A Catalytic Synthesis of Spiro[dibenzo[b,i]xanthene-13,30-Indoline]-Pentaones Using Heterogeneous Strong Basic Copper(II) Oxide Nanoparticles Catalyst Under Green Conditions
Ali Gharib1, 2, *, Leila Vojdani Fard3, Nader Noroozi Pesyan4, Mina Roshani1
1Department of Chemistry, Islamic Azad University, Mashhad, Iran
2Department of Chemistry, Agricultural Researches and Services Center, Mashhad, Iran
3Education Ministry, Education Organization of Razavi Khorasan, Mashhad, Iran
4Department of Chemistry, Faculty of Science, Urmia University, Urmia, Iran
Abstract
The protocol offers advantages in terms of higher yields, and mild reaction conditions, with reusability of the catalyst. Green chemistry protocols with the reusability of the nanoparticle as catalyst and water as a green solvent in the synthesis of spiro[dibenzo[b,i]xanthene-13,30-indoline]-pentaones is described. The copper(II) oxide nanoparticles functions as highly effective catalyst for the reactions of 2-hydroxynaphthalene-1,4-dione (1) and isatins (2a-d) and reflux conditions to afford the corresponding spiros in moderate to good yields. The catalyst is inexpensive, stable, can be easily recycled and reused for several cycles with consistent activity.
Keywords
Nanoparticles, Copper(II) Oxide, Catalyst, Isatin, Spiro, Heterogeneous
Received: March 21, 2015
Accepted: April 6, 2015
Published online: April 19, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Indole and indoline fragments are important moieties in a large number of natural biologically active compounds [1], and some indolines that are spiro-annulated with heterocycles at the 3 position have shown high biological activity [2,3]. The spirooxindole system is the core structure of many pharmacological agents and natural alkaloids [4,5]. Therefore, a number of methods have been reported for the preparation of spirooxindoles [6].
Molecules with a naphthoquinone structure, due to their biological properties, constitute one of the most interesting classes of compounds in organic chemistry. They have also industrial applications and can potentially be used as intermediates in the synthesis of heterocycles [7]. A series of related naphthoquinone pigments (streptocarpone, adunnione, dunniol, and dunnione) from Streptocapus dunnii have been isolated and characterized [8]. On the other hand, pyrazolo[3,4-b]quinolines belong to a class of highly fluorescent compounds which emit mostly in the blue spectral range and have been classified as promising materials for optoelectronics [9]. Attempts have been made to produce redshifts in their fluorescence spectra by changing substituents [10]. The quinone moiety is involved in a wide variety of biochemical processes including electron transport and oxidative phosphorylation [11]. Various biological properties including enzyme inhibition, antibacterial, antifungal, and anticancer activities have been reported for quinones and quinone derivatives [12]. The antitumor activity of the quinone moiety has been studied thoroughly, and it is known that they act as topoisomerase inhibitors via DNA intercalation [13] . Quinone-annulated heterocycles are found in nature, and most of them exhibit interesting biological activities. The chemistry of quinone-annulated heterocycles is dependent largely on the substituent being either on the quinone or on adjacent rings [14]. These activities, combined with diverse chemical behavior make quinones attractive targets in organic synthesis. Solid base catalysts have several advantages over homogeneous organic basic catalysts, such as easy recovery of the catalyst, simple product isolation, and recyclability. Thus, the heterogeneous solid base catalysts have been recognized as potential alternatives to homogeneous organic basic catalysts [15]. Nanocrystalline metal oxides find excellent applications as active adsorbents for gases and destruction of hazardous chemicals [16]. They are also gaining tremendous importance due to their distinct catalytic activities for various organic transformations. Production of nanoparticles can be carried out by chemical and physical methods. Super Critical Water Hydrothermal Synthesis (SCWHS) is among the most active fields of green chemistry. It is a relatively simple and environmentally benign process.
