Brain Activity and Special Relativity: Estimation and a Novel Hypothesis to Explain Time Perception
Amir Hosein Ghaderi*
Cognitive Neuroscience Lab., University of Tabriz, Tabriz, Iran
Abstract
The theory of special relativity suggests that, time is a byproduct of velocity and it is created during movement, obligatory. Subjective time is perceived during all senses and this perception also is obligatory. In this paper, I suppose that psychological perceived time is analogous to physical relative time. The brain uses many neural pathways in sensory system for perception. Overall the length of these pathways is very large and the information network is very huge. On the other hand, binding in this network is occurred in very little time. So the velocity of data transfer and integration in this network is too high. I suggest that time perception is related to this high speed. In this paper the internal clock and other dedicated models have been considered as Newtonian timing systems (an invalid theory). Also two time perception models which are based on the theory of special relativity are criticized and a novel hypothesis based on special relativity and brain activity is presented. The proposed hypothesis suggests that, the velocity of integration in the human cortex is near the speed of light and subjective time dilation and compression is occurred due to this relativistic speed. Many time distortions during psychological tasks and many physiological evidences are consistent with this novel hypothesis.
Keywords
Time Perception, Einstein Special Relativity, Brain Activity, Integration
Received:May 8, 2015
Accepted: May 18, 2015
Published online: June 23, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
At the beginning of the twentieth century, "time" was a challenging topic in physics. Earlier, the absolute time and space had been introduced by Sir Isaac Newton in 1687. According to Newton, time is an imperceptible and incomprehensible variable (the true and mathematical time) and he believed that humans perceive a relative, apparent and common aspect of time [1]. In 1905 Albert Einstein presented the special relativity. This theory states that the time is a relative variable and it is created by motion. Einstein’s first postulate, states that the laws of physics are the same in every inertial frame of reference.
As a result, the special relativity theory proves that a moving clock run slower than an observer’s stationary clock. Sensible time dilation occurs when the clock is moving close to the speed of light [2]. The special relativity theory was confirmed by many experiments. Also this theory explained many physical observations and therefore the Newton theory about true and mathematical time was failed.
However, the subjective time 12 is a confusing topic. In the past 50 years, many time perception models have been proposed. A traditional model, called internal clock, suggests that the time is an absolute parameter and it is produced by a pacemaker [3,4]. In this model, an accumulator counts ticks and elapsed time is estimated in terms of tick rate. Although this modular and dedicated model [5] justifies some aspects of time perception but there are not sufficient evidences in physiology that indicate a specific module for time perception [6]. Cerebellum, striatum, prefrontal cortex, parietal cortex and other areas of the brain are maintained in time encoding, storage and retrieval [5-10]. While patients with cerebellar disorder are impaired in time reproduction and time discrimination tasks [5,7], in some of studies the role of cerebellum is completely excluded [8]. Although it seems that all of these areas are involved in timing, but we cannot confine time perception only to these areas. Recently, new evidences suggest that other areas such as insular and temporal cortex can be involved in time perception [9,10].
In recent years intrinsic models have been developed and have found strong evidences. This idea that neural activity can inherently create time is claimed by intrinsic models [11]. Computer modelling shows that there is no a linear metric of time and time is encoded by changes in neural network state [12]. A similar model assumes that spatiotemporal processing is a consequence of hidden neuronal states, such as short-term synaptic plasticity [13]. An intrinsic measure for duration is obtained by changes in heart rate or interoceptive signals such as the body temperature [11,14]. Therefore, the activity of anterior insular cortex, a critical area involve in emotional awareness, is considerable in time perception [10].
Moreover, a significant interesting model suggests that the time is encoded intrinsically by the energy spent in the brain [6]. In this model the brain activity level is associated with perceived duration. During many physiological changes, such as repetition suppression [15,16], less neural activity during a saccade [6,17], less activation when viewing a darker [6,18], or a smaller stimuli [6] and several others, a time distortion is observed. In this model, more brain activity leads to perceived time dilation [6,19,20].
Time dilation and time compression are specific concepts in the theory of special relativity. In the recent years the saccadic compression of space and subjective time is investigated in this framework and it is suggested that time compression during a saccade is a relativistic effect [21,22]. But there is a fundamental mistake in this theory.
