Mg-Al Hydrotalcite Catalyzed Efficient One-Pot Synthesis of 4-H-Benzo[b]Pyrans, 2-Aryl Benzimidazole and 2-Aryl-4,5-Dihydro-1H-Imidazole Derivatives
Rohit Gupta1, Savita Ladage2, Lakshmy Ravishankar3, *
1Department of Chemical Sciences, Indian Institute of Science Education and Research-Kolkata (IISER-K), Mohanpur, Nadia,West Bengal, India
2Department of Chemistry, Homi Bhabha Centre for Science Education, Tata Institute of Fundamental Research, Mumbai, Maharashtra, India
3Department of Chemistry, KET’s V.G. Vaze College of Arts, Science & Commerce, Mulund (East), Mumbai, Maharashtra, India
Abstract
An environment friendly synthesis of 2-amino-3-cyano-4-aryl-7,7-dimethyl-5-oxo-4H-5,6,7,8-tetrahydrobenzo[b]pyran, 2- aryl benzimidazole and 2-aryl-4, 5-dihydro-1H-imidazole derivatives using Mg-Al hydrotalcite as an efficient catalyst has been developed. The excellent efficiency of the catalyst can be attributed to its Lewis acidic sites which resulted in higher reaction yields for different substituted benzaldehydes and shorter reaction time compared to conventional synthetic procedures. An added advantage is reusability of the catalyst, which demonstrates consistent activity over significant number of cycles after filtration and washing with ethanol.
Keywords
Multicomponent Reactions, Green Reactions, Mg-Al Hydrotalcite, Pyrans, Benzimidazole, Imidazole, Lewis Acid
Received: November 16, 2014
Accepted: January 15, 2015
Published online: February 2, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
In recent years, the growing environmental concern has led to the development of various green chemistry techniques which aim at reducing chemical waste, maximizing yields and minimizing costs without the use of toxic solvents or catalysts. In the mainstream of the current interest, multicomponent reactions have emerged as a powerful strategy to access diversified library of complex compounds from simple structures1.
Heterocyclic compounds form an important class of natural and synthetic compounds which are the essential component for synthesis of numerous molecules with high medicinal values2. Benzimidazole and Imidazole derivatives are an important group of heterocyclic scaffolds widely known for their high biological and pharmaceutical values. Benzimidazole and its derivatives are found in variety of drugs displaying a broad range of biological activities such as antifungal, anti-hypertension, anti-cancer and anti-viral effects3-5. In addition, imidazole and its derivatives are used as synthetic intermediates6, chiral auxiliaries7, chiral catalysis8, ligands for asymmetric catalysis9 and also have significant applications as important pharmaceutical compounds10.
4-H-Benzo[b]pyrans and their derivatives are another class of important heterocyclic compounds which have gained much attention in the last decade due to their biological and pharmacological activities, such as anticoagulants, spasmolytics, diuretic, anticancer and antianaphylactic activities11. 2-Amino-4H-pyran derivatives are useful as pigments and have also been used as photoactive materials12,13.
Various methods and catalysts have been employed for the synthesis of these compounds but no common catalyst has been reported which can be used for their synthesis. As a part of our detailed study on the development of new heterogeneous Lewis acid catalyst for the synthesis of the above mentioned heterocyclic compounds, herein we report Mg-Al hydrotalcites (Mg-Al HTs)as an efficient heterogeneous Lewis acid catalyst for the one-pot synthesis of 4-H-Benzo[b]pyrans, 2-aryl benzimidazole and 2-aryl-4, 5-dihydro-1H-imidazole derivatives.