2. Experimental Section
2.1. Chemicals and Apparatus
Melting points were determined by Thieles tube method (Table 1) and were uncorrected. 1H-NMR spectra were recorded on a Bruker AM 300 MHz and 13C NMR (Brucker Gemini 100 MHz) spectrometer using CDCl3 as a solvent and tetramethylsilane as an internal standard. The chemical shifts are expressed in δ (ppm). FT IR spectrometer of Perkin Elmer was used for study. Thin layer chromatography (TLC) was done with pre-coated silica gel plates (GF254 Merck) using benzene:ethyl acetate (9.5:0.5, v/v) as the mobile phase.
2.2. Preparation of the Ordinary Copper Oxide
In order to study physicochemical properties of the obtained CuO nanoparticles, bulk of CuO powder (non-nanometric) as comparison reference was prepared by a two step calcination procedure. At first, crystalline copper nitrate trihydrate was charged into a porcelain crucible, and gently heated on a hot plate until most of the hydration water was removed. Then, the crucible was transferred into a muffle furnace and heated up to 400 oC for 22 h and at the end, produced CuO product was gently crushed and screened.
2.3. Preparation of the CuO Nanoparticles Catalyst
Copper(II) nitrate trihydrate (Merck A G. For synthesis) was used as the precursor for synthesis of nano copper oxide. Preparation of CuO took place in a stainless steel (316 L) autoclave that was able to endure working temperature and pressure of 550 oC and 610 atm, respectively. Concentration of Cu(NO3)2 was 0.05 mol dm-3, and heating period about 2 h. Synthesis was carried out at 500 oC to accelerate the hydrolysis reactions and thus shorten the fabrication period. In order to maintain the safety margin, the 200 cm3 stainless steel autoclave was loaded by only 60-80 cm3 of the solution. After removing from furnace, the autoclave was quenched by cold water and CuO nano particles were recovered from discharged solution by high speed centrifugation at 14,000 rpm for about 60 min. The produced nanoparticles were then three times washed in the same centrifuge with ultra pure water, and then dried at ambient temperature. Nanoparticles were characterized by XRD (Philips PW 1800), and TEM (LEO 912AB) tests.
2.4. Procedure for the Synthesis of Spiro[dibenzo[b,i]xanthene-13,3-indoline]-Pentaones (3a-d)
A mixture of 2-hydroxynaphthalene-1,4-dione (4 mmol), an isatin (2 mmol) and copper(II) oxide (0.5 g) in refluxing water (25 mL) was stirred for 20 h (the progress of the reaction was monitored by TLC). After completion, the reaction mixture was filtered and the obtained precipitate washed with water and recrystallized from EtOH/H2O (1:3) to afford pure production.
2.5. Selected Spectral Data
1'-methyl-1'H-spiro[dibenzo[b,i]xanthene-13,4'-quinoline]-3',5,7,12,14(2'H)-pentaone (3a)
1H NMR (300 MHz, CDCl3): δH 3.35 (3H, s, CH3), 6.85-8.00 (12H, m, H-Ar). 13C NMR (100 MHz, CDCl3): δc 206, 183, 178.2, 160.7, 147.5, 135, 131.2, 130, 128., 126.7, 120.5, 118.8, 110.3, 67.4, 45.5, 37.9. IR (KBr, cm−1): ν 3065, 1710, 1676, 1600. Anal. Calcd for C29H15NO6: C, 73.57; H, 3.19; N, 2.96. Found: C, 73.53; H, 3.16; N, 2.91. HRMS (EI) Calcd. for C29H15NO6 [M]+, 487.1003, Found 487.1007.
6'-bromo-1'-methyl-1'H-spiro[dibenzo[b,i]xanthene-13,4'-quinoline]-3',5,7,12,14(2'H)-pentaone (3b)
1H NMR (300 MHz, CDCl3): δH 3.33 (3H, s, CH3), 7.01-8.12 (11H, m, H-Ar). 13C NMR (100 MHz, CDCl3): δc 206, 183, 178.3, 160.9, 146.7, 135, 134.8, 131.9, 130.9, 126.7, 121.2, 120.6, 116.7, 112.8, 67.6, 45, 37.6. IR (KBr, cm−1): ν 3017, 1714, 1603. Anal. Calcd for C29H14BrNO6: C, 63.06; H, 2.55; N, 2.54. Found: C, 63.02; H, 2.58; N, 2.59. HRMS (EI) Calcd. for C29H14BrNO6 [M]+, 565.0004, Found 565.0006.