In the present paper, an alternative timing model based on special relativity is proposed. The proposed hypothesis suggests that, velocity of information exchange in human cortex is on the order of light speed and time dilation and time compression in perceived durations is occurred due to this relativistic speed. Physiological aspects of time perception are consistent with this idea. This hypothesis is based on this fact that, time in the mind follows from the physical rules (as the first postulate of special relativity).
The section 2 of this paper is devoted to state of Newtonian aspects of dedicated models. The association between brain activity and time perception is considered in the section 3. In section 4 other relativistic time perception models are criticized and a novel time perception model based on special relativity theory is presented in section 5. Conclusions are stated in section 6.
2. Dedicated Timing Models and Newtonian Timing System
In a dedicated timing model three structures are important: 1- A pacemaker that regularly pulses as ticks of a clock, 2- An attention (or mode) switch and 3- An accumulator that counts ticks. In a comparison process, the output of these structures is compared with the current time in the working memory [3,4,8,23]. However, the internal clock model has found little evidence in physiology [6].
In some studies a cortico striatal network is assumed as the internal timing system [8,24]. In an fMRI study [8], this cortico-striatal network is investigated and three individual processes are assumed; an oscillatory pacemaker emitting pulses (right caudate nucleus) and an accumulator (pre supplementary motor area), a working memory system as a memory stage for storing current time (bilateral pre motor cortex and inferior prefrontal cortex) and a mechanism that compares the current intervals with those in reference memory (anterior cingulate cortex).
In the Newtonian timing model there is an absolute time measure which is called mathematical time. According to Newton, the mathematical or true time is passed without relation to anything external. This time is a sum of successive pulses and these pulses are occurred regularly and with same intervals [1]. In this framework, the relative time is our perception of the mathematical time which is achieved by changes in moving objects such as moon, sun and the passing of seasons. All of changes in these objects are consequent of mathematical time. Consistent with this theory, same structures is observed in internal clock timing model. The pacemaker in the internal clock model is a form of mathematical time. This clock works regularly and it is not perceived directly.
The relative sense of time is a function of mode or attention switch, accumulator, working memory and decision structures. This perceived time is a consequent of pacemaker ticks (Fig. 1). Thus, from the perspective of the internal clock, the time is an absolute variable. In this framework, subjective time is an estimation of counted ticks. This description of time is such as the relative time in Newtonian timing system. However, in the theory of special relativity, time is a byproduct of movement [1] or information exchange [25]. Thus, in the internal clock framework, relativistic nature of time is completely neglected and time is not relative to velocity. Accordingly, we need to a comprehensive model that is based on true meaning of the time in the physics. Since the Newtonian timing model is a rejected hypothesis in physics, it is suggested that the internal clock model cannot correctly describes actual aspects of the perceived time.

Figure 1. A comparison between mathematical timing system and internal clock model. There is an absolute pulse in both models and the perceived time is a consequent of another structure.
3. The Brain Activity and the Time Perception
3.1. Saccadic Time Compression
In some brain areas such as middle temporal (MT/V5) and magnocellular pathway, a reduced activity is seen during a saccade [26–29]. On the other hand, saccadic eye movement provides a compression in spatiotemporal space [30–32] such that 100ms interval is perceived to half of its true value [22,31]. In some studies the relation between saccadic time compression and the brain activity reduction in MT and magnocellular system is investigated [6,17]. These studies suggest that, time could be encoded by the magnitude of neural activity [5] and more energy spent lead to time dilation [6]. Hence, the time compression during a rapid eye movement could be a consequence of saccadic neural activity reduction [6,17]. Internal clock model does not have an explanation for this phenomenon.
3.2. Repetition Suppression
When a stimulus is presented repeatedly, a reduction in neural activity is occurred [15,33,34]. This reduction involves many cortical areas such as fusiform [15,33], inferior prefrontal cortex [35], inferotemporal cortex [36] and mirror system in inferior parietal lobule [37]. On the other hand, a repeated stimulus is perceived compressed while an oddball stimulus is dilated [6,19,20]. As a result, Eagleman and colleagues suggest that there is a correlation between neural response amplitude and time dilation [38]. According to the internal clock model, this effect is caused by attention. But in some studies it has been under doubt [20].