Mg-Al HT is an eco-friendly material, and it has been shown that it can be synthesized even from the effluent of Friedal – Crafts Reaction14.They have been reported as efficient base catalysts for various organic transformations owing to the Lewis and Bronsted base sites present on the surface15-18 but to the best of our knowledge there have been no reports on their use as Lewis acid catalysts. Mg- Al HTs when calcined at temperatures above 1000 ⁰C exhibit Lewis acid sites similar to γ- Al2O319. Thus, exploiting the Lewis acidity of Mg-Al HTs, we tried to investigate the one pot synthesis of 4H-tetrahydrobenzo[b]pyran, 2-aryl benzimidazole and 2-aryl-4, 5-dihydro-1H-imidazole derivatives using these HTs.
2. Materials and Methods
The synthesis of Mg-Al HT was done as per the reported method. All the reagents were purchased from commercial sources and used without further purification. The characterization of the HT samples was done using powered X-ray Diffraction analysis measured on PANalyticalXpert pro X-ray diffractometer using Cu Kα radiation (0.154178nm). Melting points (mp.s) were determined on Veego model no. VMP-D melting point apparatus and are uncorrected.
2.1. General Procedure for Synthesis of Mg-Al Hydrotalcite
Mg-Al HT (Mg/Al = 0.33) was prepared using the coprecipitation method20, by simultaneous addition of aqueous solutions (25mL) of Mg(NO3)2.6H2O (0.033mol) and of Al(NO3)3.9H2O (0.1 mol) to a solution (40mL) containing NaOH(0.27mol) and Na2CO3 (0.05mol) with vigorous stirring at room temperature for 8h (pH =8–10). The precipitate of HT was collected and washed with water until the pH of the filtrate became 7. The Mg-Al HT was dried at 80 oC for 16h followed by activation by calcining it at1050 oC for 6h.As all the products are reported, they were characterized by comparison of their m.p.s with literature data.
2.2. General Procedure for Synthesis of 2-Aryl Benzimidazole Derivatives
A mixture of a substituted benzaldehyde (1.1 mmol), o-phenylenediamine (1 mmol) and Mg-Al HT (15 % w/w), in acetonitrile (5 mL) was stirred at room temperature. The progress of the reaction was monitored using TLC. After completion of the reaction, the solvent was evaporated, and the crude product was dissolved in 5mL of hot ethanol. The catalyst was recovered by filtration, the product obtained as filtrate was concentrated and cooled to give crystals of the pure product.
2.3. General Procedure for Synthesis of 2-Aryl-4, 5-Dihydro-1H-Imidazole Derivatives
A mixture of a substituted benzaldehyde (1.1 mmol), ethylenediamine (1 mmol) and Mg-Al HT (15 % w/w), in acetonitrile (5 mL) was stirred at room temperature. The progress of the reaction was monitored using TLC. After completion of the reaction, the solvent was evaporated, and the crude product was dissolved in 5mL of hot ethanol. The catalyst was recovered by filtration, the product obtained as filtrate was concentrated and cooled to give crystals of the pure product.
2.4. General Procedure for Synthesis of 2-Amino-3-Cyano-4-Aryl-7,7-Dimethyl-5-Oxo-4H-5,6,7,8-TetraHydrobenzo[b]Pyran Derivatives
A mixture of a substituted benzaldehyde (1 mmol), malononitrile (1mmol), dimedone (1 mmol), Mg-Al HT (15 % w/w), and water-ethanol (1:1) (10 mL) was stirred under reflux condition for an appropriate time. The progress of the reaction was monitored using TLC. After completion of the reaction, the mixture was kept at room temperature and the resulting solid product obtained was collected by filtration. The solid was then treated with hot ethanol and filtered to recover and reuse the catalyst. The product obtained as filtrate was concentrated and cooled to give crystals of the pure product.
3. Results and Discussions
To study the effect of Mg/Al ratio and the calcination temperature of Mg-Al HT on their Lewis acidity, HTs with different Mg/Al ratios (0.33, 0.6, 1.0 and 3.0) were calcined at 450 ⁰C , 750⁰C and 1050 ⁰C for 6h and were used to catalyse the reaction between benzaldehyde and o-phenylenediamine (Scheme 1). Mg-Al HT (Mg/Al = 0.33) calcined at 1050 ⁰C was found be to the most efficient in comparison to HTs with ratios 0.6, 1.0 and 3.0 (Table 1).