1'H-spiro[dibenzo[b,i]xanthene-13,4'-quinoline]-3',5,7,12,14(2'H)-pentaone (3c)
1H NMR (300 MHz, CDCl3): δH 6.75-8.14 (12H, m, H-Ar), 10.85 (1H, s, NH). 13C NMR (100 MHz, CDCl3): δc 206, 183, 178.3, 160.9, 146.7, 135, 131.7, 130.9, 129.8, 126.8, 120.6, 118.3, 117.2, 110.7, 60.5, 45.6. IR (KBr, cm−1): ν 3410, 3090, 1720, 1668, 1603. Anal. Calcd for C28H13NO6: C, 73.21; H, 2.85; N, 3.05. Found: C, 73.26; H, 2.81; N, 3.11. HRMS (EI) Calcd. for C28H13NO6 [M]+, 473.1000, Found 473.1005.
1'-benzyl-1'H-spiro[dibenzo[b,i]xanthene-13,4'-quinoline]-3',5,7,12,14(2'H)-pentaone (3d)
1H NMR (300 MHz, CDCl3): δH 5.31 (2H, J = 15.2 Hz, CH2), 6.76-8.30 (17H, m, H-Ar). 13C NMR (100 MHz, CDCl3): δc 206, 183, 178.3, 160.9, 147.5, 138.8, 131.9, 130.8, 128.5, 127.7, 127.0, 126.5, 118.8, 110.2, 64.7, 45.8. IR (KBr, cm−1): ν 3015, 1720, 1673, 1612.Anal. Calcd for C35H19NO6: C, 76.50; H, 3.48; N, 2.55. Found: C, 76.46; H, 3.41; N, 2.60. HRMS (EI) Calcd. for C35H19NO6 [M]+, 563.1009, Found 563.1014.
3. Results and Discussions
We wish to report an efficient and green protocol for reaction of 2-hydroxynaphthalene-1,4-dione (1) and isatins (2a-d) in the presence of copper(II) oxide nanoparticles catalyst as an inexpensive and readily available catalyst proceeded smoothly in water at reflux for 22 h to produce spiro[dibenzo[b,i]-xanthene-13,30-indoline]-20,5,7,12,14-pentaones (3a-d) in 78.5-86.5% yields (Scheme 1).
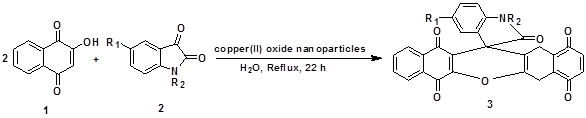
Scheme 1. Synthesis of spiro[dibenzo[b,i]-xanthene-13,30-indoline]-20,5,7,12,14-pentaones (3a-d) in presence of copper(II) oxide nanoparticles
catalyst and water under reflux conditions.
Table 1. Synthesis of spiro[dibenzo[b,i]-xanthene-13,30-indoline]-20,5,7,12,14-pentaones (3a-d) in presence of copper(II) oxide nanoparticles
catalyst and water under reflux conditions for 22 h.
| Entry | R1 | R2 | aYield (%) | Mp(oC) | Product |
| 1 | H | CH3 | 78.5 (78, 78, 77, 77)b | 360oC dec. | 3a |
| 2 | Br | CH3 | 80 (80, 80, 79,79)b | 365oC dec. | 3b |
| 3 | H | H | 84 (83, 83, 82.5, 82.5)b | 350 oC dec. | 3c |
| 4 | H | C6H5CH2 | 86.5 (86, 86, 85.5, 85)b | 358oC dec. | 3d |
aIsolated yield.bYield of catalyst recycled four times. dec= decomposition
As shown in Table 1, the yields of the reaction of 2-hydroxynaphthalene-1,4-dione (1) and isatins (2a-d) in the presence of copper(II) oxide nanoparticles catalyst. We examined Without copper(II) oxide nanoparticles catalyst, for long periods of time (66 h), the yields of products were low (<20%).