3.3. Brightness, Size, Flicker and Other Stimulus Properties
Terao et al. suggest that reduction of stimulus visibility could cause a compression in perceived time. This effect is same as a saccadic time compression and it is associated with a reduction in neural activity [17]. On the other hand, the stimulus brightness is a distortion factor in time perception and a brighter stimulus is perceived with dilation [6]. Also duration of bigger stimulus is perceived longer [6] and subjective duration of a flickering stimulus is dilated compared to a non flickering stimulus [39]. All of these time dilations are associated with increasing activity in higher cortical areas. The activity of neurons in the primary visual cortex (V1) is increased when the stimulus is brighter or bigger [18,40]. Moreover, the effect of numerosity, motion, pattern complexity and looming on neural activity and time perception, is similar to the effect of mentioned properties (for a detailed review, see [6]).
Altogether, this section shows that time perception is associated with the activity of several cortical and some subcortical areas. Less activity during saccade and stimulus repetition causes time compression while the more neural activity leads to time dilation.
4. Time Perception and Einstein Special Relativity
4.1. Previous Attempts
Table 1. Investigation of timing models from the standpoint of the special relativity and absolutism. Previous models are not completely consistent with the Einstein special relativity model. The proposed model is based on special relativity theory.
| Timing Model | Absolute time | Perceived time | Time reference |
| Newtonian time | Mathematical time | External measure: An hour, a day, a month and etc. | Does not exist |
| Internal-Clock | Pacemaker | Mode switch, accumulator, working memory and decision structures. | Does not exist |
| Special Relativity | Does not exist | Relative time; via speed of moving observer. | Stationary observer |
| Energy Model | Does not exist | Energy expended during neural activity. | Does not exist |
| SDN Model | Does not exist | Network state and short term plasticity. | Does not exist |
| Multiple Clocks | Three physiological clocks | Mode switch, accumulator, working memory and decision structures. | Does not exist |
| Current Model | Does not exist | Relative time; via firing rate and speed of network development | Stationary activation |
In the intrinsic models, there is no an "absolute time" such as a pacemaker. However, these models are not exactly consistent with theory of special relativity (Table 1). The relativistic nature of time is considered in the recent perceptual timing models [21,22,41]. Buhusi and Meck suggest that physiological time is represented by multiple clocks. They suggest that, in the rats timing system, three clocks are operated independently and a given time interval is perceived differently by these individual clocks. They suggest that three absolute clocks represent a relative time and they claim that this model is consistent with the Einstein relativity theory [41]. But two essential postulate of Einstein is completely ignored. On the other hand, this model is dominated by an absolute time. Since, the time is not considered related to the speed of the light and time is presented by absolute clocks, this dedicated model is a Newtonian timing model (Table 1).
In another relativistic approach, Morrone and collagenous suggest that saccadic compression in time and space is a relativistic effect. But other time distortions during neurophysiological effects such as repetition suppression are not considered from this framework. Furthermore, a fundamental error is occurred in development of the Morrone and her colleagues’ hypothesis. They explain that, a fast remapping occurs during saccade and the velocity of the information exchange is 0.87 speed of light. They write that [21]:
For neural propagations at about 87% maximum speed, objects will be compressed in both space and time by a factor of 2.
This sentence, that is the base of their hypothesis, is completely wrong. According to Einstein special relativity, in moving references, time is dilated while length is contracted [2]. The length of any object in a moving frame is contracted and the amount of contracted length is achieved by Lorentz transformation:
![]() (1)
(1)
where L is the measured length in the stationary reference, L0 is the measured length in the moving reference, v is the velocity of the object and c is the speed of the light in the vacuum [2]. On the other hand, according to the Lorentz transformation, a clock in a moving frame run slowly and time is dilated. The dilated time is achieved by:
![]() (2)
(2)
where t is the elapsed time in the stationary reference, t0 is the elapsed time in the moving reference, v is the velocity of the object and c is the speed of the light in the vacuum [2].
Therefore, if we consider that velocity of remapping during a saccade is in the order of the light speed, then the perceived time will be dilated and it is inconsistent with saccadic time compression. Although saccadic length contraction is correctly explained by Morrone and her colleagues’ hypothesis, but saccadic time contraction is not satisfied by this model. Hence, it is suggested that saccadic time contraction is occurred with a mechanism same as other time distortions. A possible mechanism is proposed in the next section as a novel hypothesis.
4.2. The Brain Activity and the Special Relativity: A New Hypothesis
The theory of special relativity suggests that, time is a byproduct of velocity and it is created during movement, obligatory. Subjective time is perceived during all senses and this perception also is obligatory. I suppose that subjective time is created as a depended sense as well as it is a depended variable in Einstein physics.
