Comparative Study of Fatty Acid Composition of Dennettia tripetala Leaves Extracted with Different Solvents
Nnenna E. Okoronkwo1, *, Michael O. Ajomiwe1, Chioma A. Ike-Amadi2
1Department of Pure and Industrial Chemistry, Abia State University, Uturu, Nigeria
2Department of Chemistry, Abia State Polytechnics, Aba, Nigeria
Abstract
The fatty acids contents of different solvent extracts of Dennettia tripetala leaves were characterized using GC/MS. Methanol, ethanol and benzene extracted eleven, ten and five fatty acid compounds respectively in varying amounts. The fatty acids extracted by the different solvents identified include: caprylic, capric, lauric, myristic, myristoleic, palmitic, palmitoleic, stearic, oleic, linoleic and linolenic acids. Benzene extracted the highest composition of linolenic acid (omega-3-fatty acid) with percentage composition of 97.76%, while methanol and ethanol extracted highest composition of palmitic acid with percentage compositions of 26.17% and 25.64% respectively. This study revealed that solvent used in extraction has great influence on the type and amount of fatty acids extracted.
Keywords
D. tripetala, Extraction, Fatty Acids, Leaves, Solvents
Received: March 20, 2015
Accepted: April 9, 2015
Published online: April 10, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Dennetia tripetala (annonanceae) is a well known plant in communities of some southern states of Nigeria. It is also found mostly in tropical rainforest region and sometimes in the Savannah area. Its common name is pepper fruit; locally called ‘mmịmị’ by Igbos and ‘nkarika’ by Efik of Calabar. The plant possesses characteristic aroma and fragrance. It ranges from small to medium size or shrub-like with alternate simple and estipulate leaves. The young leaves have spicy taste and are sometimes used to prepare ‘pepper soup’ delicacies and as condiments in some special local dishes [1]. The fruits are green at first and eventually red when ripened with spicy taste [2,3,4]. The leaves and the roots in addition to the fruits are utilized for `medicinal purposes [5]. It is also used by local herbalists to treat cough, vomiting and stomach upset and also for masking of mouth odour [6,7]. The chemical compositions of the leaves have been reported and important nutritive and non-nutritive components such as proximate, vitamin minerals and phytochemicals were found in the leave of the plant [8]. A phenanthrenic alkaloid has also been isolated and characterized from the fruit of the plant [9].
Moreover, plants serve as one of the sources of fatty acids and essential oils. Fatty acids are carboxylic acids with long hydrocarbon chains with a chain of even numbers of carbon from 4 to 28. They are one major group of lipids. Most frequently occurring fatty acids in nature are unbranched because they are synthesized from acetate, a compound of two carbons. Most naturally occurring fatty acids are usually derived from triglyceride or phospholipids. They are known as free fatty acids when they are not attached to other molecules [10]. Fatty acids are important source of fuel because their metabolism yields large quantities of ATP (Adenosin Triphosphate). The double bonds in naturally occurring unsaturated fatty acids have the cis configuration and are always separated by CH2 group. A cis configuration means that hydrogen atoms are on the same side of the double bond. Essential fatty acid is the type of fatty acids that are required by the human body but cannot be made in sufficient quantities and therefore must be obtained from food.
Solvent extraction is mostly useful technique for isolation of plant active compounds though the extract yields of the plant materials are highly dependent on the nature of extracting solvent, due to the presence of different active compounds present on the plant material to be extracted which have varied chemical characteristics and polarities that may or may not be soluble in a particular solvent [11]. Methanol and ethanol have been extensively used to extract active compounds from various plants parts including the leaves, stem, root and bark and even the fruits and fruit peels [11,12,13,14].
The extraction of compounds from natural product sources requires a proper selection of plant, extraction method and screening method for discovering bioactive molecules. Among the key steps in natural product processing is the selection of suitable solvent for extraction [15], since the components extracted is highly dependent on the extraction solvent used. Studies on the influence of extraction solvents from different plants materials have been reported [16,17].
This research, therefore, is aimed at using Gas Chromatography/Mass Spectrometer (GC/MS) to characterise the fatty acids compositions of D. tripetala (annonanceae) leaves using three different organic solvents for the extraction.