Table 1. Effect of Mg/Al ratio and calcination temperature on the lewis acidity of Mg-Al HTs in benzimidazole synthesis.
| Mg+2/Al+3 Ratio | Calcination Temp.(⁰C) | Yield % |
| 0.33 | 450 | 67 |
| 0.33 | 750 | 73 |
| 0.33 | 1050 | 80 |
| 0.66 | 450 | 62 |
| 0.66 | 750 | 67 |
| 0.66 | 1050 | 70 |
| 1.0 | 450 | 60 |
| 1.0 | 750 | 62 |
| 1.0 | 1050 | 65 |
| 3.0 | 450 | 58 |
| 3.0 | 750 | 60 |
| 3.0 | 1050 | 62 |
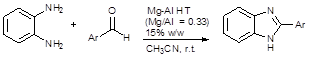
Scheme 1. Synthesis of 2-aryl benzimidazole derivatives using Mg-Al HT.
Benzimidazole derivatives are selectively synthesized in high yields via the condensation of o-phenylenediamine and substituted benzaldeydes using Mg-Al HT (Mg/Al=0.33) calcined at 1050 ⁰C (Scheme 1).The reaction was carried out using acetonitrile as solvent at room temperature. Similar observations were recorded for the condensation of ethylenediamine with substituted benzaldehydes using Mg-Al HT (Mg/Al =0.33) to give imidazole derivatives in high yields (Scheme 2).It was observed that for both benzimidazole and imidazole synthesis, substitution of electron withdrawing group on the aromatic aldehyde led to increase in the rate of reaction and high yields were obtained in shorter reaction time in comparison to substituents with electron donating substituents (Table 2,3).
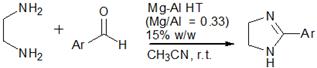
Scheme 2. Synthesis of 2-aryl-4,5-dihydro-1H-imidazolederivatives using Mg-Al HT.
Table 2. Synthesis of 2-aryl benzimidazole derivatives catalysed by Mg-Al HT.
| Aromatic aldehydes Ar- | Time (min) | Yield (%)* | mp (⁰C)(Lit. mp) |
| C6H5 | 60 | 79 | 289-291 (288-290)21 |
| 2-Cl C6H4 | 50 | 83 | 236 -237 (233-234)21 |
| 4-Cl C6H4 | 50 | 81 | 290-293 (292-293)21 |
| 4-F C6H4 | 45 | 86 | 248-250 (250-251)22 |
| 4-NO2 C6H4 | 45 | 91 | 310-312 (311-312)23 |
| 3-NO2 C6H4 | 55 | 85 | 208-210 (205-207)24 |
| 4-OMe C6H4 | 50 | 86 | 224-226 (227-228)23 |
| 2-OH C6H4 | 55 | 81 | 237-239 (236-237)25 |
| 4-Me C6H4 | 60 | 75 | 268-269 (270)26 |
| Cinnamaldehyde | 70 | 71 | 200-202 (199-202)27 |
| Furaldehyde | 70 | 71 | 284-285 (288)26 |
*isolated yield
Table 3. Synthesis of 2-aryl-4,5-dihydro-1H-imidazole derivatives catalysed by Mg-Al HT.
| Aromatic aldehydes Ar- | Time (min) | Yield (%)* | mp (⁰C)(Lit. mp) |
| C6H5 | 65 | 75 | 142-144 (140-142)28 |
| 4-Cl C6H4 | 60 | 77 | 245-247 (248)28 |
| 4-NO2 C6H4 | 55 | 81 | 308-311 (310-312)28 |
| 4-OMe C6H4 | 60 | 80 | 152-153 (152-154)28 |
| 4-Me C6H4 | 60 | 73 | 220-221 (223)28 |
*isolated yield

Scheme 3. Synthesis of 4H- tetrahydrobenzo[b]pyran derivatives using Mg-Al HT.
