Inhibition of Peroxidase Activity by N-(3-Aminophenyl)-Arylsulfonamides Hydrochloride
Serkan Dayan1, Burcu Somturk1, Nilgun Kayaci1, Nalan Ozdemir1, Nilgun Kalaycioglu Ozpozan1, *, Hasan Ozdemir2
1Erciyes University, Faculty of Science, Department of Chemistry, Kayseri, Turkey
2Ataturk University, Faculty of Science, Department of Chemistry, Erzurum, Turkey
Abstract
A new series of [N-(3-aminophenyl)-arylsulfonamides] hydrochloride (Aryl= 4-methoxybenzene, 4-tert-butylbenzene, 4-nitrobenzene, 4-chlorobenzene) compounds (1-4) were synthesized successfully with a simple method. The compounds (1-4) were elucidated on the basis of elemental and spectral analyses (NMR, FT-IR, EA) and their inhibitory effects on the activity of purified peroxidase (POD) enzyme were evaluated by UV-vis spectrophotometers. The POD enzyme was purified from red cabbage (Brassica oleracea var. capitata f. rubra) using affinity chromatography. The half maximal inhibitory concentration (IC50) values were found to be 0.144 mM, 0.2 mM, 0.117 mM and 0.185 mM for compounds 1, 2, 3 and 4 respectively. The inhibition results show that all the compounds (1-4) inhibited POD enzyme activity. Particular, the N-(3-aminophenyl)-4-nitrobenzenesulfonamide hydrochloride compound (3) was found as the most active compound for POD compared to the others.
Keywords
Sulfonamide, Peroxidase, Inhibition, Synthesis
Received: March 16, 2015
Accepted: April 9, 2015
Published online: April 19, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
The structures containing sulfonamide fragment are generally obtained from R-sulfonyl chloride with primary or secondary amines in basic media [1]. Recently, there has been an increase in studies regarding compounds bearing sulfonamide moiety because of their potential applications such as in the metal complexes of sulfonamide [2], medicinal chemistry [3], as catalysts [4], in chemical luminescence [5] and in material chemistry [6]. Additionally, sulfonamide derivatives are known to contain inherent pharmacological properties such as antimicrobial [7], anti-inflammatory [8], antiviral [9], antiproliferative [10], angiogenesis [11] and enzyme inhibitory (α, β, δ-carbonic anhydrase, human carbonic anhydrase, adenine nucleotide translocator etc.) activities [12-16].
Peroxidases (PODs, E.C 1.11.1.X) are a member of the oxidoreductase family and they catalyse the hydrogen peroxide-dependent oxidation reaction of various substrates containing aromatic groups [17-19]. They are widely found in different kinds of organisms and are associated with numerous physiological and biochemical functions in living organisms such as in the protection of tissue from physical damage, defence against pathogens, general stress response, wound healing etc. [20-22]. Because of their wide substrate specificity and high sensitivity, the PODs are useful tools for several biomedical, biotechnological, clinical and industrial applications [23,24]. PODs are also used in various reactions such as the synthesis of polymers, the development of drugs and organic molecules etc. Additionally, POD is one of the most important enzymes used to remove H2O2 enzymatically [18,19].
Organic compounds or metal complexes bearing sulfonamide moiety are widely used as inhibitors for various enzymatic reactions [25,26]. Although sulfonamide analogues are often used in the inhibition of carbonic anhydrase, these compounds are rarely used in the inhibition of peroxidase [27]. This case also raises a potential area of research. In this study, a new series of water soluble [N-(3-aminophenyl)-arylsulfonamides] hydrochloride (sulfonamide analogues (1-4)) were fabricated using 1,3-diaminobenzene, arylsulfonyl chloride and HCl solution and their structures were characterized by 1H-NMR, 13C-NMR, elemental analysis and FT-IR. In addition, their inhibitory effects of these compound on the activity of purified POD from red gabbage (Brassica oleracea var. capitata f. rubra) were evaluated for the first time. In the literature, there have been many studies have focused on the purification [17,22,28,30] and investigation of the POD inhibition by different compounds [27,31,34]. However, our study is the first investigation of the inhibition of purified the POD from red cabbage (Brassica oleracea var. capitata f. rubra) using these compounds.