2. Materials and Methods
The sample used for this research was gotten from Umuekeogo Ogbor Ovuru in Abo Mbaise Local Government Area of Imo State, Nigeria and was identified by Mr. Ibe of Forestry Department, Michael Okpara University of Agriculture, Umudike, Abia State. The green leaves were detached from the stem, air dried and ground into powdered form. This was then stored in an air tight container until required for analysis.
The extraction of the crude sample of the leaves of the plant sample was done using three different solvents - methanol, ethanol and benzene. The cold percolation method of extraction was used. 15.0 g of ground D. tripetala leaves was put into conical flask to which 150 ml of methanol was added into it and then allowed to stand for 24 hours. It was then filtered and the residue was again re-soaked for another 24hrs with fresh 150 ml of methanol and filtered for total extraction of the plant material. The filtrate was then allowed to evaporate to dryness. This process was repeated for both the ethanol and benzene solvents. The extracts were then used to determine and characterise the fatty acid contents by subjecting them to GC/MS adapted basically for fatty acids characterisation (Hewllet placard 6890 series, England).
3. Results and Discussion
The chromatogram of GC/MS characterisation of fatty composition of methanol, ethanol and benzene extracts of Dennettia tripetala leaves showed sixteen (16), fifteen (15) and five (5) peaks suggesting same number of compounds respectively and the result is shown in Tables 1 and 2.
The study revealed that the solvents used extracted different fatty acids in varying amounts. Methanol extracted eleven different fatty acids which were identified as Caprylic, Capric, Lauric, Myristic, Myristoleic, Palmitic, Palmitoleic, Stearic, Oleic, Linoleic and Linolenic. Ethanol extracted ten fatty acids identified as Caprylic, Capric, Lauric, Myristic, Myristoleic, Palmitic, Stearic, Oleic, Linoleic and Linolenic. Ethanol while benzene extracted only five fatty acids identified as Myristic, Palmitic, Palmitoleic, Oleic and Linolenic.
The fatty acids identified from the different solvents (Table 2) are represented by structures 1 – 11 (Fig 1): caprylic (1), capric (2), lauric (3), myristic (4), myristol (5), palmitic (6), palmitoleic (7), stearic (8), oleic (9), linoeic (10) and linolenic (11) acids.
However, for the methanol extract, palmitic acid had the highest composition with a value of 26.1711% at the retention time of 8.247 mins while caprylic acid had the least percentage composition with a value of 0.484% at the retention time of 1.76 mins. The ethanol extract also showed similar trend while benzene extracted on major fatty acid which showed the highest composition and identified as linolenic acid at a retention time of 15.855 with a percentage composition value of 97.76%.
The variation in the retention times of identified fatty acids of the three different solvents were very minimal while there was great difference between the percentage compositions extracted by them. Though the retention time of the identified compound were approximately at same range, the percentage composition extracted for each of the solvents varied greatly. Furthermore, benzene extracted other fatty acids present in a very minimal percentage composition while that of methanol and ethanol were relatively similar but differences in palmitoleic acid and other unidentified compounds present in the crude extracts.
The results of the present study revealed that among all the solvents used in the extraction; methanol and ethanol extracted more of the fatty acids though their compositions varied and in much lower percentages. This may be due to the fact that they are often extracted in higher amounts in more polar solvents such as methanol/ethanol as compared to benzene [18,19,20].
The differences in the extract yields of the different solvent in the present analysis might be ascribed to the different availability of extractable components, resulting from the varied chemical composition of the fatty acids [21]. The amount of the extractable components that can be extracted from any plant material is mainly affected by the extraction procedure, which may in turn vary from sample to complex precedure. Other contributing factors includes: efficiency of the extracting solvent to dissolve bioactive components present the materials to be extracted [18,20].
The methanol and ethanol solvents also extracted more five other unidentified compounds (Table 1) and Table 2 shows the identified fatty acid and the IUPAC names.