It is thought that compounds (3) result from initial addition of 2-hydroxynaphthalene-1,4-dione (1) to the isatin (2a-d) to yield intermediates (4), which react further with another molecule of compound (1). Finally, cyclization afforded the corresponding products (3a-d) (Scheme 2).
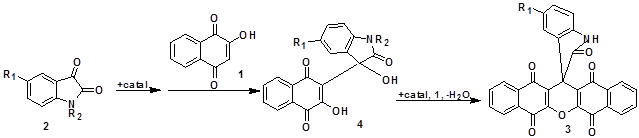
Scheme 2. Steps of addition of 2-hydroxynaphthalene-1,4-dione (1) to the isatin (2a-d) to intermediates (4) and products (3a-d).
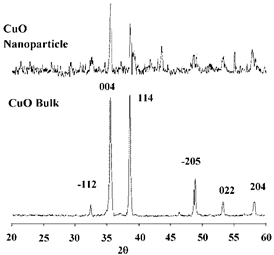
Figure 1. XRD spectrum of CuO prepared by calcination of Cu(NO3)2(H2O)3.
The XRD spectrum of copper nitrate trihydrate nano-sized is shown in Figure 1. All diffraction peaks of X-ray are indexed to the monoclinic crystal system of CuO.
No characteristic peaks are observed for other possible impurities, such as Cu(OH)2,Cu2O or Cu(OH)3NO3. Average size of the obtained CuO particle shown in Figure 2 is 5 nm. The crystallite size was also calculated by X-ray line broadening analysis using the Scherrer equation, we found that the average CuO crystallite size was 8 nm.

Figure 2. Transmission electron micrographs (TEM) of CuO nanoparticles.
The mean value of surface area of CuO catalyst was 32.457 m2/g from BET analysis. Interpretation of this phenomena is that When water is heated up to its critical point (Tc = 374 oC, Pc = 22.1 MPa), it changes from a polar liquid to a fluid with a low dielectric constant. Adschiri et al. [17] also pointed out that dielectric constant of water, the most significant factor which controls the solvent power of water declines at supercritical condition, hence the formed metal oxide can no longer stay in aqueous phase and must precipitate. As a result, at high temperature supersaturation for precipitation of metal oxide becomes greater which consequently leads to creation of more nucleation centers [18], and thus formation of smaller particles (i.e., nanoparticles). At the same time dissociation constant of water (Kw) remarkably increases, giving rise to correspondingly increased concentrations of H+ and OH-. From this point, the key rule is played by OH- ions whose enhanced concentration according to Adschiri et. al. [19] leads to a rigorous hydrolysis of the metal salts that is immediately followed by a dehydration step (Scheme 3).
![]()
Scheme 3. Hydrolysis of the metal salts.
3.1. Solvents Effects
Different organic solvents were examined for the reaction and we found that water was the solvent of choice (Table 2). Ethanol, methanol and CHCl3 proved to be almost as good as THF, with CH2Cl2 giving a slightly better yield than CCl4. When the reactions were conducted in water, the expected products were obtained in good yields and compared to organic solvents (Tables 1 and 2).
Table 2. Solvent effects in the synthesis of 1'-benzyl-1'H-spiro[dibenzo[b,i]xanthene-13,4'-quinoline]-3',5,7,12,14(2'H)-pentaone (Table 1, entry 4, product 3d) in the presence of copper(II) oxide nanoparticles catalyst under reflux conditions for 22 h.
| Entry | Solvent | aYield (%) | Time (h) |
| 1 | H2O | 86.5 | 22 |
| 2 | C2H5OH | 80 | 31.5 |
| 3 | CH3OH | 78 | 33.5 |
| 4 | CH3CN | 73 | 51 |
| 5 | CHCl3 | 75 | 53 |
| 6 | CH2Cl2 | 69 | 54 |
| 7 | CCl4 | 59 | 66 |
| 8 | DMF | 61 | 77 |
| 9 | DMSO | 60 | 77 |
| 10 | THF | 70 | 44.5 |
a Isolated yield.