In the central nervous system, each neuron is considered as a single-unit processor. Many single-unit processors (neurons) are involved in perception of sensory inputs. An input signal distributes in a neuronal network and it passes from many neurons. For example, in visual perception, there are above 1,000,000 axons between retia and visual cortex that information is transferred through them [42]. Overall total distance that is passed by an input signal is very large. On the other hand, Einstein special relativity suggests that time is not an independent concept rather it is created via motion. It is the key point that indicates time in the brain (as a physical system) is perceived through motion.
The average number 152 of neocortical neurons is approximately 20 billion in the human brain [43]. The white matter occupies 44% of human cortex volume and makes long range cortical connection [44]. Estimations show that information exchange in white matter of human brain is accomplished via approximately 150000−180000 km myelinated nerve fibres [45,46]. On the other hand 60% of the gray matter is composed of neural connections; axons and dendrites [44]. As estimation, in the neocortex of an awake human, the time of neuron firing is between 10ms (origin of gamma frequency) and 100ms (origin of alpha frequency) [47]. An axon in the gray matter of human cortex [48], can convey information signals over distance 1 mm.
The huge number of cortical neurons and long length of the myelinated nerve fibres can be lead to very fast signal distribution. Although there is no accurate estimation to velocity of signal distribution in brain, a simple example can be clarified. As an example, we consider a postman that delivered pieces of a message to many destinations. The complete massage will integrate when all of pieces are delivered. The postman passes too many ways to deliver these message pieces at a given time (Fig. 2). Therefore the velocity of message transfer is equal to distance traveled divided by the time elapsed. In the brain, the postman is like as neural transmission that it distribute neural signal in many neuronal pathways. As indicated in many studies, the time required for data integration is in order of 100 ms (this is in the order of Libet consciousness time) [49–51]. The information is distributed in gray matter network and as estimation, the length that is passed by information is:
![]() (3)
(3)
where N is the number of excited neurons and L is the average length of nerves fibre between two neurons. Moreover, there are over Ltw = 1.5 × 108 m myelinated nerve fibres in the white matter that constitute long-term connections between cortical areas [45,46]. As estimation, in the white matter, the information transfers from the path length that is equal to:
![]() (4)
(4)
where N is the number of activated neurons and N0 is the number of total cortical neurons (N0 ≃ 2×1010). Therefore the total path that is passed by information is:
![]() (5)
(5)
and the velocity of distributed information is:
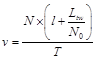 (6)
(6)
where T is the elapsed time for integration. These estimations show that, this velocity is very high and it is near the speed of light. As an example, in the rest state, if 15% of cortical neurons (N0 = 3×109) are in active mode, after T = 100 ms, the velocity of integration is:
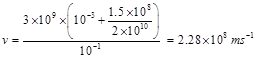 (7)
(7)
this is approximately 0.76 of light speed (0.76 × c). However, since this activity is the base activity of the cortex in the rest state, it can be ignored. On the other words, for a stationary observer (brain state) in this system v = 0m and it is a stationary reference in the special relativity. Therefore a clock measures 100ms in this reference as indicated in Fig 3. When, the activity increases during a task, the velocity is increased and the velocity difference is equal to:
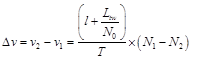 (8)
(8)
where N2 is the number of neurons in more activity state (state 2) and N1 is the number of neurons in the rest state (state 1). In this equation, since the state 1 is the stationary reference, composition law for velocities is not used [2]. If the number of activated neurons increase to 25% of total cortical neurons (10% increase relative to the rest state), the velocity difference is:
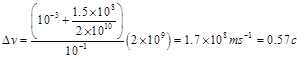 (9)
(9)
In the state 2, the velocity of integration is same as velocity of moving reference in special relativity and according to theory of relativity, if a clock moves nearly the light speed, then time obviously passes slowly and time dilation is occurred (Fig. 2). Thus, according to Lorentz equation (Eq. 2), 100ms in the moving reference is dilated and it is equal to:
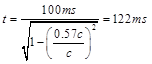 (10)
(10)
in the stationary reference. Einstein suggests that multiple clocks are created during two or several references that are moving relative to each other [2]. In the cortex there are many moving references that their velocities are different from stationary or rest state of brain. The duration of 100ms in the moving state is seen to 122ms from the viewpoint of an observer in the stationary state. On the other words, when activation of the brain is increased as 10% total cortical neurons, then 100ms time is dilated for activated cortex (moving reference) and this time is measured 122ms, as a viewpoint of an observer in stationary reference (rest state of brain).
