Parametric and Experimental Study of Desulfurization Process from Valuable Fuels
Mohammad Amin Seifi1, Farshad Farahbod2, *
1Department of Chemical Engineering, Shahreza Branch, Islamic Azad University, Shahreza, Iran
2Department of Chemical Engineering, Firoozabad Branch, Islamic Azad University, Firoozabad, Iran
Abstract
Today, in the young field of nanotechnology (sometimes shortened to "nanotech"), scientists and engineers are taking control of atoms and molecules individually, manipulating them and putting them to use with an extraordinary degree of precision. Nano catalytic sulphur removal process is surveyed in this paper, experimentally. The qualification of sulphur removal from liquefied natural gas process is investigated due to catalytic bed geometries which contains nano aluminium and copper oxide and operating conditions. The purpose of experiments is finding the conditions which lead to the lowest amount of sulphur content in the out stream.
Keywords
Operating Pressure, Operating Temperature, Bed, Dimensionless Group, Performance
Received: June 1, 2016
Accepted: June 13, 2016
Published online: July 21, 2016
@ 2016 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY license. http://creativecommons.org/licenses/by/4.0/
1. Introduction
Today, in the young field of nanotechnology (sometimes shortened to "nanotech"), scientists and engineers are taking control of atoms and molecules individually, manipulating them and putting them to use with an extraordinary degree of precision. Word of the promise of nanotechnology is spreading rapidly, and the air is thick with news of nanotech breakthroughs. Governments and businesses are investing billions of dollars in nanotechnology R&D, and political alliances and battle lines are starting to form. Nanotechnology is the manipulation of matter on an atomic and molecular scale. The earliest, widespread description of nanotechnologyhas been referred to the particular technological goal of precisely manipulating atoms and molecules for fabrication of macro scale products, also now is referred to as molecular nanotechnology [1]. A more generalized description of nanotechnology was subsequently established by the National Nanotechnology Initiative, which defines nanotechnology as the study and application of fine particles which are sized from 1 to 100 nanometres in all of the science fields [2].
Sulphur compounds in fuels such as liquefied petroleum gas cause problems on two fronts: they release toxic gases during combustion process, and they damage metals and catalysts in engines and fuel cells [3]. They usually are removed using a liquid treatment that adsorbs the sulphur compounds from the liquefied petroleum gas, but the process is cumber some and requires that the hydrocarbon be cooled and reheated, making the fuel less energy efficient [4]. To solve these problems, researchers have turned to solid metal oxide adsorbents, but those have their own sets of challenges [5]. While they work at high temperatures, eliminating the need to cool and re-heat the fuel, their performance is limited by stability issues. They lose their activity after only a few cycles of use [6].
The sulphur compounds and sulphurs in hydrocarbon and petroleum cuts lead to environmental pollution and corrosion problems in pipelines and storage tanks. So it is necessary to decrease sulphur and sulphurs concentrations in hydrocarbon cuts to international standard levels in petroleum industries. For this purpose, DMD process has been developed as a desulphurization process providing the possibility of desulphurization of different hydrocarbon cuts even crude oil as a feed.
Previous studies found that sulphur compounds adsorption works best at the surface of solid metal oxides [7]. So, the authors set out to create a material with maximum surface area. The solution seems to be tiny grains of copper and aluminium oxide nano particles, uniting high surface area, high reactivity and structural integrity in a high-performance sulphur adsorbent [8]. Aluminium oxide has been numerously used for removing of sulphide compounds from liquefied petroleum gas streams in processes like reforming [9,10], integrated gasification combined cycle [11] and fuel cell [11,12,13]. Although, ![]() has been well evaluated with sulphide compounds feed stocks, the performance of aluminium and copper oxide nano structure with different operating conditions and structural characteristics in sulphur removal has not been specially evaluated in details. This work is devoted to using experimental design methodology to identify the optimum conditions for sulphur removal by nano copper and aluminium oxide catalysts. Clearly, the nano-sized
has been well evaluated with sulphide compounds feed stocks, the performance of aluminium and copper oxide nano structure with different operating conditions and structural characteristics in sulphur removal has not been specially evaluated in details. This work is devoted to using experimental design methodology to identify the optimum conditions for sulphur removal by nano copper and aluminium oxide catalysts. Clearly, the nano-sized ![]() is more reactive than the same material in bulk form, enabling complete sulphur removal with less material, allowing for a smaller reactor. The nano particles stay stable and active after several cycles [10].
is more reactive than the same material in bulk form, enabling complete sulphur removal with less material, allowing for a smaller reactor. The nano particles stay stable and active after several cycles [10].