3.2. Regeneration of Catalyst
The possibility of recycling the catalyst was examined. For this reason, the reaction of isatin with 2-hydroxynaphthalene-1,4-dione was studied in water in the presence of copper(II) oxide nanoparticles. When the reaction was complete, the mixture was filtered, the residue was washed with warm ethanol and recycled catalyst was reused in the next reaction. No appreciable loss of catalytic activity was observed after foure cycles (Figure 3).
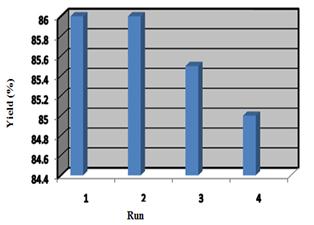
Figure 3. Recyclability of copper (II) oxide nanoparticles catalyst in the synthesis of 1'-benzyl-1'H-spiro[dibenzo[b,i]xanthene-13,4'-quinoline]-3',5,7,12,14(2'H)-pentaone (3d)
4. Conclusion
In conclusion, We have developed an efficient, facile and environmentally acceptable synthetic methodology for the synthesis of spiro[dibenzo[b,i]-xanthene-13,30-indoline]-20,5,7,12,14-pentaone derivatives using copper(II) oxide nanoparticles catalyst under green solvent (water as solvent) conditions. The copper(II) oxide nanoparticles was applied for the synthesis of spiro[dibenzo[b,i]-xanthene-13,30-indoline]-20,5,7,12,14-pentaones from isatin with 2-hydroxynaphthalene-1,4-dione in a simple and straight forward protocol. High yields, environmentally friendly conditions simplicity of operation, reusability, green solvent and easy work-up are some advantages of this method.
References
- R. J. Sundberg, "The chemistry of indoles," Academic, New York, 1996.
- Da Silva, J. F. M.;Garden, S. J.;Pinto, A. C. Journal Braz Chem Soc. 2001, 12, 273.
- Zhu, S. L.;Ji, S. J.;Yong, Z. Tetrahedron. 2007, 63, 9365.
- Ma, J.;Hecht, M.Chem Commun. 2004, 1190.
- Usui, T.;Kondoh, M.;Cui, C. B. Biochem J. 1998, 333, 543.
- Nandakumar, A.;Thirumurugan, P.;Perumal, P. T. Bioorg Med Chem Lett. 2010, 20, 4252.
- Thomson, R. H. "Naturally occurring quinines," 4th edn. Chapman & Hall, London, 1997.
- Perez, A. L.;Lamoureux, G.;Sanchez-Kopper, A. Tetrahedron Lett. 2007, 48, 3735.
- Tao, Y. T.;Balasubramaniam, E.;Danel, A. Journal Mater Chem, 2001, 11, 768.
- Gondek, E.;Kityk, I. V.;Danel, A. Mater Lett. 2006, 60, 3301.
- Pratt, Y. T.;Drake, N. L. Journal American Chemical Society. 1960, 82, 1155.
- Ryu, C. K.;Choi, K. U.;Shim, J. Y. Journal Bioorg Med Chem. 2003, 11, 4003.
- Denny, W. A.;Bagulet, B. C. "In Molecular Aspects of Anticancer Drug-DNA Interactions," 2nd ed.; Waring, M. J., Neidle, S., Eds.; Macmillan: London, 1994, pp. 270-311.
- Tisler, M. "Advances in Heterocyclic Chemistry. In Heterocyclic Quinones," A. R. Katriztky, Ed.; Academic: London, 1989, 45, 37.
- Seshu Babu, N.;Nayeem Pasha, K. T.;Venkateswara Rao, P. S. Tetrahedron Lett. 2008, 49, 2730.
- Klabunde, K. J.;Mulukutla, R."Nanoscale Materials in Chemistry," Wiley Interscience, New York, 2001.
- Adschiri, T.;Hakuta, Y.;Sue, K. Journal Nanopart Res. 2001, 3, 227.
- Adschiri, T.;Arai, K. "Supercritical Fluid Technology in Materials Science and Engineering," Marcel Dekker Inc., Tokyo, 2003.
- Adschiri, T.;Kanazawa, K.;Arai, K. Journal American Ceram Society. 1992, 75, 1019.



