One-Pot Synthesis of 1,8-Dioxooctahydroxanthenes Catalyzed by Mg-Al Hydrotalcites
Rohit Gupta1, Savita Ladage2, Lakshmy Ravishankar3, *
1Department of Chemical Sciences, Indian Institute of Science Education and Research-Kolkata (IISER-K), Mohanpur, Nadia, West Bengal, India
2Department of Chemistry, Homi Bhabha Centre for Science Education, Tata Institute of Fundamental Research, Mumbai, Maharashtra, India
3Department of Chemistry, KET’s V.G. Vaze College of Arts, Science & Commerce, Mulund (East), Mumbai, Maharashtra, India
Abstract
An environment friendly synthesis of 1,8-dioxooctahydroxanthenes derivatives using Mg-Al hydrotalcite as an efficient Lewis acid catalyst has been developed. Water was used as a green solvent and the reaction gave products in high yields in shorter reaction time and involved easy work up. The reaction works well with both electron deficient and electron rich aldehydes.
Keywords
Multicomponent Reactions, Green Reactions, Mg-Al Hydrotalcite, Xanthenes, Lewis Acid
Received: November 5, 2014
Accepted: January 19, 2015
Published online: January 29, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Multicomponent reactions (MCRs) have emerged as a powerful tool for the synthesis of complex structures from simple substrates with high selectivity and high atom efficiency.1-8One-pot MCRs are the useful path for the facile generation of combinatorial libraries of various organic molecules of high synthetic and biological value.9,10Xanthene and its derivatives are of considerable interest due to a plethora of biological and pharmaceutical activities like antiviral,11anti-inflammatory12and anti-bacterial activities.13 They also find their applications in photodynamic therapy,14 in laser technology15,16and as pH sensitive fluorescent material for the visualization of bio-molecular assemblies.17Numerous methods have been reported for the synthesis of 1,8-dioxooctahydroxanthenes. The reaction of aromatic aldehydes with 1,3- diketones has been promoted by various catalysts such as p-dodecylbenzenesulfonic acid in aqueous media,18p-toluenesulfonic acid (p–TsOH) in organic solvents,19benzyltriethylammonium chloride,20diammonium hydrogen phosphate,21MCM-41 functionalized sulfonic acid under ultrasonic irradiation22 and heterogeneous catalysts (NaHSO4-SiO2 and silica chloride),23 InCl3·4H2O in ionic liquids,24 Amberlyst-15,25polyaniline-p-toluenesulfonate salt26 and PPA-SiO2.27These methods have certain drawbacks which include prolonged reaction time, use of hazardous organic solvents, toxic catalysts and low catalytic efficiency.
Hydrotalcites (HTs) are the anionic clays which are layered double hydroxides whose structure is similar to brucite [Mg(OH)2]. They are represented by a general formula [M1-x2+Mx3+(OH)2(An-)x/n.yH2O], where M2+ andM3+are divalent andtrivalent cations respectively and An-is the interlayer charge balancing inorganic or organic anion. The divalent cation in most of the cases occupies the octahedral voids with anion and water in the interlayer’s. Mg-AlHTs upon calcinationare known to possess both acidic and basic sites.28In recent years, Mg-Al HTs have gained much attention as heterogeneous base catalysts for various chemical transformations such as the aldol and Claisen–Schmidt condensations or the Knoevenagel and Michael additions owing to their surface Lewis basic sites.29-31Recently, we have studied the effect of different phases of Mg-Al HTs formed by calcination on the Knoevenagel reaction of benzaldehydes and malononitrile.32 However, there has been no report about the use of Mg-Al HTs as heterogeneous Lewis acid catalysts. Mg-Al HTs exhibit Lewis acid sites similar to γ-Al2O3 when calcinated at higher temperatures28. Herein we report Mg-Al hydrotalcite (HT) as a new efficient heterogeneous Lewis acid catalyst for preparation of 1,8-dioxooctahydroxanthenes which helps to overcome the problems inherent in the procedures reported earlier. To the best of our knowledge, this methodology has not been reported previously.
2. Materials and Methods
Mg-Al HT was prepared by the reported method. 33All the reagents were purchased from commercial sources and used without further purification. The characterization of the HT samples was done using powered X-ray Diffraction analysis measured on PANalytical Xpert pro X-ray diffractometer using CuKα radiation (0.154178nm). Melting points (mp.s) were determined on Veego model no. VMP-D melting point apparatus and are uncorrected.
2.1. General Procedure for the Synthesis of Mg-Al Hydrotalcite
Mg-Al HT (Mg/Al = 0.33) was prepared using the co-precipitation method, 33by the simultaneous addition of aqueous solutions (25mL) of Mg(NO3)2∙6H2O (0.033mol) and of Al(NO3)3∙9H2O (0.1 mol) to a solution (40mL) containing NaOH (0.27mol) and Na2CO3 (0.05mol) with vigorous stirring at room temperature for 8h (pH =8–10). The precipitate of HT was collected and washed with water until the pH of the filtrate was 7. The Mg-Al HT was dried at 80 oC for 16h followed by activation by calcination by heating the catalyst to 1050 oC in the furnace for 6 h and then slowly cooling it to room temperature.
