 88 |
| 20 |
| 134 | 84.5 |
| 21 |
| 159 | 90 |
| 22 |
| 131 | 89.5 |
| 23 |
| 129 | 91.5 |
| 24 |
| 210 | 99 |
| 25 |
| 236 | 96.5 |
aYields refer to the isolated pure products.

Figure 3. Plausible mechanism for the ZnO nanoparticles catalysed synthesis of 1,8-dioxooctahydroxanthene derivatives and its open chain form.
A plausible mechanism for the synthesis of various 9-aryl-1,8-dioxooctahydroxanthane derivatives (4a-z) catalysed by ZnO nanoparticle is proposed (Scheme 3). Reaction of dimedone in its enol form is expected to react with aromatic aldehyde, under the influence of ZnO nanoparticle, to give rise to ortho-quinone methides (o-QMs) III by Knoevenagel adduct formation. The intermediate III may further undergo Michael addition with another molecule of dimedone to form intermediate IV. The presence of water as solvent might not have facilitated dehydration reaction to give the cyclised product 4. However, when reaction was carried out in the absence of water a facile dehydration reaction took place to give rise to the cyclic compound 4.
This nano catalyst also showed excellent reusability in these reactions (Table 5) and the catalyst could be recycled five times for the model reaction without significant loss of activity (Table 5). No observation of any appreciable loss in the catalytic activity of ZnO nanoparticles catalyst was observed (Table 4, entry 1).
Table 5. Recyclability of ZnO nanoparticles catalyst in the synthesis of 3,3,6,6-tetramethyl-9-phenyl-3,4,5,6,7,9-hexahydro-1H-xanthene-1,8(2H)-dione (Table 4, entry 1) under reflux conditions and water as green solvent for 20 min.
| Entry | Cycles | aYield(%) |
| 1 | 1 | 94 |
| 2 | 2 | 93 |
| 3 | 3 | 93 |
| 4 | 4 | 92 |
| 5 | 5 | 91.5 |
aIsolated yield.
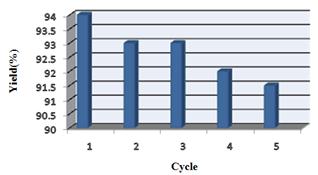
Figure 4. Recyclability of ZnO nanoparticles catalyst in the condensation reaction of 5,5-dimethyl-1,3-cyclohexanedione and benzaldehyde.
The possibility of recycling the catalyst was examined using the condensation reaction of 5,5-dimethyl-1,3-cyclohexanedione and benzaldehyde under the optimized conditions. When the reaction was completed, the mixture was filtered and the remaining was washed with warm ethanol, and the catalyst reused in the next reaction. The recycled catalyst could be reused four times without any additional treatment. After the completion of the reaction, the reaction mixture was cooled to room temperature, and hot ethanol was added. The catalyst was separated by simple filtration, dried at 60 °C under vacuum for 1 h, and reused for a similar reaction. As shown in Figure 1, the catalyst could be reused at least five times without significant loss of activity.
4. Conclusion
In conclusion, we have developed a simple and method for the synthesis of various 1,8‐dioxooctahydro xanthene derivatives by using the reaction of 1,3‐cyclohexanedione with aromatic aldehydes under environmentally benign conditions in the presence of ZnO nanoparticles catalyst. These nano catalysts mediated aqueous phase organic reactions are useful both from economical and environmental points of view. This methodology also overcomes the formation of unwanted by‐products, low yields, and use of hazardous solvents and high temperatures. A simple and highly efficient method for the synthesis of 14-substituted-14H-dibenzo[a,j]xanthenes has been developed via the condensation of β-naphthol with aldehydes catalyzed by ZnO nanoparticles catalyst in water at reflux conditions. The catalyst was recovered and reused without any noticeable loss of reactivity. The mild reaction conditions and simplicity of the procedure offers improvements over many existing methods. The present approach offers the advantages of simple methodology, clean and mild reaction conditions, high atom-economy, short reaction time, low environmental impact, wide substrate scope, high yield, and excellent product purity.
References
- Hideo, T. Jpn Tokkyo Koho JP 56005480,1981.
- Poupelin, J. P.;Saint‐Ruf, G.;Foussard‐Blanpin, O.;Narcisse, G.;Uchida Ernouf, G.;Lacroix, R.Eur. J. Med. Chem.1978, 13, 67.
- Lambert, R. W.;Martin, J. A.;Merrett, J. H.;Parkes, K. E. B.;Thomas, G. J. PCT Int. Appl.WO9706178, 1997.
- Ion, R. M.;Planner, A.;Wiktorowicz, K.;Frackowiak, D.Acta Biochim. Pol. 1998,45, 833.