Mg-Al hydrotalcite (Mg/Al=0.33) calcined at 1050 ⁰C also efficiently catalyzes the synthesis of 2-amino-3-cyano-4-aryl-7,7-dimethyl-5-oxo-4H-5,6,7,8-tetrahydrobenzo[b]pyran derivatives in high yields. The reaction was carried out through the one-pot, three-component reaction of benzaldehyde, malononitrile, and dimedone using water and 1:1 water/ethanol mixture as solvent under reflux conditions (Scheme 3). To explore the scope of this reaction, the reaction was extended to various aromatic aldehydes. The reaction time, yield and the physical constants of various products obtained by the reaction are given in (Table 4).
High yields were obtained regardless of the structural variations in the aromatic aldehyde, but with varied reaction time under these reaction conditions. Aromatic aldehydes substituted with electron-donating groups led to products with slightly lower yields and have taken longer reaction time as compared to the aldehydes containing electron-withdrawing groups. As seen in (Table 4), in case water and 1:1 water/ ethanol mixture as solvents it was observed that 1:1 water/ ethanol was more efficient and resulted in high yields of compounds in shorter reaction time.
Table 4. Synthesis of 2-amino-3-cyano-4-aryl-7,7-dimethyl-5-oxo-4H-5,6,7,8-tetra-hydrobenzo[b]pyran derivatives catalyzed by Mg-Al HT.
| Aromatic aldehydes Ar- | Time (min) | Yield (%)* | mp (⁰C) (Lit. mp)* | ||
| (H2O) | (1:1, H2O:Ethanol) | (H2O) | (1:1, H2O:Ethanol) | ||
| C6H5 | 50 | 40 | 85 | 87 | 227-229 (226-228)29 |
| 2-Cl C6H4 | 80 | 70 | 86 | 89 | 205-207 (217-218)30 |
| 4-Cl C6H4 | 60 | 50 | 84 | 86 | 211-213 (209-211)29 |
| 4-F C6H4 | 80 | 65 | 81 | 91 | 178-180 (176-178)31 |
| 4-NO2 C6H4 | 70 | 45 | 92 | 95 | 184-186 (177-178)30 |
| 3-NO2 C6H4 | 60 | 45 | 89 | 90 | 205-208 (208-210)30 |
| 4-OMe C6H4 | 90 | 80 | 90 | 92 | 191-193 (199-201)30 |
| 4-Me C6H4 | 90 | 80 | 80 | 81 | 204-205 (214-216)30 |
| Furfuraldehyde | 100 | 80 | 73 | 74 | 205-207 (218-220)29 |
| Cinnamaldehyde | 105 | 85 | 75 | 77 | 206-208 (205-207)31 |
*isolated yield
The high catalytic efficiency of Mg-Al hydrotalcites (Mg/Al = 0.33) in all the three reactions may be attributed to the Lewis acid sites similar to γ-Al2O3 attained on calcining it to higher temperatures around 1050 ⁰C. The melting points of all the compounds compared well with the reported values.
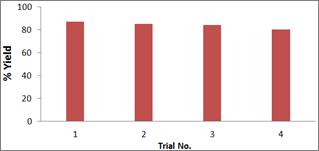
Figure 1. The reusability of Mg-Al HT catalyst for the synthesis of 4-H-tetrahydrobenzo[b]pyrans.
Apart from the eco-friendly conditions and the high catalytic efficiency, the catalyst provides the simplicity of product isolation and the advantage of possible recovery and recyclability. To study the reusability of Mg-Al HTs, repeated trials of the one pot synthesis of 4-H-tetrahydrobenzo[b]pyrans was done. The catalyst was separated by filtration after dissolving the products in hot ethanol and was directly reused after washing with ethanol for subsequent trials. It was found that the catalyst retains much of its catalytic activity even after four consecutive trials (Figure 1).