Table 1. Fatty Acid Composition of D. tripetala leaves from different solvent.
| S/No | Fatty Acid composition | Methanol Extract | Ethanol Extract | Benzene Extract | |||
| Retention time (mins) | % composition | Retention Time (mins) | % Composition | Retention Time (mins) | % Composition | ||
| 1 | Caprylic | 1.768 | 0.48457 | 1.794 | 0.31639 | - | - |
| 2 | Capric | 2.509 | 1.97692 | 2.560 | 2.03306 | - | - |
| 3 | NI | 3.157 | 0.61039 | 3.238 | 0.56079 | - | - |
| 4 | Lauric | 4.129 | 5.84952 | 4.202 | 4.24876 | - | - |
| 5 | Myristic | 5.324 | 8.42352 | 5.398 | 7.63337 | 5.638 | 0.68536 |
| 6 | Myristoleic | 6.588 | 9.00358 | 6.729 | 7.14934 | - | - |
| 7 | NI | 7.761 | 0.91107 | 7.886 | 0.73886 | - | - |
| 8 | Palmitic | 8.247 | 26.17119 | 8.368 | 25.64715 | 8.598 | 0.47536 |
| 9 | Palmitoleic | 9.015 | 1.15090 | - | - | 9.264 | 0.33880 |
| 10 | NI | 9.492 | 0.51084 | - | - | - | - |
| 11 | Stearic | 10.879 | 6.00062 | 11.043 | 6.17610 | - | - |
| 12 | Oleic | 11.737 | 17.90309 | 11.979 | 18.70296 | 11.932 | 0.73567 |
| 13 | NI | 12.786 | 2.91463 | 12.979 | 2.70693 | - | - |
| 14 | Linoleic | 13.427 | 2.64410 | 13.665 | 3.32730 | - | - |
| 15 | NI | 14.257 | 2.11140 | 14.463 | 1.33090 | - | - |
| 16 | Linolenic | 15.197 | 13.33366 | 15.470 | 18.09214 | 15.855 | 97.76769 |
| 17 | NI | - | - | 19.372 | 1.33596 | - | |
Key: NI = Not Identified
Table 2. Fatty Acid Compounds Identified from D. tripetala leaves.
| S/No | Molecular weight | Molecular formula | No of double bonds | Trival Name | IUPAC Name |
| 1 | 144.21 | C8H16O2 | - | Caprylic | Octanoic acid |
| 2 | 172.26 | C10H20O2 | - | Capric | Decanoic acid |
| 3 | 200.32 | C12H24O2 | - | Lauric | Dodecanoic acid |
| 4 | 228.37 | C14H28O2 | - | Myristic | Tetradecanoic acid |
| 5 | 226.36 | C14H26O2 | 1 | Myristoleic | Tetradec-9-enoic acid |
| 6 | 256.42 | C16H32O2 | - | Palmitic | Hexadecanoic acid |
| 7 | 254.41 | C16H30O2 | 1 | Palmitoleic | Hexadec-9-enoic acid |
| 8 | 284.48 | C18H36O2 | - | Stearic | Octadecanoic acid |
| 9 | 282.46 | C18H34O2 | 1 | Oleic | Cis-9- Octadecenoic acid |
| 10 | 280.45 | C18H32O2 | 2 | Linoleic | Cis, cis-9,12- Octadecadienoic acid |
| 11 | 278.43 | C18H30O2 | 3 | Linolenic | Cis, cis-9,12,15- Octadecatrienoic acid |
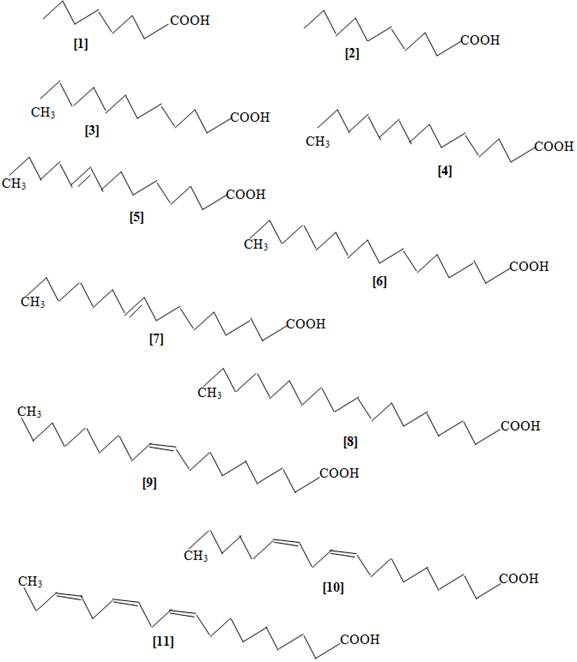
Fig. 1. Structures of the identified fatty acids from the leaves of D. tripetala.