- Hatakeyma, S.;Ochi, N.;Numata, H.;Takano, S.J. Chem. Soc.Chem. Commun.1988, 1202.
- Hilderbrand, S. A.;Weissleder, R.Tetrahedron Lett.2007,48, 4383.
- Menchen, S. M.;Benson, S. C.;Lam, J. Y. L.;Zhen, W.;Sun, D.;Rosenblum, B. B.;Kha, S. H.;Taing, M.US Patent, 2003,6,583, 168.
- Banerjee, A.; Mukherjee, A. K.Stain Technol.1981,56, 83.
- Reynolds, G. A.; Tuccio, S. A.; Peterson, O. G.; Specht, D. P.German Patent, 1971, DE2109040.
- Horning, E. C.;Horning, M. G.J. Org. Chem.1964,11, 95.
- Wang, J. Q.;Harvey, R. G.Tetrahedron.2002,58, 5927.
- Casiraghi, G.;Casnati, G.;Cornia, M.Tetrahedron Lett. 1973,14, 679.
- Casiraghi, G.;Casnati, G.;Catellani, M.;Cornia, M.Synthesis.1974, 564.
- Takeshiba, H. JP 56 005 480, 1981.
- Poupelin J. P.;Saint-Rut, G.;Foussard-Blanpin, O.;Narcisse, G.;Uchida-Ernouf, G.;Lacroix, R. Eur J Med Chem. 1978, 13, 67.
- Lambert, R. W.;Martin, J. A.;Merrett, J. H.;Parkes, K. E. B.;Thomas, G. J. Chem Abstr. 1997, 126, 212377y.
- Ion, R. M.;Frackowiak, D.;Planner, A.;Wiktorowicz, K. Acta Biochim Pol. 1998, 45, 833.
- Saint-Ruf, G.;De, A.;Hieu, H. T. Bull Chim Ther. 1972, 7, 83.
- Saint-Ruf, G.;Hieu, H. T.;Poupelin, J. P. Naturwissenschaften. 1975, 62, 584.
- Banerjee, A.;Mukherjee, A. K. Stain Technol. 1981, 56, 83.
- Menchen, S. M.;Benson, S. C.;Lam, J. Y. L.; Zhen, W.;Sun, D.;Rosenblum, B. B.;Khan, S. H.;Taing, M. U. S. US 6 583 168, 2003.
- Sirkecioglu, O.;Talinli, N.;Akar, A. J Chem Res, Synop. 1995, 502.
- Bekaert, A.;Andrieux, J.;Plat, M. Tetrahedron Lett. 1992, 33, 2805.
- Knight, C. G.;Stephens, T. Biochem J. 1989, 258, 683.
- Casiraghi, G.;Casnati, G.;Cornia, M. Tetrahedron Lett. 1973, 14, 679.
- Knight, D. W.;Little, P. B. J Chem Soc, Perkin Trans 1. 2001,1771.
- Sen, R. N.;Sarkar, N. N. J Am Chem Soc. 1925, 47, 1079.
- Papini, P.; Cimmarusti, R. Gazz Chim Ital. 1947, 77, 142.
- Ota, K.;Kito, T. Bull Chem Soc Jpn. 1967, 49, 1167.
- Rajitha, B.;Kumar, B. S.;Reddy, Y. T.;Reddy, P. N.;Sreenivasulu, N. Tetrahedron Lett. 2005, 46, 8691.
- Khosropour, A. R.;Khodaei, M. M.;Moghannian, H. Synlett. 2005, 955.
- Hatakeyma, S.;Ochi, N.;Numata, H.;Takano, S.;J Chem Soc, Chem Commun.1988, 1202.
- Hafez, E. E.;Elnagdi, M. H.;Elagamey, A. G. A.;El-Taweel, F. M. A. A. Heterocycles. 1987, 26, 903.
- Abdel Galil, F. M.; Riad, B. Y.;Sherif, S. M.;Elnagdi, M. H. Chem Lett. 1982, 1123.
- Ellis, G. P. The chemistry of heterocyclic compounds. In Chromene, Chromanes and Chromone. Volume 2. Edited by: Weissberger A, Taylor ECE. John Wiley: New York. 1977, 13.
- Callan, J. F.; De Silva, P.;Magri, D. C. Tetrahedron.2005, 61, 8551.
- Sirkecioglu, O.;Talinli, N.;Akar, A. J Chem Res (S). 1995, 502.
- Poupelin, J. P.;Saint-Rut, G.;Foussard-Blanpin, O.;Eu J Med Chem.1978, 13, 67.
- Banerjee, A.;Mukherjee, A. K. Stain Technol. 1981, 56, 83.
- Jin, T. S.;Zang, J. S.;Wang, A. Q.;Li, T. S. Synth Commun. 2005, 35, 2339.