2. Experimental
2.1. Materials and Methods
All chemicals used in the synthesis process (reagents and solvents) were purchased from chemical companies (Sigma-Aldrich, Merck and Alfa Aesar) and used as received unless otherwise stated. The 400 MHz 1H-NMR and 100.56 MHz 13C-NMR spectra were recorded at ambient temperature on a Bruker 400 NMR spectrometer in CDCl3 as solvent. NMR signals are given in parts per million (ppm) as δ downfield from tetramethylsilane (TMS) (δ 0.00) as an internal standard. Coupling constants are given in Hertz. The multiplicity of NMR peaks is abbreviated as follows: br = broad, d = doublet, m =multiplet, s = singlet, t = triplet. The elemental analyses were carried out using a Truspec MICRO (LECO) instrument. A Perkin-Elmer Spectrum 400 FTIR system with universal ATR sampling accessory was used to obtain the FT-IR spectra. For melting point determination, an Electrothermal 9100 instrument was used with open capillary tubes. A pH meter (SCHOTT CG840) was used for adjusting the pH. UV-Vis absorption measurements for the inhibition experiments were performed with Perkin-Elmer Lambda 25 UV/Vis spectrophotometers.
2.2. Plant Materials
The POD enzyme was purified from red cabbage (Brassica oleracea var. capitata f. rubra) using affinity chromatography in experiments. Fresh the red cabbage samples were purchased from a local market. Before homogenization, the red cabbage leaves were separated from the root, and the root was washed, dried, and crumbled with a blender.
2.3. Preparation of the Crude Extract
The homogenate preparation procedure for the POD was applied in accordance with the method described by the literature [18]. All the homogenization, centrifugation, dialysis, and chromatography processes were performed at +4 °C. For the preparation of crude extract, the red cabbage root sample (approximately 20 g) was shred, mixed with liquid nitrogen, and crushed in a mortar. Addition, the KH2PO4 (50 mL, 0.3 M, pH 7.0) solution was added. After this period, the mixture was centrifuged at 9.000 rpm for 60 min. Finally, the pellet was separated and discarded, and the raw extract was stored at −20 °C in small aliquots until use.
2.4. Standard Peroxidase Activity Assay
The POD activity was determined by the colorimetric method using guaiacol substrate and the increase in absorption due to the formation of 3,3'-dimethoxy-4,4'-biphenoquinone was measured at 470 nm by using a double beam UV-Vis spectrophotometer. In the all experiments were performed under optimum conditions (pH 6.0, 0.2 M KH2PO4, 25 °C). The activity assay mixture was prepared in accordance with the method described by the literature [18]. The activity assay mixture was prepared as follows: 1 mL of 45 mM guaiacol and 1 mL of 22.5 mM H2O2 were mixed in a test tube. Then, the POD enzyme (0.1 mL) was added to the activity assay mixture. The final volume of this mixture was adjusted to 3 mL by the addition of 0.2 M phosphate buffer. The changes of absorbance were monitored for 3 min at 25 °C. In the experiments, one unit of enzyme activity was defined as the amount of enzyme that changes the absorbency 0.01 per minute.
2.5. In Vitro Inhibition Studies
For the inhibition studies of the synthesized compounds, the POD activity was measured in the presence of different concentrations of inhibitors (N-(3-aminophenyl)-arylsulfonamide hydrochloride). As stated before, the activity assay mixture was prepared in accordance with the method described by the literature [18]. After the preparation of the activity assay mixture, the inhibition reaction was started by addition of certain amounts (25 µL, 50 µL and 75 µL) of inhibitors. The concentrations of activity assay compounds, i.e. H2O2 and guaiacol were kept constant (15 and 7.5 mM, respectively) while the concentration of inhibitor varied from 0.1375-0.4125 mM. The activity of POD was measured decrease at 470 nm using a double beam UV-Vis spectrophotometer. The decrease of activity (%) was plotted against inhibitor concentration in mM. A control sample without inhibitor (N-(3-aminophenyl)-arylsulfonamide hydrochloride) was used to determine 100% activity. The experiments were repeated three times and the results were given as mean ± SD.