Caprylic acid (octanoic acid) is used in commercial production of esters used in perfumery. It is also used in the treatment of some bacterial infections [22]. Capric acid, a saturated fatty acid is used in organic synthesis and industrially in the manufacture of perfumes and pharmaceuticals [23]. Lauric acid is a non-toxic fatty acid used used in the manufacture of flavourings, cocoa butter, margarine, soaps, shampoos and other surface active agents [24]. It possesses antibacterial, antioxidant and antiviral properties [25]. Myristic acid is used in cosmetics and medicinal preparations. Palmitic acid increases the risk of developing cardiovascular diseases. Palmitoleic acid helps to reduce weight gain which in turn combats obesity [26]. Stearic acid is useful as an ingredient in making dietary supplements and cosmetics. It is used to harden soaps particularly those made with vegetable oil [27]. Oleic acid is a monounsaturated omega-9 fatty acids found in various animal and vegetable fats. Linoleic acid (omega-6 fatty acids) is one of the essential fatty acids that human and animals ingest for good health [28]. It is used in the treatment of dermatitis and also in making of soap emulsifiers and quick drying oil. Linoleic acid has anti-inflammatory-acne reductive and moisture retentive properties when applied on the skin [22,29]. Linolenic acid is an unsaturated fatty acid considered essential to the diet [25] and an important component of drying oils [30]. Linoleic (omega-6 fatty acid) and linolenic acid (omea-3 fatty acid) are essential fatty acids for mammals. Mammals cannot synthesize them yet require them for normal body function and must be included in their diet [10].
4. Conclusion
D. tripetala leaves have been found from this study to contain essential fatty acid and the various fatty acids composition found from this study have their important uses. Also, the percentage composition of the various fatty acids varied with solvent used for their extraction of which benzene extracted the highest percentage composition of linolenic acid (omega-3-fatty acid).
References
- B. O. Ejechi and D. Akpomedaye ( 2005). Activity of Essential Oil and Phenolic Extracts of Pepper Fruits D, tripetala G. Baker against some Food Born Microorganisms. Afr. J. Biotechnol. pp 258 – 261.
- S. C. Achinewhu, C. C. Ogbonna and A. D. Hart (1995). Chemical Composition of indigenous wild Herbs Species Fruits. Nuts and Leafy Vegetables used as Food. Plants Food for Human Nutrition, Kluwer Publishers Netherland Vol. 48, No. 4: 341 - 388
- C. A. Stace (1980). Plant Taxanomy and Biosystematics. Edward Arnold Publication Ltd., London.
- P. Shukla and S. P. Misra (1979). An Introduction to the Taxonomy of Angiosoerm. Vikas Publishing House PVT Ltd.
- M. M. Iwu (1989). Food for Medicine, In Dietary Plants and Masticators as Source of Biological Active Substances. University of Ife Press. pp 303 – 310.
- I. A. Oyemitan, E. O. Iwalewa, M. A. Akanmu, S. O. Asa and T. A. Olugbade (2006). The Abusive Potential of Habitual Consumption of the Fruits of D. tripetala G. Baker (Annonaceae) among the People in Ondo Township (Nigeria). Nig. J. Natural Products Med. 2006. pp 55 – 62.
- I. A. Oyemitan (2006). Evaluation of D. tripetala G. Baker (Annonaceae) for Central Nervous System Activities. Ile Ife Nigeria. Department of Pharmacology, Obafemi Awolowo University. An M. Phil Thesis. pp 40 – 72.
- N. E. Okoronkwo, A. K. Mbachu and N. C. Nnaukwu (2011). Chemical Compositions and GC/MS Charactrisation of Fatty Acids Content of Dennettia Tripetala Leaves. In Nekang, Frida Menkan (ed.) Proceedings of International Conference on Sustainable Development, Vol.7. No. 10., Uyo: International Research and Development Institute [IRDI]/Kan Educational Books. pp 18 – 24.
- D. E. Okwu, F. N. Morah and E. M. Anam (2005). Isolation and characterization of Phenanthrenic Alkaloid Uvariopsine from Dennettia tetrapetala Fruit. J. Med. Aromatic Plant Science. pp 496 – 498.
- P. Y. Bruice (2007). Organic Chemistry 5th ed. Pearson Education International USA.