- Shi, D. Q.;Wang, Y. H.;Lu, Z. S.;Dai, G. Y. Synth Commun. 2000, 30, 713.
- Jin, T. S.;Zang, J. S.;Xiao, J. C.;Wang, A. Q.;Li, T. S. Synlett. 2004, 866.
- Das, B.;Thirupathi, P.;Reddy, K. R.;Ravikanyh, B.;Nagarapu, L. Catal Commun. 2007, 8, 535.
- S. Ghomi, J.;Ghasemzadeh, M. A A.;Zahedi S, J. Mex. Chem. Soc. 2013, 57, 1.
- Horning E C and Horning M G, J Org Chem. 1946, 11, 95.
- Wang, X. S.;Shi, D. Q.;Li, Y. L.;Chen, H.;Wei, X. Y.;Zong, Z. M, Synth Commun. 2005, 35, 97.
- Jin, T. S.;Zhang, J. S.;Xiao, J. C.;Wang, A. Q.;Li, T. S, Synlett. 2004, 866.
- Darvish, F.;Balalaei, S.;Chadegani, F.;Salehi, P, Synth Commun. 2007, 37, 1059.
- Jin, T. S.;Zhang, J. S.;Wang, A. Q.;Li, T. S, Ultrason Sonochem. 2006, 13, 220.
- Dabiri, M.;Baghbanzadeh, M.;Arzroomchilar, E, Catal Commun. 2008, 9, 939.
- Das, B.;Thirupathi, P.;Mahender, I.;Reddy, V. S.;Rao, Y. K, J Mol Catal A Chem. 2006, 247, 233.
- Das, B.;Thirupathi, P.;Mahender, I.;Reddy, K. R.;Ravikanth, B.;Nagarapu, L, Catal Commun. 2007, 8, 535.
- Srihari, P.;Mandal, S. S.;Reddy, J. S. S.;Srinivasa, Rao, R.;Yadav, J. S, Chin Chem Lett. 2008, 19, 771.
- Rostamizadeh, S.;Amani, A. M.;Mahdavinia, G. H.;Amiri, G.;Sepehrian, H, Ultrason Sonochem.2010, 17, 306.
- Mahdavi, G. H.;Bigdeli, M. A.;Saeidi, H. Y,Chin Chem Lett. 2009, 20, 539.
- Imani Shakibaei, G.;Mirzaei, P.;Bazgir, A, Appl Catal A: General. 2007, 325, 188.
- Kantevari, S.;Bantu, R.;Nagarapu, L, J Mol Catal A: Chem. 2007, 269, 53.
- Maghsoodlou, M .T.;Habibi Khorassani, S. M.;Shahkarami, Z.;Maleki, N.;Rostamizadeh, M, Chin Chem Lett. 2010, 21, 686.
- Karthikeyan, G.;Pandurangan, A, J Mol Catal A: Chem. 2009, 311, 36.
- Khosropour, A. R.;Khodaei, M. M.;Moghannian, H. A. Synlett. 2005, 955.
- Mahdavi, G. H.;Bigdeli, M. A.;Saeidi Hayeniaz, Y. Chin Chem Lett. 2009, 20, 539.
- Imani Shakibaei, G.;Mirzaei, P.;Bazgir, A. Appl Catal A: General.2007, 325, 188.
- Lu, C. H.;Yeh, C. H, Ceram. Int. 2000, 26, 351.
- Shah, M.A.;African Phys. Rev. 2008, 2, 106.
- Gharib, A.;Noroozi Pesyan, N.;Vojdani Fard, L.; Roshani, M, J Chem Eng and Chemistry Research. 2014, 1, 1.

 4a
4a 4b
4b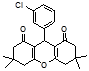 4c
4c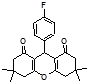 4d
4d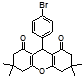 4e
4e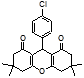 4f
4f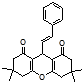 4g
4g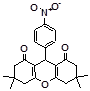 4h
4h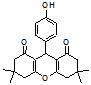 4i
4i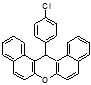 4j
4j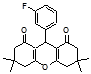 4k
4k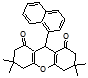 4l
4l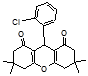 4m
4m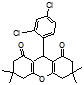 4n
4n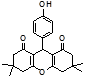 4o
4o 4p
4p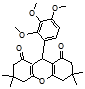 4q
4q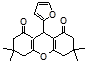 4r
4r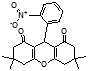 4s
4s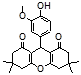 4t
4t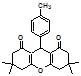 4u
4u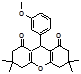 4w
4w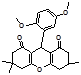 4x
4x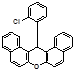 4y
4y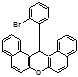 4z
4z