Although the synaptic velocity is a classic speed (in order to 1 m/s) but since each of the neurons participate to the perception and an input signal passes through a huge number of neurons in the fraction of a second, it can be said that signal is distributed in the processing network with relativistic velocity (as estimated above). This relativistic speed cause to time distortion. 1 second is always absolute when it is measured by a stationary external clock. But this time is distorted when it is measured by the brain. I suggest that, this distortion is a 206 consequent of the special relativity effect and very high velocity of integration in the cortex.
Calculations show that, when the activity of the cortex is 10% more than activity of the cortex during the rest state, then a time dilation on the order of 25% actual duration is occurred. It is in consistent with time dilation for oddball stimulus when it is represented between repetitive stimuli. Also other time dilations during more brain activation (such as time dilation for brighter, bigger and complex stimuli) are in consistent with this relativistic approach. On the other words, more activation leads to more velocity of integration and since this velocity is near the speed of light, a relativistic time dilation is occurred. During a saccade, the activation of cortex is reduced and the velocity is decreased. According to special relativity theory a time frame in the less speed, is compressed. Since a 100ms interval is perceived approximately 50ms, during a saccade [31], the velocity most be reduced equal to 0.85c. Thus, the Eq. 8 predicts that, approximately 3 × 109 neuron is inhibited during saccade. This is in consistent with experimental observations [26–29].
Thus, in the mammalian’s brain, the relativistic speed is accessible and also higher speed is imaginable. Theory of relativity describes that higher speed of light is unavailable. This theory suggests that, in higher speed of light, time arrow is reversed and causality is violated. But memory is a good example for violation in causality. We can transfer in time with memory without any problem. As a suggestion the mentioned hypothesis suggests that brain can be considered as a "Time Machine" and memory is a tool for transferring in time. Although this prediction requires more investigation in experimental studies.
5. Conclusions
In this paper a novel time perception model based on the theory of special relativity is presented. This hypothesis estimate that, velocity of integration in the corticocortical paths may be close to the speed of light (eq. 7). Thus, relativity of subjective time is related to this speed. When the activity of cortex is increased, the velocity of information exchange and integration is increased (in order of the speed of light) and based on the theory of special relativity, the perceived time is dilated. On the other hand, when the activity is less than the rest state (for example: during a saccade), the integration speed is reduced (in order of the light speed) and perceived time is compressed. This model is consistent with this fact that time is perceived as a depended sense with other senses and respects to relativistic essence of time. Moreover many physiological and psychological observations in time perception such as time compression during saccade or repetition suppression and time dilation for brighter, bigger and complex stimuli are explained by this approach. Also this hypothesis suggests that brain can be considered as a "Time Machine". However, more investigation by specific evidences is required.

Figure 2. A postman that delivered pieces of a message to many destinations. He traveled a great distance at a given time and the velocity of message transfer is equal to distance traveled divided by the time elapsed.
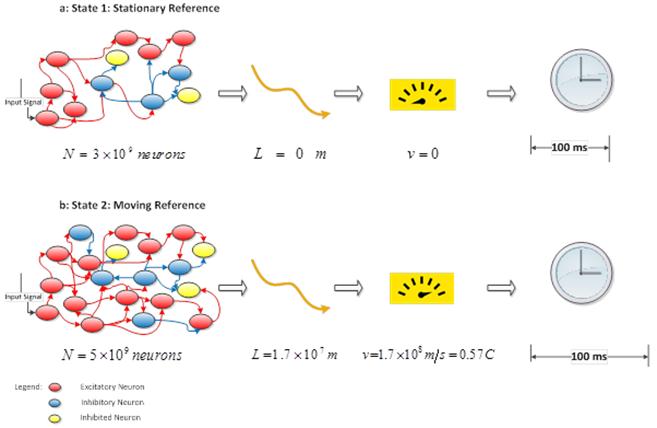
Figure 3. Time dilation during more activity of the cortex, a: in the rest state, velocity of an observer is equal to 0. Thus time is not distorted, b: when the cortical activity is increased as 10% of total capacity, observer (brain state) leaves the stationary state and its velocity is 0.57c. In this moving reference, the time is dilated.