2.6. General Procedure for the Synthesis of Ligands 1-5
Stage-1: A solution of N(CH2CH3)3 (13 mmol) in THF (tetrahydrofuran) (10 ml) was slowly added to a solution of R-sulfonyl chlorides (10 mmol) in THF (10 ml), immediately followed by 1,3-diaminobenzene, (10 mmol) in THF (10 ml). After the reaction time, the mixture was filtered and the volatiles were removed in vacuo. The residue was dissolved in DCM (dichloromethane) (15 ml) and washed with H2O (3 x 10 ml), dried over anhydrous MgSO4, filtered and concentrated to half of its volume in vacuo. The solution was purified by precipitation with diethyl ether from a CH2Cl2 solution to give a microcrystalline product. All the desired products were checked by FT-IR and NMR.
Stage-2: The HCl solution (5 mmol) was slowly added to DCM solutions of the each of prepared compounds (5mmol) with a dropping funnel and stirred at ambient temperature in a Schlenk tube for 2 h. After the times, the corresponding mixture was filtered. The crude products can be used without further purification. Analytically pure samples were characterized with NMR, FT-IR and EA (Figure-1).
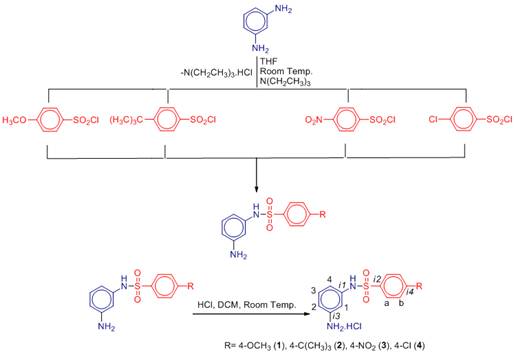
Figure 1. Synthesis of the compounds 1-4 together with NMR numbering scheme.
(1) N-(3-aminophenyl)-4-methoxybenzenesulfonamide hydrochloride
Color: White, Yield: 84 %, Mp: 268 oC, 1H-NMR (CDCl3, δ ppm):3.82 (s, 3H, -OCH3), 6.43 (d, 1H, J=8 Hz, -H2), 6.70 (br, -NH-, HCl), 6.52 (s, 1H, -H1), 6.77 (d, 1H, J=8 Hz, -H4), 6.89 (d, 2H, J=8 Hz, Hb), 6.95 (t, 1H, J=8 Hz, -H3), 7.72 (d, 2H, J=8 Hz, -Ha). 13C-NMR (CDCl3, ppm): 55.7(-OCH3), 107.5(-C1), 111.1(-C2), 111.8(-C4), 114.1(-Cb), 129.0(-Ca), 130.0 (-C3), 130.7(-Ci2), 137.9(-Ci1), 147.3 (-Ci3), 163.2(-Ci4). IR (cm-1): 3408 (NH2), 3318 (NH2), 3212 (N-H), 3092, 2944, 2851, 2801, 2770, 2657, 1608, 1576, 1481, 1450, 1421, 1403, 1331 (SO2), 1276, 1260, 1167, 1146 (SO2), 1078, 1006, 998, 897, 861, 837, 811, 797, 721, 690, 660, 629, 550. Anal. Calcd. For: [C13H15ClN2O3S], C: 49.60, H: 4.80, N: 8.90, S: 10.19 Found: C: 49.38, H: 4.91, N: 8.77, S: 10.25.
(2) N-(3-aminophenyl)-4-tert-butylbenzenesulfonamide hydrochloride
Color: White, Yield: 90 %, Mp: 215 oC, 1H-NMR (CDCl3, δ ppm): 1.32 (s, 9H, -C(CH3)3), 6.40 (d,J=8 Hz1H, -H2),6.44 (d, J=8 Hz 1H, -H4), 6.55 (s, 1H, -H1), 6.98 (t, 1H, J=16, 8 Hz, -H3),7.42 (d, 2H, J=8 Hz, -Hb), 7.77 (d, 2H, J=8 Hz, -Ha). 13C-NMR (CDCl3, ppm): 31.6(-C(CH3)3), 35.1 (-C(CH3)3), 107.3 (-C1), 110.8 (-C2), 111.8 (-C4), 126.1 (-Cb), 127.1 (-Ca), 130.1 (-C3), 136.3 (-Ci2), 137.7 (-Ci1), 147.4 (-Ci3), 156.7 (-Ci4). IR (cm-1): 3474 (NH2), 3391 (NH2), 3286 (N-H), 3063, 2920, 2909, 2831, 2770, 2701 1612, 1594, 1509, 1485, 1430, 1402, 1363, 1338, 1321, 1312 (SO2), 1296, 1284, 1197, 1167 (SO2), 1103, 1091, 960, 921, 846, 840, 770, 747, 691, 603, 555, 545, 529, 508, 497, 491, 478, 460, 452. Anal. Calcd. For: [C16H21ClN2O2S], C: 56.38, H: 6.21, N: 8.22, S: 9.41, Found: C: 56.51, H: 6.10, N: 8.34, S: 9.27.