4. Conclusion
We present a new and efficient methodology for the synthesis of 2-amino-3-cyano-4-aryl-7,7-dimethyl-5-oxo-4H-5,6,7,8-tetrahydrobenzo[b]pyran , 2- aryl benzimidazole and 2-aryl-4,5-dihydro-1H-imidazole derivatives using Mg-Al hydrotalcites (Mg/Al = 0.33) calcined at 1050 ⁰C as an efficient Lewis acid catalyst. The eco-friendly nature of the catalyst, safer reaction conditions, high yields, shorter reaction times and possible reuse of the catalyst makes these reported procedures an effective contribution to the existing processes in the field of 4H-benzo[b]pyrans, benzimidazole and imidazole derivatives synthesis.
Acknowledgements
This work was done by Rohit Gupta as a part of National Initiative on Undergraduate Science (NIUS) Chemistry Programme (Batch – V, 2008-2010) of Homi Bhabha Centre for Science Education (HBCSE, TIFR). We thank Bhabha Atomic Research Centre (BARC) and Tata Institute of Fundamental Research (TIFR) for the powder XRD analysis. We also acknowledge the support extended by the staff members at the chemistry laboratory of HBCSE.
References
- (a) Ugi, I. Pure Appl. Chem. 2001, 731, 87.(b) Dömling, A.; Ugi, I. Angew. Chem. Int. 2000, 39, 3168.
- (a) Varma, R. S.J. Heterocycl. Chem. 1999, 36, 1565. (b) Eicher, T.; Hauptmann The Chemistry of Heterocycles: Structure, Reactions, Syntheses, and Applications 2nd Ed. 2003, Wiley - VCH, Weinheim.
- Preston, P. N. Chem. Rev. 1974, 74, 279.
- Horton, D. A.; Bourne, G. T.; Sinythe, M. L. Chem. Rev.2003, 103,893.
- Alamgir, M.; Black, St. C. D.; Kumar, N. T. Heterocycl. Chem. 2007, 9, 87.
- (a) Jones, R. C. F.; Nichols, J. R. Tetrahedron Lett. 1990, 31, 1771. (b) Jones, C.; Smallridge, M. J.; Chapleo, C. B. J. Chem. Soc. Perkin Trans. 1991,385. (c) Hayashi, T.; Kishi, E.; Soloshonok, V. A.; Uozumi, Y.Tetrahedron Lett.1996, 37, 4969. (d) Lin, Y. R.; Zhou, X. T.; David, L. X.; Sun, J. J. Org. Chem.1997,62, 1799. (e) Hsiao, Y.; Hegedus, L. S. J. Org. Chem. 1997,62,3586. (f) Jung, M. E.; Huang, A. Org. Lett.2000, 2, 2659.
- (a) Jonesv, R. C. F.; Turner, I.;Howard, K. J.;Tetrahedron Lett.1993,34,6329. (b) Langlois, Y.;Dalko, P. I.J. Org. Chem.1998, 63, 8107. (c) Jones, R. C. F.;Howard,S.; Naith, K. J.Tetrahedron Lett.1996,37,1707.
- (a) Corey, E. J.;Grogan, M. J.Org. Lett.1999, 1,157. (b) Isober, T.; Fukuda,K.; Araki, Y.;Ishikawa, T.Chem. Commun.2001,243.
- (a) Botteghi, C.;Schionato, A.;Chelucci, G.; Brunner, H.;Kurzinger, A.;Obermann,U. J.Organomet. Chem.1989, 370, 17. (b) Morimoto, T.; Tachibana, K.;Achiwa, K.Synlett,2003, 783.(c) Davinport, A. J.; Davies, D. L.;Faweett, J.;Russell, D. R.J.Chem. Soc., Perkin Trans2001,11,500. (d) Menges, F.;Neuburger, M.;Pfaltz,A.Org. Lett.2002, 44,713. (e) Boland,N. A.; Casey, M.; Hynes, S. J.;Matthews,J. W.;Muller-Bunz, H.;Wilkes, P. Org. Biomol. Chem.2004, 2, 1995.