- W. Peschel, F. Sanchez-Rabaneda, W. Dn, A. Plescher, I. Gartzia, D. Jimenez, R. Lamuela-Raventos, S. Buxaderas, and C. Condina, (2006). An industrial approach in the search of natural antioxidants from vegetable and fruit wastes. Food Chem., 97, 137-150.
- M. H. Abdille, R. P. Singh, G. K.. Jayaprakasa, and B. S. Jens, (2005). Antioxidant activity of the extracts from Dillenia indica fruits. Food Chem., 90, 891-896.
- Z. U. Rehman, (2006). Citrus peel extract- A natural source of antioxidant. Food Chem., 99, 450-454.
- Y. Li, C. Guo, J. Yang, J. Wei, J. Xu, and S. Cheng, (2006). Evaluation of antioxidant properties of pomegranate peel extract in comparison with pomegranate pulp extract. Food Chem., 96,254-260.
- A. Taamalli, D. Arraez–Roman, E. Barrajon - Catalan, V. Ruiz - Torres, A. Perez - Sanchez, M. Herrero, E. Ibanez, V. Micol, M. Zarrouk, A. Segura-Carretero and A. Fernandez-Gutierrez (2012). Use of advanced techniques for the extraction of phenolic compounds from Tunisian olive leaves: phenolic composition and cytotoxicity against human breast cancer cells. Food Chem. Toxicol. 50:1817 – 1825.
- J. Alzeer, B. R. Vummidi, R. Arafeh, W. Rimawi, H. Saleem and N. W. Luedtke (2014). The influence of extraction solvents on the anticancer activities of Palestinian medicinal plants. Journal of Medicinal Plant Research. Vol. 8(8), pp. 408 – 415.
- B. Sultana, F. Anwar and M. Ashraf (2009). Effect of Extraction Solvent/Technique on the Antioxidant Activity of Selected Medicinal Plant. Extracts Molecules 14, 2167-2180.
- P. Siddhuraju, and K. Becker (2003). Antioxidant properties of various extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera lam.) leaves. J.Agric. Food Chem. 51, 2144-2155.
- F. Anwar, A. Jamil, S. Iqbal, and M. A. Sheikh (2006). Antioxidant activity of various plant extracts under ambient and accelerated storage of sunflower oil. Grasas Aceites Sevilla, 57, 189-197.
- B. Sultana, F. Anwar, and R. Przybylski (2007).. Antioxidant activity of phenolic components present inbarks of barks ofAzadirachta indica, Terminalia arjuna, Acacia nilotica, and Eugenia jambolana Lam. trees. Food Chem., 104, 1106-1114.
- B. Hsu, I. M. Coupar, and K. Ng (2006). Antioxidant activity of hot water extract from the fruit of the Doum palm, Hyphaene thebaica. Food Chem. 98, 317-328.
- J. Beare-Roger, A. Dieffebacher and J. V. Holm (2001). Lexicon of lipid nutrition IUPAC Publisher. pp 685 -744.
- M. John (2008) Organic Chemistry. 7th Ed. Thompson Brooks/Cole.
- T. Nakatsuji, M. C. Kao and C. M. Huangi (2009). Antimicrobial properties of Lauric Acid against Propionibacterium Acnes. Journal of Investigative Dermatology, Ching. pp 2480 – 2488.
- D. R. Lide (2005). CRC Handbook of Chemistry and Physics. 86th Ed. Boca Raton CRC Press.
- P. Nestel, P. Clifon and M. Noakes (1994). Effects of increasing dieting palmitoleic acid, palmitic acid and oleic acid on plasma lipid of hypercholesterolemic men. Journal of Lipid Research, Vol. 35. pp 656 – 662.
- E. A. Emken (1994). Metabolism of Dietary Stearic Acid Relatively to other Fatty Acids in Human Subjects. American Journal of Clinical Nutrition Vol. 60 (6:) 10235 – 10285.
- S. Cunnane and M. Anderson (1997). Pure linoleate deficiency in the ratinfluence on growth. Lipid resource publishers. pp 2216-2223
- M. Ayasse and R. Paxton (2002). Brood Production in Social Insects. Blackwell Berlin. pp 117 – 148.
- T. Alfred (2002). Fats and Fatty Oils. Ullmann’s Encylopedia of Industrial Chemistry. Weinheim.