(3) N-(3-aminophenyl)-4-nitrobenzenesulfonamide hydrochloride
Color: White, Yield: 87 %, Mp: 209 oC, 1H-NMR (DMSO-d6, δ ppm):6.20 (d, 1H, J=8 Hz, -H2), 6.23 (d, 1H, J=8 Hz, -H4),6.33 (s, 1H, -H1), 6.83 (t, 1H, J=8 Hz, -H3),7.98 (d, 2H, J=8 Hz, Ha), 8.37 (d, 2H, J=8 Hz, -Hb). 13C-NMR (DMSO-d6, δ ppm): 106.3 (-C1), 108.4 (-C2), 111.0 (-C4), 125.0 (-Cb), 128.7 (-Ca), 129.9 (-C3), 137.9 (-Ci1), 145.7 (-Ci2), 149.9 (-Ci3), 150.2 (-Ci4). IR (cm-1): 3436 (NH2), 3396 (NH2), 3228 (N-H), 3099, 3065, 2977, 2892, 2752, 2695, 1605, 1594, 1526, 1493, 1477, 1435, 1401, 1367, 1349, 1302 (SO2), 1273, 1180, 1152 (SO2), 1108, 1088, 1010, 969, 939, 875, 854, 835, 803, 782, 742, 680, 633, 602, 575, 548 (SO2), 528, 490, 465. Anal. Calcd. For: [C12H12ClN3O4S], C: 43.71, H: 3.67, N: 12.74, S: 9.72, Found: C: 43.71, H: 3.67, N: 12.74, S: 9.72.
(4) N-(3-aminophenyl)-4-chlorobenzenesulfonamide hydrochloride
Color: White, Yield: 89 %, Mp: 239 oC, 1H-NMR (DMSO-d6, δ ppm):6.38 (d, 1H, J=8 Hz, -H2), 6.44 (d, 1H, J=8 Hz, -H4), 6.51 (s, 1H, -H1), 6.99 (t, 1H, J=8 Hz, -H3), 7.38 (d, 2H, J=8 Hz, Ha), 7.72 (d, 2H, J=8 Hz, -Hb). 13C-NMR (DMSO-d6, δ ppm): 107.9 (-C1), 111.3 (-C2), 112.4 (-C4), 128.7 (-Cb), 129.4 (-Ca), 130.2 (-C3), 137.1 (-Ci1), 137.5 (-Ci4), 139.5 (-Ci2), 147.5 (-Ci3). IR (cm-1): 3390 (NH2), 3325 (NH2), 3214 (N-H), 3068, 2978, 2894, 2830, 2767, 2698, 1595, 1585, 1505, 1490, 1475, 1432, 1395, 1339, 1326, 1303 (SO2), 1279, 1184, 1172, 1149 (SO2), 1087, 1014, 997, 982, 962, 875, 837, 821, 787, 767, 749, 702, 681, 610, 551 (SO2), 518, 496, 478, 462. Anal. Calcd. For: [C12H12Cl2N2O2S], C: 45.15, H: 3.79, N: 8.78, S: 10.05, Found: C: 45.27, H: 3.65, N: 8.90, S: 10.17.