- (a) Preston, P. N.; Stevens, M F. G.;Tennant, G.Benzimidazoles and Congeneric Tricyclic Compounds, Part 2, John Wiley & Sons: New York 1980. (b) Cedillo-Rivera, R.;Munoz, O.J. Med. Microbiol1992, 37, 221 (c) Chavez, B.;Cedillo-Rivera, R.;Martiner-Palomo, A.J. Protozool1992,39,510. (d) Navarrete-Vazquez, G.;Cedillo, R.; Hernandez-Campos, A.;Yepez, L; Hernandez-Luis, F.; Valdez, J.; Morales, R.;Cortes, R.; Hernandez, M.;Castillo, R.Bioorg. Med. Chem. Lett.2001,11,187.
- Mobinikhaledi, A.; Fard, M.A.B.ActaChim. Slov.2010,57,931.
- Ellis, G. P.In The Chemistry of Heterocyclic Compounds. Chromenes, Chromanes and Chromones;Weissberger, A., Taylor, E. C., Eds.; John Wiley: New York, 1977, chapter 11, pp 11.
- Armetso, D.; Horspool, W. M.; Martin, N.; Ramos, A.J. Org. Chem.1989, 54, 3069.
- Kshirsagar, S. W.; Patil, N. R.; Samant, S. D.Green Chem. Lett. and Rev.2010,3,335.
- Suzuki, E.; Ono, Y.Bull. Chem. Soc. Jpn.1988,61,1008.
- Suzuki, E.; Okamoto, M.; Ono, Y.J. Mol. Catal. 1990,61,283.
- Cativiela, C.;Figueras, F.; Garcia, J. I.; Mayoral, J. A.; Zurbano, M. M.Synth. Commun.1995,25,1745.
- Cativiela, C.; Figueras, F.; Fraile,J.M.; Garcia, J. I., Mayoral, J. A.Tetrahedron Lett.1995,36, 4125.
- Shen, J.; Kobe, J. M.; Chen, Y.; Dumesic, J. A.Langmuir1994,10,3902.
- Cavani,F.;Trifiro, F.; Vaccari, A.Catal. Today1991, 11, 173.
- Sharghi, H.; Beyzavi, M. H.; Doroodmand, M. M.Eur. J. Org.Chem. 2008,24,4126.
- Du, L. H.; Wang, Y. G.Synthesis2007,675.
- Chakrabarty, M.; Mukherjee, R.;Karmakar, S.;Harigaya, Y. Monatsh Chem.2007, 138, 1279.
- Heravi, M.M.;Tajbakhsh, M.;Ahmadi,A. N.;Mohajerani, B.Monatsh Chem. 2006,137, 175.
- Sahu, D. P.; Ponnala, S.Synth. Commun. 2006, 36, 2189.
- Allouma,A. B.;Bougrin, K.;Soufiaoui,M.Tetrahedron Lett.2003, 44,5935.
- Han, X.; Ma, H.; Wang, Y. Russ.J. Org. Chem.2008, 44, 863.
- Haneda, S.; Okui, A.; Ueba, C.; Hayashi, M.Tetrahedron2007, 63, 2414.
- Wang, X. S.; Shi, D. Q.; Tu, S. J.; Yao, C. S. Synth. Commun.2003, 33, 119.
- Jin, T.S.; Wang, A. Q.; Wang, X.; Zhang, J. S.; Li, T.S.Synlett.2004, 871.
- Gurumurthi, S; Sundari, V.; Vallippan, R.E-J Chem.2009, 6(S1), S466.