Thermal Swing Regeneration is a common industry process used for desulphurization process. In that process, chemical sponges called sorbents remove toxic and flammable gases, such as rotten-egg smelling hydrogen sulphide from natural gas.
The liquefied petroleum gas must first be treated with a solution of chemical sorbents that are dissolved in water. That solution must then be heated up and boiled to remove the sulphide compounds, in order to prepare the sorbent for future use. Once the sulphide compounds are boiled off, the sorbent is then cooled and ready for use again. The repeated heating and cooling requires a lot of energy and markedly reduces the efficiency of the process, scientists say.
In the adsorption process by nano aluminium and copper oxide, sweetening of liquefied petroleum gas is occurred with minimum heat flux comparing with the other sweetening methods. Also, approximately, 70 to 80 percentage of the initial amount of sulphur is removed from the liquefied petroleum gas by the proposed adsorption process. Also, aluminium and copper oxide catalyst is produced due to feasible method and is not expensive comparing with the other catalysts. So, this method is beneficial. Undoubtedly, the aluminium and copper oxide nano particles as sorbents have large active surface. So, they can be reused again and again. This method will be developed as soon as possible and will be applied in the industrial scale.
In this work, a fixed bed reactor is set up which is equipped by nano aluminium and copper oxide catalysts. Some experiments have been held to investigate the effect of different operating pressure, temperature, catalyst diameter, bed height on the performance of sulphur removal. Also, the capability of nano catalysts is surveyed toward changing the amount of sulphur in the feed stream and also changing feed superficial velocity. The results are illustrated as the ratio of outlet sulphur concentration per inlet sulphur concentration. In addition, this work contains the cost estimations for the various operating pressures and temperatures. Consequently the optimum conditions are introduced.
2. Materials and Methods
2.1. Synthesis Method of Nano-sized Al2O3
The amount of 1.16 g of Aluminium oxide in powder form is dissolved in de-ionized water contains 0.38 g of citric acid crystals. Then Ammonium hydroxide is added gradually (till obtaining pH=7) into the mixture and is homogenized effectively in magnetic stirrer simultaneously.
2.2. Synthesis Method of Nano-sized CuO
CuO nanoparticles were synthesized by aqueous precipitation method using 55 g of copper sulphate 5-hydrate (Merck) mixed with 25 g of hydroxyl ammonium chloride (Sigma Aldrich) in 125 ml distilled water.
2.2.1. Characterization of Nanoparticles
The XRD patterns of nano CuO were recorded on a (BRUKER AXS) diffractometer in the scanning range of 20-70° (2θ) using CuKα as radiation source having a wavelength of 1.54060 A° at the scanning rate of 15.50 s with the instrument temperature of 25°C. The morphology of prepared NPs before and after adsorption was examined by SEM technique. SEM images were taken by Joel Quick Auto Coater and by using ion sputtering device.
2.2.2. The Average Size of Nanoparticles
The average size of nano particles which is used in this paper is 60nm, approximately. The colour of produced nano particles is white. The 60nm as average size of metal oxide can improve the mass transfer specific area.
2.3. Set up Description
One laboratory cylindrical vessel equipped with the nano-sized ![]() and CuO catalytic fixed bed is applied for sulphur adsorption process, in this work. The process temperature is adjusted by one steam jacket around the vessel. Liquefied petroleum gas stream from a tank reservoir is mixed by sulphur and is fed into the bed containing aluminium and copper oxide (50%-50%) nano particles.
and CuO catalytic fixed bed is applied for sulphur adsorption process, in this work. The process temperature is adjusted by one steam jacket around the vessel. Liquefied petroleum gas stream from a tank reservoir is mixed by sulphur and is fed into the bed containing aluminium and copper oxide (50%-50%) nano particles.