3. Results and Discussion
3.1. Synthesis
For the preparation of N-(3-aminophenyl)-arylsulfonamides hydrochloride compounds (1-4), the direct reaction was occurred between an equivalent amount of the corresponding N-(3-aminophenyl)-arylsulfonamides and one of the HCl solutions. All the compounds are air and moisture-stable in the solid state as well as in solutions. They were isolated as solids in very good yields and fully characterized by 1H-NMR, 13C-NMR, IR spectroscopy and elemental analyses and their melting points were determined. In the 1H-NMR spectra for the compound bearing sulfonamide moiety (1-4), for 1, the -H1, -H2, -H3, -H4 protons were assigned at 6.52 ppm as singlet, at 6.43 ppm as a doublet, at 6.95 ppm as a triplet and at 6.77 ppm as a doublet, respectively. The –Ha and –Hb protons were located at 7.72 ppm and 6.89 ppm as doublets, respectively. Also, over the sulfonamide ring substituent the –OCH3 protons were found at 3.82 ppm as a singlet peak. For the compound 2, the -H1, -H2, -H3, -H4 protons were observed at 6.55 ppm as a singlet, at 6.40 ppm as a doublet, at 6.98 ppm as a triplet and at 6.44 ppm as a doublet, respectively. The -C(CH3)3 protons were assigned at 1.32 ppm as a singlet peak. For 3, the chemical shifts of -H1-4 protons were located at 6.33 ppm as a singlet, at 6.20 ppm as a doublet, at 6.83 ppm as a triplet and at 6.23 ppm as a doublet, respectively. Similarly, for the compound 4, similarly, the chemical shifts of the -H1-4 protons were found at 6.51 ppm as a singlet, at 6.38 ppm as a doublet, at 6.99 ppm as a triplet and at 6.44 ppm as a doublet, respectively.
In the IR spectra for 1-4, the N-H stretching frequency peaks belonging to the sulfonamide fragment appeared at 3212, 3286, 3228 and 3214 cm-1 and NH2 stretching frequency peaks were observed at 3408-3318 for 1, 3474-3391 for 2, 3436-3396 for 3, and 3390-3325 for 4. Additionally, the SO2 stretching peaks were appeared at 1331 – 1146 for 1, 1312 – 1167 for 2, 1302 – 1152 for 3 and1303 – 1149 for 4. All the measurements and interpretations are compatible with the proposed structure of compounds 1-4.
3.2. Inhibition Effect of Peroxidase in Presence of 1-4
As known, the half maximal inhibitory concentration (IC50) value is a suitable parameter to show the inhibitor effect of a compound on a biological or biochemical function. To obtain the IC50 values, the POD activity was measured in the presence of different concentrations of N-(3-aminophenyl)-arylsulfonamide hydrochloride (0.1375-0.4125 mM) and an activity [N-(3-aminophenyl)-arylsulfonamide hydrochloride] plot was drawn (Figure 2). By plotting the activity (%) against inhibitor concentration, the amount of inhibitor that caused 50% rate reduction (IC50) was obtained. The half maximal inhibitory concentration (IC50) values were found to be 0.144 mM, 0.2 mM, 0.117 mM and 0.185 mM for 1, 2, 3 and 4 respectively, from the graph. When the investigated of results, the N-(3-aminophenyl)-4-nitrobenzenesulfonamide hydrochloride compound (3) was the most active compound for the POD compared to the others.
Horseradish peroxidase (HRP) is a well-known, highly investigated and characterized member of the peroxidase family. Also, several investigations on the purification and the inhibition of HRP by different compounds were carried out [30-33]. However, it is well known that the HRP has low stability with respect to hydrogen peroxide and acid concentration. This feature of HRP is the main disadvantage of its applications. The higher stability and wide substrate specificity of the POD purified from different sources allow the development of new analytical procedures or the improvement of its utilization in different applications.
Table 1. Inhibition of peroxidase by 1-4 compounds.