References
- Newton, I. Sir Isaac Newton's mathematical principles of natural philosophy and his system of the world. (University of California Press, Berkeley, 1934).
- Einstein, A. (1920). Relativity: The Special and the General Theory, (trans. Lawson, R. W.) (Methuen, London, 1920).
- Treisman, M. Temporal discrimination and the indifference interval: Implications for a model of the" internal clock". Psychological Monographs: General and Applied 77, 1-31 (1963).
- Treisman, M., Faulkner, A., Naish, P. L., Brogan, D. The internal clock: Evidence for a temporal oscillator underlying time perception with some estimates of its characteristic frequency. Perception 19, 705-743 (1990).
- Ivry, R. B., Schlerf, J. E. Dedicated and intrinsic models of time perception. Trends in cognitive sciences 12, 273-280(2008).
- Eagleman, D. M., Pariyadath, V. Is subjective duration a signature of coding efficiency?. Philosophical Transactions of the Royal Society B: Biological Sciences, 364, 1841-1851 (2009).
- Allman, M. J., Meck, W. H. Pathophysiological distortions in time perception and timed performance. Brain 135, 656-677 (2012).
- Pouthas, V. et al. Neural network involved in time perception: an fMRI study comparing long and short interval estimation. Human brain mapping 25, 433-441 (2005).
- Sadeghi, N. G., Pariyadath, V., Apte, S., Eagleman, D. M., Cook, E. P. Neural correlates of subsecond time distortion in the middle temporal area of visual cortex. Journal of cognitive neuroscience 23, 3829-3840 (2011).
- Craig, A. D. How do you feel now? the anterior insula and human awareness. Nature Reviews Neuroscience 10, 59-70 (2009).
- Wittmann, M. The inner sense of time: how the brain creates a representation of duration. Nature Reviews Neuroscience 14, 217-223 (2013).
- Karmarkar, U. R., Buonomano, D. V. Timing in the absence of clocks: encoding time in neural network states. Neuron 53, 427-438 (2007).
- Buonomano, D. V., Maass, W. State-dependent computations: spatiotemporal processing in cortical networks. Nature Reviews Neuroscience 10, 113-125 (2009).
- Wittmann, M. The inner experience of time. Philosophical Transactions of the Royal Society B: Biological Sciences 364, 1955-1967 (2009).
- Henson, R. N. A., Rugg, M. D. Neural response suppression, haemodynamic repetition effects, and behavioural priming. Neuropsychologia 41, 263-270 (2003).
- Summerfield, C., Trittschuh, E. H., Monti, J. M., Mesulam, M. M., Egner, T. Neural repetition suppression reects fulfilled perceptual expectations. Nature neuroscience 11, 1004-1006 (2008).
- Terao, M., Watanabe, J., Yagi, A., Nishida, S. Y. Reduction of stimulus visibility compresses apparent time intervals. Nature neuroscience 11, 541-542 (2008).
- Kinoshita, M., Komatsu, H. Neural representation of the luminance and brightness of a uniform surface in the macaque primary visual cortex. Journal of Neurophysiology 86, 2559-2570 (2001).
- Eagleman, D. M. Human time perception and its illusions. Current opinion in neurobiology 18, 131-136 (2008).
- Pariyadath, V., Eagleman, D. The effect of predictability on subjective duration. Plos one 2, e1264 (2007).
- Morrone, M. C., Ross, J., Burr, D. C. in Space and time in perception and action (eds Nijhawan, R., Khurana, B.) 52-62 (Cambridge University Press, New York 2010).
- Burr, D. C., Ross, J., Binda, 276 P., Morrone, M. C. Saccades compress space, time and number. Trends in cognitive sciences 14, 528-533 (2010).
- Allman, M. J., Teki, S., Griffiths, T. D., Meck, W. H. Properties of the internal clock: first-and second-order principles of subjective time. Annual review of psychology 65, 743-771 (2014).
- Meck, W. H., Penney, T. B., Pouthas, V. Cortico-striatal representation of time in animals and humans. Current opinion in neurobiology 18, 145-152 (2008).
- Capria, M. M., Superluminal Waves and Objects: Theory and Experiments. A Panoramic. Physics Before and After Einstein, 267 (2005).
- Burr, D. C., Morrone, M. C., Ross, J. Selective suppression of the magnocellular visual pathway during saccadic eye movements. Nature 371, 511-513 (1994).