2.2. General Procedure for the Synthesis of 1,8-Dioxooctahydroxanthenes
A mixture of a substituted benzaldehyde (1 mmol), dimedone (280 mg, 2 mmol), Mg-Al HT (42 mg, 15 % w/w), and water or ethanol (10 mL) was stirred at reflux for the appropriate time (Table1). The progress of the reaction was monitored using silica gel TLC (EtOAc : hexane = 2:8). After completion of the reaction, the mixture was kept at room temperature and the resulting solid was collected. It was treated with hot ethanol (5 mL) and the mixture was filtered to recover the catalyst. The filtrate was concentrated and cooled to give the pure product in crystalline form.
3. Results and Discussions
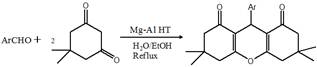
Scheme 1. Synthesis of 1,8-Dioxooctahydroxanthenes
Several 1,8-dioxooctahydroxanthenes were synthesized under reflux conditions in water and ethanol. Table 1shows that both electron-deficient and electron-rich aromatic aldehydes reacted with 2 equivalents of dimedone to afford the corresponding 1,8-dioxooctahydroxanthenes in high yields using Mg-Al HT as catalyst (Scheme 1). The catalytic activity may be attributed to the presence of mild acidic sites in Mg-Al HT (Mg/Al = 0.33) when calcined at 1050 ⁰C. Aldehydes bearing electron-withdrawing groups gave higher yields than those substituted with electron-donating groups. The use of water and ethanol both resulted in excellent yields of 1,8-dioxooctahydroxanthenes ranging from 73-95%, although, reaction in water required less time. In order to test the scope of the reaction, formaldehyde and acetaldehyde were treated with dimedone in water and in ethanol, however, no product was formed even after stirring at reflux for eight hours. Interestingly, the reaction of benzaldehdye with acetylacetone did not yield any product under identical conditions, this failure may be due to the fact that acetylacetone exists mostly in its enol form (80%) and even more so in polar solvents. This fact may explain why it does not react because it is tied up to the acidic sites of the catalyst. Enolization is negligible in dimedone.34
Table 1. Synthesis of 1,8-Dioxooctahydroxanthenes catalyzed by Mg-Al HT.
| Aldehydes | Time (h) | Yield (%) | mp (⁰C)(Lit. mp) | ||
| (Water) | (Ethanol) | (Water) | (Ethanol) | ||
| C6H5CHO | 3 | 4 | 85 | 87 | 204-205 (202-204)16 |
| 2-Cl C6H4 CHO | 3.5 | 4 | 86 | 89 | 228-230 (228-230)16 |
| 4-Cl C6H4 CHO | 3 | 3.5 | 84 | 86 | 225-226 (228-230)16 |
| 4-F C6H4 CHO | 3 | 3.5 | 88 | 90 | 224-225 (224-226)22 |
| 4-NO2 C6H4CHO | 3 | 3.5 | 92 | 95 | 226-227 (226-228)16 |
| 3-NO2 C6H4CHO | 3 | 4 | 89 | 90 | 168-170 (168-170)16 |
| 4MeOC6H4CHO | 4 | 4.5 | 90 | 92 | 243-245 (242-244)16 |
| 2-HO C6H4 CHO | 4 | 4.5 | 83 | 86 | 202-205 (205-206)35 |
| 4-Me C6H4 CHO | 4 | 4.5 | 80 | 81 | 218-220 (217-218)16 |
| Cinnamaldehyde | 4.5 | 5 | 75 | 77 | 177-178 (175-177)16 |
| Furfuraldehyde | 4.5 | 4.5 | 73 | 74 | 62-63 (62-64)16 |
| HCHO | 8 | 8 | - | - | |
| CH3CHO | 8 | 8 | - | - | |
4. Conclusion
In conclusion, a novel one-pot synthesis of 1,8-dioxooctahydroxanthenes using aromatic aldehydes and dimedone has been developed. It is notable that the catalyst is easy to prepare, less toxic than some of the earlier reported sulfonic acid catalysts and is environmental friendly. High yields, mild reaction conditions, short reaction times and the use of green solvent such as water are the notable advantages of this procedure. In addition the reaction works well with both electron deficient and electron rich aldehydes.
Acknowledgements
The experimental work was done by Rohit Gupta as a part of the National Initiative on Undergraduate Science (NIUS) Chemistry Programme (Batch –V, 2008-2010) of Homi Bhabha Centre for Science Education (HBCSE, TIFR). We thank Bhabha Atomic Research Centre (BARC) and Tata Institute of Fundamental Research (TIFR) for the powder XRD analysis. We also acknowledge the support extended by the staff members at the chemistry laboratory of HBCSE.