| Type of inhibitor | IC50 values (mM) |
| (1)- N-(3-aminophenyl)-4-methoxybenzenesulfonamide hydrochloride.HCl | 0.144±0.003 |
| (2)- N-(3-aminophenyl)-4-tert-butylbenzenesulfonamide hydrochloride.HCl | 0.2 ±0.002 |
| (3)-N-(3-aminophenyl)-4-nitrobenzenesulfonamide hydrochloride.HCl | 0.117±0.002 |
| (4)-N-(3-aminophenyl)-4-chlorobenzenesulfonamide hydrochloride.HCl | 0.185±0.003 |
In one study, Onsa et al. [30] purified two membrane-bound the PODs from sago palm (Merroxylon sagu) and the inhibitory effect of ascorbic acid, metabisulfite, l-cysteine and p-coumaric acid. In another study, Ruprapa et al. [29] purified an intracellular POD from the genetically transformed root cultures of red beet and investigated the inhibitory effect of potassium periodate and sodium azide. The POD from cassava leaves (Manihot esculenta Crantz of cv. KU50) was isolated and the inhibitory effect of some thiols and pesticides containing thiol groups was investigated by Wischada Jongmevasna et al. [34]. In all the studies, different inhibitory effects of the tried compounds were reported by the researchers. In one study, Kalın et al. [18] purified PODs from- Turkish blackradish (Raphanus sativus L.) and turnip (Brassica rapa L.) using 4–amino benzohydrazide affinity chromatography. They investigated the inhibitory effects of 4-aminobenzohydrazide on purified POD. The IC50 value was reported to be 0.726 mM for Turkish blackradish-POD and 1.167 mM for turnip-POD by reseachers [18]. In another study, a POD enzyme was isolated and purified from red cabbage (Brassica oleracea var. capitata f. rubra) using the same affinity chromatography and the inhibitory effect of the same inhibitor was investigated by Somtürk et al. [19]. The IC50 value was reported as 1.047 mM by the reseachers [19]. Similarly, in our study, the POD enzyme was purified from red cabbage (Brassica oleracea var. capitata f. rubra) using 4-aminobenzohydrazide affinity chromatography. The inhibitory effect of newly synthesized water soluble [N-(3-aminophenyl)-arylsulfonamides] hydrochloride compounds was investigated. The IC50 values were found to be 0.144±0.003 mM, 0.200±0.002 mM, 0.117±0.002 mM, 0.185±0.003 mM for 1, 2, 3 and 4 respectively, ([N-(3-aminophenyl)-arylsulfonamides] hydrochloride (Aryl= 4-methoxybenzene, 4-tert-butylbenzene, 4-nitrobenzene, 4-chlorobenzene) (1-4)). The inhibition results indicated that all the compounds (1-4) effectively inhibited the purified POD enzyme activity. In particular, the N-(3-aminophenyl)-4-nitrobenzenesulfonamide hydrochloride compound (3) was found as the most active compound.
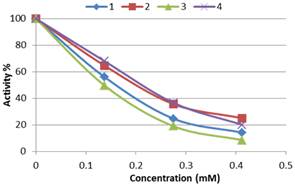
Figure 2. Activity (%) plot for compounds 1-4 for peroxidase in three different inhibitor concentrations.
4. Conclusion
Herein, we were synthesized N-(3-aminophenyl)-arylsulfonamide hydrochloride compounds (1-4) and the peroxidase inhibitory effect of the sulfonamide analogues (1-4) was tested. The most suitable parameters to show the effect of inhibition are the IC50values because these values determine the inhibitory effect of a compound on a biological or biochemical function. To determine the IC50value, the purified peroxidase activity was measured in the presence of three different concentrations of 1-4. A control sample without ligand was used to determine 100 % activity. Based on the graph, the IC50 values were determined to be 0.144±0.003 mM, 0.200±0.002 mM, 0.117±0.002 mM and 0.185±0.003 mM for 1-4 compounds for the red cabbage (Brassica oleracea var. capitata f. rubra) peroxidase, respectively. It was found that compound 3 was a more potent inhibitor on peroxidase activity than the other inhibitors. We believe that our inhibition results will shed light on further studies.
References
- Andersen K.K.; Jones D.N. Pergamon Press. Oxford, 1979, 3, 345.
- a) González-Alvarez, M.; Alzuet, G.; Borrás, J.; del Castillo Agudo, L.; García-Granda, S.; Montejo-Bernardo, J.M. Comparison of protective effects against reactive oxygen species of mononuclear and dinuclear Cu(II) complexes with N-substituted benzothiazolesulfonamides. Inorg Chem. 2005, 44, 9424-9433.
- Cumaoglu, A.; Dayan, S.; Agakaya, O. A.; Ozkul, Z.; Ozpozan Kalaycioglu N.; J Enz. Inhib. Med. Chem. 2015, DOI: 10.3109/14756366.2014.940938.
- Dayan, S.; Ozpozan Kalaycıoğlu, N.; Daran, J-C.; Labande, A.; Poli, R. Synthesis and characterization of half-sandwich ruthenium complexes containing aromatic sulfonamides bearing pyridinyl rings: Catalysts for transfer hydrogenation of acetophenone derivatives. Eur. J. Inorg Chem. 2013, 18, 3224-3232.
- Chohan, Z.H.; Shad, H.A.; Supuran, C.T. Synthesis; characterization and biological studies of sulfonamide Schiff's bases and some of their metal derivatives. J Enzyme Inhib Med Chem. 2012, 1, 58-68.