3. Results and Discussion
Anyone knows, the sulphide compound is corrosive and toxic, severely. Meanwhile, this component is in several industrial. We know the current technologies use huge resources of energy for removing the sulphur component. Therefore, the researchers try to enhance the performance of sweetening process. So, in this paper the aluminium and copper oxide are applied as nano catalysts for removal of sulphur. These metal oxides are available and also, are not expensive comparing with the other metal oxides. Several experiments were held to determine operational conditions that would optimize the amount of sulphur removed from gas in order to gas sweetening.
Some major parameters are considered experimentally in the gas desulphurization process by nano particles. The effects of operating conditions, properties of catalytic bed and copper and aluminium oxide catalyst are investigated on the process performance. The ratio of sulphur concentration in the product stream on the initial concentration in the input stream (![]() ) represents the process performance. The purpose of the experiments is to decrease the amount of sulphide compounds below the 4 ppm in the outlet stream. Experimental results are presented in the following Figures.
) represents the process performance. The purpose of the experiments is to decrease the amount of sulphide compounds below the 4 ppm in the outlet stream. Experimental results are presented in the following Figures.
3.1. The Effect of Operating Temperature and Pressure
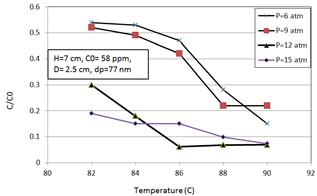
Figure 1. The effect of operating temperature and pressure on the quality index.
The increase in the amount of operating pressure from 6atm to 9atm at temperatures of 82C to 88C decreases the amount of C/C0. For higher amounts of pressure from 12atm to 15atm the increase in the amount of temperature from 86C to 90C decreases the amount of C/C0. The strange behavior in the decreasing trend of C/C0 is obtained at temperature of 90C which the higher amount of C/C0 is obtained at 9atm comparing with 6 atm. Also, the equal amounts of C/C0 are obtained at 12atm and 15atm and at 90C. The amounts of concentrations which are obtained at 15atm are higher than those are obtained at pressure of 12atm in ranges of temperatures from 86C to 90C. The Figure 1 shows the changes in the amount of C/C0 due to the changes in temperatures and pressures. Results indicate the optimum amount of pressure and temperature is 12atm and 66C which leads to the amount of 0.061 ppm.
3.2. The Effect of Bed Characteristics
The height and diameter of catalytic bed and also catalyst diameter are considered as the shape effects in this section. Bed length is changed in the range of 6, 7 and 9cm. Also the bed diameter changes are in range of 1.25, 1.8 and 2.5cm. The effect of bed characteristics are investigated on the amount of sulphur removal. The optimum amounts of height and diameter of the nano catalytic bed are studied in this section. Operating temperature and pressure is adjusted at 86°C and 12![]() , respectively.
, respectively.
3.3. The Effect of Bed Diameter
The effect of changes in diameter of bed on sulphur removal is shown in the Figure 2. The increase in value of bed diameter provides higher mass transfer area and also leads higher rate of mass transfer. So, the decrease trend is obtained with higher values of bed diameter. The diameter changes from 1.25cm to 2.5cm and the ratio of C/C0 changes from 0.048 to 0.04.
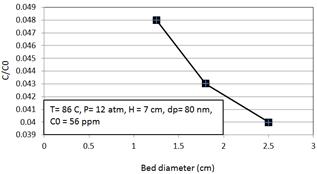
Figure 2. The effect of bed diameter on the amount of C/C0.
So, the increase of 50% in the amount of bed diameter improves the amount of C/C0 about 0.16. Bed height values are 6cm, 7cm and 9cm.
4. Conclusions
Nano catalyst development in various areas proposes to perform many processes economically and efficiently. The optimum operating conditions and reactor characteristics for sulphur removal with aluminium and copper oxide nano catalyst are investigated experimentally in this work. The process performance is introduced as the ratio of the outlet concentration of sulphur per the inlet concentration and is presented as value of ![]() . Experiments are done in the cylindrical reactor in different temperature, pressure, bed height, bed diameter and copper and aluminium oxide catalyst diameter to find the best condition to reach the
. Experiments are done in the cylindrical reactor in different temperature, pressure, bed height, bed diameter and copper and aluminium oxide catalyst diameter to find the best condition to reach the ![]() value of 0.04 for sulphur at the product.
value of 0.04 for sulphur at the product.