References
- Ugi I, Pure Appl. Chem. 2001, 73, 187 and references therein; For a monograph, see: Multicomponent Reactions; Zhu J &Bienayme H, Eds.; Wiley-VCH: Weinheim, Germany 2005.
- Domling A, Chem. Rev. 2006, 106, 17.
- D’Souza D M & Mueller T J J, Chem. Soc. Rev. 2007, 36, 3169.
- Cariou C C A, Clarkson G J & Shipman M J, J. Org. Chem. 2008, 73, 9762.
- Alizadeh A, Mobahedi F &Esmaili A, Tetrahedron Lett. 2006, 47, 4469.
- Umkeherer M, Kalinski C, Kolb J &Burdack C, Tetrahedron Lett. 2006, 47, 2391.
- Syamala M, Org. Prep. Proced. Int. 2005, 37, 103.
- Syamala M, Org. Prep. Proced. Int. 2009, 41, 1.
- Elders N, Schmitz R F, de Kanter F J J, Ruijter E, Groen M B &Orru R V A, J. Org. Chem. 2007, 72, 6135.
- Groenendaal B, Ruijter E &Orru R V A, Chem. Commun. 2008, 43, 5474.
- Lambert R W, Martin J A, Merrett J H, Parkes K E B & Thomas G J,PCT Int. Appl. WO,1997, 9706178,; Chem. Abstr. 1997,126, 212377y.
- Poupelin J P, Saint-Ruf G, Foussard-Blanpin O, Narcisse G, Uchida-Ernouf G &Lacroix R, Eur. J. Med. Chem. 1978, 13, 67.
- Hideo T &Teruomi J, Jpn. Patent 56005480, 1981; Chem. Abstr. 1981, 95, 80922b.
- (a) Ion R M, Prog. Catal. 1997, 6, 55; (b) Ion R M, Planner A, Wiktorowicz K &Frackowiak D, ActaBiochim. Pol. 1998, 45, 833.
- Sirkencioglu O, Talinli N &Akar A J, Chem. Res. 1995, 502.
- Ahmad M, King T A, Ko D K, Cha B H & Lee J, J. Phys. D: Appl. Phys. 2002, 35, 1473.
- Knight C G & Stephens T, Biochem. J. 1989, 258, 683.
- Jin T S, Zhang J S, Xio J C, Wang A Q & Li T S, Synlett 2004, 866.
- Khosropour A R, Khodaei M M&Moghannian H, Synlett 2005, 955.
- Wang X S, Shi D Q, Li Y L, Chen H, Wei X Y &Zong Z M, Synth. Commun. 2005, 35, 97.
- Darvish F, Balalaei S, Chadegani F &Salehi P, Synth. Commun. 2007, 37, 1059.
- Jin T S, Zhang J S, Wang A Q & Li Y S, Ultrason. Sonochem. 2006, 13, 220.
- Das B, Thirupathi P, Reddy K R, Ravikanth B &Nagarapu L, Catal. Commun. 2007, 8, 535.
- Fan X, Hu X, Zhang X & Wang J, Can. J. Chem. 2005, 83, 16.
- Das B, ThirupathiP, Mahender I, Reddy V S & Rao Y K, J. Mol. Catal. A: Chem. 2006, 247, 233.
- John A, Yadav J P &Palaniappan S, J. Mol. Catal. A: Chem. 2006, 248, 121.
- Kantevari S, Bantu R &Nagarapu L, J. Mol. Catal. A: Chem. 2007, 269, 53.
- (a)De Roy A, Forano C, Malki El, Besse K J P, Synthesis ofMicroporous Materials; Van Nostrand-Reinhold: New York, 1992. (b) Figueras F, Top. Catal, 2004, 29, 189. (c)Shen J, Kobe J M, Chen Y &Dumesic J A, Langmuir 1994, 10, 3902.
- Suzuki E & Ono Y, Bull. Chem. Soc. Jpn1988, 61, 1008.
- Suzuki E, Okamoto M & Ono Y, J. Mol. Catal. 1990, 61, 283.
- (a) Cativiela C, Figueras F, Garcia J I, Mayoral J A &Zurbano M M, Synth. Commun.1995, 25, 1745. (b) Cativiela C, Figueras F, Fraile J M, Garcia J I & Mayoral J A, Tetrahedron Lett.1995, 36, 4125.
- Gupta R, Kshirsagar S, Ladage S, Samant S D, SMC Bulletin2014, 3 (4), 29.
- Cavani F,Trifiro F &Vaccari F, Catal. Today 1991, 11, 173.
- Cook G &Feltman P M,J. Chem. Educ.2007, 84(11), 1827.
- Horning E C &Horing M G, J. Org. Chem. 1946, 11, 95.