- Dayan, S; Arslan, F.; Ozpozan Kalaycıoğlu, N. Ru(II) impregnated Al2O3, Fe3O4, SiO2 and N-coordinate ruthenium(II) arene complexes: Multifunctional catalysts in the hydrogenation of nitroarenes and the transfer hydrogenation of aryl ketones. App. Cat. B. Environ. 2015, 164, 305-315.
- Turkmen, H.; Zengin, G.; Buyukkircali, B. Synthesis of sulfanilamide derivatives and investigation of in vitro inhibitory activities and antimicrobial and physical properties. Bioorg Chem. 2011, 3, 114-119.
- Gangapuram, M.; Mazzio, E.; Eyunni, S.; Soliman, K.F.; Redda, K.K. Synthesis and Biological Evaluation of Substituted N-[3-(1H-Pyrrol-1-yl)methyl]-1;2;5;6-tetrahydropyridin-1-yl]benzamide/benzene Sulfonamides as Anti-Inflammatory Agents. Arch. Pharm. 2014, 5, 360-9.
- Chen, Z.; Xu, W.; Liu, K.; Yang, S.; Fan, H.; Bhadury, P.S.; Hu, D.Y.; Zhang, Y. Synthesis and antiviral activity of 5‑(4‑chlorophenyl)-1;3;4-thiadiazole sulfonamides. Molecules. 2010, 12, 9046-9056.
- Bashandy, M.S.; Alsaid, M.S.; Arafa, R.K.; Ghorab, M.M. Design; synthesis and molecular docking of novel N;N-dimethylbenzenesulfonamide derivatives as potential antiproliferative agents. J Enzyme Inhib Med Chem. 2013, 3, 1-9.
- Funahashi, Y.; Sugi, N.H.; Semba, T.; Yamamoto, Y.; Hamaoka, S.; Tsukahara-Tamai, N.; Ozawa, Y.; Tsuruoka, A.; Nara, K.; Takahashi, K.; Okabe, T.; Kamata, J.; Owa, T.; Ueda, N.; Haneda, T.; Yonaga, M.; Yoshimatsu, K.; Wakabayashi, T. Sulfonamide derivative; E7820; is a unique angiogenesis inhibitor suppressing an expression of integrin alpha2 subunit on endothelium. Cancer Res. 2002, 21, 6116-6123.
- Güzel, O.; Innocenti, A.; Vullo, D.; Scozzafava, A.; Supuran, C.T. 3-phenyl-1H-indole-5-sulfonamides: structure-based drug design of a promising class of carbonic anhydrase inhibitors. Curr Pharm Des. 2010, 29, 3317-3326.
- Owa, T.; Okauchi, T.; Yoshimatsu, K.; Sugi, N.H.; Ozawa, Y.; Nagasu, T.; Koyanagi, N.; Okabe, T.; Kitoh, K.; Yoshino, H. A focused compound library of novel N-(7-indolyl)benzenesulfonamides for the discovery of potent cell cycle inhibitors. Bioorg Med Chem Lett. 2000, 11, 1223-1226.
- O'Brien, T. M.; Oliveira, P. J.; Wallace, K. B. Inhibition of the adenine nucleotide translocator by N-acetyl perfluorooctane sulfonamides in vitro; Toxic. Appl. Pharma. 2008, 227, 184–195.
- Vullo, D.; Prete, S. D.; Osman, S. M.; De Luca V.; Scozzafava A.; AlOthman Z.; Supuran C. T.; Capasso C.; Sulfonamide inhibition studies of the d-carbonic anhydrase from the diatom Thalassiosira weissflogii; Bioorg. & Med. Chem. Lett. 2014, 24, 275–279.
- Nishimori, I.; Vullo, D.; Minakuchi, T.; Scozzafava, A.; Capasso, C.; Supuran, C. T.; Sulfonamide inhibition studies of two b-carbonic anhydrases from the bacterial pathogen Legionella pneumophila; Bioorg. & Med. Chem. 2014, 22, 2939–2946.
- Şişecioğlu, M.; Gülçin, İ.; Çankaya, M.; Atasever, A.; Şehitoglu, M. H.; Kaya, H. B. Özdemir, H.; Purification and characterization of peroxidase from Turkish black radish (Raphanus sativus L.); J. Medic. Plants Res. 2010, 4, 1187-1196.