References
- Yuxiao Niu, Mingyang Xing, Baozhu Tian, Jinlong Zhang, 2012,"Improving the visible light photocatalytic activity of nano-sized titanium dioxide via the synergistic effects between sulfur doping and sulfation,’’Applied Catalysis B: Environmen., 115–116 (5) pp. 253-260.
- Corrie L.C., Kenneth J.K., 2002,"Unique Chemical Reactivities of Nanocrystalline Metal Oxides toward Hydrogen Sulfide,’’ Chem. Mater., 14 (4) pp. 1806-1811.
- Rao Mumin, Song Xiangyun, Cairns Elton J., 2012,"Nano-carbon/sulfur composite cathode materials with carbon nanofiber as electrical conductor for advanced secondary lithium/sulfur cells,’’ J. Power Source., 205 (1), pp. 474-478.
- Zhang Yongguang, Zhao Yan, Konarov Aishuak, Gosselink Denise, Soboleski Hayden Greentree, Chen P., 2013,"A novel nano-sulfur/polypyrrole/graphene nanocomposite cathode with a dual-layered structure for lithium rechargeable batteries,’’ J. Power Source., 241 (1), pp. 517-521.
- Hosseinkhani M., Montazer M., Eskandarnejad S., Rahimi M.K., 2012,"Simultaneous in situ synthesis of nano silver and wool fiber fineness enhancement using sulphur based reducing agents,"Colloids and Surfaces A: Physicochem. Eng. Aspect., 415 (5), pp. 431-438.
- Christoforidis Konstantinos C., Figueroa Santiago J.A., Fernández-García Marcos, 2012,"Iron–sulfur codoped TiO2 anatase nano-materials: UV and sunlight activity for toluene degradation,"Applied Catalysis B: Environment., 117–118 (18), pp. 310-316.
- Balouria Vishal, Kumar Arvind, Samanta S., Singh A., Debnath A.K., Mahajan Aman, Bedi R.K., Aswal D.K., Gupta S.K., 2013,"Nano-crystalline Fe2O3 thin films for ppm level detection of SULPHUR,’’Sensors Actuators B: Chemical, 181, pp. 471-478.
- Eow, D., John, S.,2002,"Recovery of sulfur from sour acid gas: A review of the technology Environmental Progress,’’ American Institute Chem. Eng., 21, pp. 143-162.
- Habibi R., Rashidi A. M., Towfighi Daryan J., Alizadeh A., 2010,"study of the rod –like and spherical nano Fe2O3 morphology on Sulphur removal from natural gas". Appl. Surf. Sci., 257, pp. 434-439.
- Novochimskii I.I., Song CH., Ma X., Liu X., Shore L., Lampert J., Farrauto R. J., 2004, "Low temperature SULPHUR removal from steam containing gas mixtures withFe2O3for fuel cell application. 2. wash- coated monolith". Ene. Fuel., 18, pp. 584-589.
- Novochimskii II., Song CH., Ma X., Liu X., Shore L., Lampert J., Farrauto R. J., 2004, "Low temperature SULPHUR removal from steam containing gas mixtures withFe2O3for fuel cell application. 1.Fe2O3particles and extrudates". Ene. Fuel., 18, pp. 576-583.
- Arthour L.K., Richard B., 1997, "Gas purification", Nielsen edition.
- Habibi R., Towfighi Daryan J., Rashidi A.M., 2009, Shape and size-controlled fabrication of Fe2O3 nanostructures using noveltemplates, J. Exp. Nanosci. 4 (1) 35-45.
- Farahbod Farshad, Bagheri Narges, Madadpour Fereshteh, Effect of Solution Content Fe2O3 Nanoparticles on Thermal Stability of Poly Vinyl Chloride, Journal of Nanotechnology in Engineerin and Medicine, DOI: 10.1115/1.4025209, In press.