- Ross, J., Burr, D., Morrone, C. Suppression of the magnocellular pathway during saccades. Behavioural brain research 80, 1-8 (1996).
- Thiele, A., Henning, P., Kubischik, M., Hoffmann, K. P. Neural mechanisms of saccadic suppression. Science 295, 2460-2462 (2002).
- Kleiser, R., Seitz, R. J., Krekelberg, B. Neural correlates of saccadic suppression in humans. Current Biology 14, 386-390 (2004).
- Yarrow, K., Haggard, P., Heal, R., Brown, P., Rothwell, J. C. Illusory perceptions of space and time preserve cross-saccadic perceptual continuity. Nature 414, 302-305 (2001).
- Morrone, M. C., Ross, J., Burr, D. Saccadic eye movements cause compression of time as well as space. Nature neuroscience 8, 950-954 (2005).
- Eagleman, D. M. Distortions of time during rapid eye movements. Nature neuroscience 8, 850-851 (2005).
- Henson, R., Shallice, T., Dolan, R. Neuroimaging evidence for dissociable forms of repetition priming. Science 287, 1269-1272 (2000).
- Dobbins, I. G., Schnyer, D. M., Verfaellie, M., Schacter, D. L. Cortical activity reductions during repetition priming can result from rapid response learning. Nature 428, 316-319 (2004).
- Wagner, A. D., Desmond, J. E., Demb, J. B., Glover, G. H., Gabrieli, J. D. Semantic repetition priming for verbal and pictorial knowledge: A functional MRI study of left inferior prefrontal cortex. Journal of Cognitive Neuroscience 9, 714-726 (1997).
- McMahon, D. B., Olson, C. R. Repetition suppression in monkey inferotemporal cortex: relation to behavioral priming. Journal of neurophysiology 97, 3532-3543 (2007).
- Chong, T. T. J., Cunnington, R., Williams, M. A., Kanwisher, N., Mattingley, J. B. fMRI adaptation reveals mirror neurons in human inferior parietal cortex. Current biology 18, 1576-1580 (2008).
- Pariyadath, V., Eagleman, D. M. Subjective duration distortions mirror neural repetition suppression. Plos one 7, e49362 (2012).
- Kanai, R., Paffen, C. L., Hogendoorn, H., Verstraten, F. A. Time dilation in dynamic visual display. Journal of Vision 6, 1421-1430 (2006).
- Murray, S. O., Boyaci, H., Kersten, D. The representation of perceived angular size in human primary visual cortex. Nature neuroscience 9, 429-434 (2006).
- Buhusi, C. V., Meck, W. H. Relativity theory and time perception: single or multiple clocks?. Plos one 4, e6268 (2009).
- Kropotov, J. Quantitative EEG, event-related potentials and neurotherapy. (Academic Press, 2010).
- Pakkenberg, B., Gundersen, H. J. G. Neocortical neuron number in humans: e_ect of sex and age. Journal of ComparativeNeurology 384, 312-320 (1997).
- Laughlin, S. B., Sejnowski, T. J. Communication in neuronal networks. Science 301, 1870-1874 (2003).
- Pakken319 berg, B. et al. Aging and the human neocortex. Experimental gerontology 38, 95-99 (2003).
- Marner, L., Nyengaard, J. R., Tang, Y., Pakkenberg, B. Marked loss of myelinated nerve fibers in the human brain with age. Journal of Comparative Neurology 462, 144-152 (2003).
- Jensen, O., Kaiser, J., Lachaux, J. P. Human gamma-frequency oscillations associated with attention and memory. Trends in neurosciences 30, 317-324 (2007).
- Kandel, E., Barres, B., Hudspeth A. J. in Principles of Neural Science, (eds Kandel, E., Schwartz, J., Jessell, T., Siegelbaum, S., Hudspeth A. J.) 21-38 (McGraw Hill Professional, New York, 2013)
- Wittmann, M. Moments in time. Frontiers in integrative neuroscience 5, 66 (2011).
- Repp, B. H. On the nature of phase attraction in sensorimotor synchronization with interleaved auditory sequences. Human Movement Science 23(3), 389-413 (2004).
- Giraud, A. L., Kleinschmidt, A., Poeppel, D., Lund, T. E., Frackowiak, R. S., Laufs, H. Endogenous cortical rhythms determine cerebral specialization for speech perception and production. Neuron 56(6), 1127-1134 (2007)