- Kalin, R.; Atasever, A.; Ozdemir, H.; Single-step purification of peroxidase by 4-aminobenzohydrazide from Turkish blackradish and Turnip roots; Food Chem. 2014, 150, 335–340.
- Somtürk, B.; Kalın R.; Özdemir, N.; Purification of Peroxidase from Red Cabbage (Brassica oleracea var. capitata f. rubra) by Affinity Chromatography. Appl. Biochem. Biotechnol. 2014, 173, 1815-1828.
- Leon, J.C.;Alpeeva, I. S.;Chubar, T. A.;Galaev, I.Y.;Csoregi,E.;Sakharov, I.Y.. Purification and substrate specificity of peroxidase from sweet potato tubers. Plant Sci. 2002, 163, 1011-1019.
- Ajila, C. M. Prasada R. Purification and characterization of black gram (Vigna mungo) husk peroxidase. J. Mol. Catal B: Enzym. 2009, 60, 36–44.
- Kouakou, T. H.; Dué, E. A.; Kouadio, N. E. J. P.; Niamké, S.; Kouadio, Y. J.; Mérillon, J M.;. Purification and Characterization of Cell Suspensions Peroxidase from Cotton (Gossypium hirsutum L.). Appl. Biochem. Biotech. 2009, 157, 575- 592.
- Colonna, S.; Gaggero, N.; Richelmi, C.; Pasta, P.; Recent biotechnological developments in the use of peroxidase; Trends. Biotechnol. 1999, 17, 163-168.
- Regalodo, C.; Garcia-Almandarez, B. E.; Duarte-Vazquez, M. A. Biotechnological applications of peroxidases. Phytochem. Rev. 2004, 3, 243–256.
- Prabhu, V.; Lui H.; King, J.; Arabidopsis dihydropteroate synthase: general properties and inhibition by reaction product and sulfonamides; Phytochemistry. 1997, 45, 23-27.
- Krungkrai, S. R.; Krungkrai, J.; Malaria parasite carbonic anhydrase: inhibition of aromatic/heterocyclic sulfonamides and its therapeutic potential Asian Pacific J. Tropical Biomed. 2011, 3, 233-242.
- Doerge, D. R.;Decker, C. J.;Inhibition of Peroxidase-Catalyzed Reactions by Arylamines: Mechanism for the Anti-Thyroid Action ofSulfamethazine Chem. Res. Toxicol. 1994, 7, 164-169.
- Srinivas, N.D.; Rashmi, K.R.; Raghavarao, K.S.M.S.; Extraction and purification of a plant peroxidase by aqueous two-phase extraction coupled with gel filtration; Process Biochem. 1999, 35, 43–48.
- Rudrappa, T.; Lakshmanan, V.; Kaunain, R.; Singara, N.M. Neelwarne, B.. Purification and characterization of an intracellular peroxidase from genetically transformed roots of red beet (Beta Vulgaris L.). Food Chem. 2007, 105, 1312–1320.
- Onsa, G. H.; Saari, N.; Selamat, J.; Bakar J. Purification and characterization of membrane-bound peroxidases from metroxylon sagu; Food Chem. 2004, 85, 365–376.
- Aspuru, E. O.; Zato´n, A. M L. Effect of glutathione on horseradish peroxidase activity; Spectrochim. Acta A Mol. Biomol. Spectrosc. 1999, 55, 2343–2346.
- Mishra B.; Priyadarsini K. I.; Mohan H.; Mugesh G. Horseradish peroxidase inhibition and antioxidant activity of ebselen and related organoselenium compounds. Bioorgan. & Medicin. Chem. Lett. 2006, 16, 5334–5338.
- Sariri, R.; Jafarian, V.; Hassan Sajedi, R.; Khajeh, Kh.; Inhibition of horseradish peroxidase by thiol type inhibitors: Mercaptoethanol and mercaptoacetic acid; J. Mol. Liq. 2006, 128, 175–177.
- Kabeya, L. M.; Marchi, A. A.; Kanashiro, A.; Lopes, N. P.; Silva, C. H. T. P.; Pupo, M. T.; Lucisano-Valim, Y. M.; Inhibition of horseradish peroxidase catalytic activity by new 3-phenylcoumarin derivatives: Synthesis and structure–activity relationships; Bioorg. & Med. Chem. 2007, 15, 1516–1524.



