Observations of the Effect of Nano Chitosan Against the Locust Schistocerca gregaria (Orthoptera: Acrididae)
Sabbour M. M.*
Department of Pests and Plant Protection, Dep. National Research Centre, Dokki, Cairo, Egypt
Abstract
The effect of Chitosan and nano Chitosan tested on the target insect pest Schistocerca gregaria. Results obtained showed that, under laboratory conditions, the LC50of the newly hatched is recorded, 278, 244 233, 247 and 241 ppm for newly hatched, nymphs, Last nymphal stage Adult ♀ and Adult ♂ respectively. Also, when S. gregaria treated with nano- Chitosan, the LC50s obtained 268,204,213,231, and 132 ppm for Newly hatched, nymph, Last nymphal stage, Adult ♀ and Adult ♂, respectively after treated with nano-Chitosan. Also, results recorded that, the Number of egg laid/femal±SE recorded, 158±2.2 and 88±0.01 eggs / female after treated with Chitosan and nano- Chitosanas compared to 258±3.11 eggs / female in the control (Table 2). The % of Adult ♀ and % of Adult ♂ significantly decreased to 18 and 10 after nano-Chitosantreatments as compared to 99 and 99% in the control. Our results showed, under semi field conditions, the number of S. gregaria were significantly decreased after the Chitosanand nano-Chitosantreatment, the number of infestations with S. gregaria decreased to 29±3.6 and 8±1.1 individuals after120 days of treatments.
Keywords
Locust, Schistocerca gregaria, Chitosan, Nano
Received:January 29, 2016
Accepted: February 19, 2016
Published online: August 19, 2016
@ 2016 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY license. http://creativecommons.org/licenses/by/4.0/
1. Introduction
Chitosan (CS)-g-poly (acrylic acid) (PAA) nanoparticles, which are well dispersed and stable in aqueous solution have been prepared by template polymerization of acrylic acid in chitosan solution. The prepared CS-PAA had a white powder shape and was insoluble in water and diluted acid [1]. The mean particles size were found to be around 50nm. FTIR spectra of CS-PAA nanoparticles for CS, the intensities of the amide band were observed clearly. The board peak appeared at 2500cm-1, which confirmed the presence of NH3 +in the CS-PAA nanoparticles. Nanoparticles synthesis is currently intensively researched due to its wide variety of potential applications [2]. As an alternative to chemical manufactured pesticides, use of nanoparticles as an antimicrobial agents has become more common as technological advances have made their production more economical [3, 4]. Numerous studies on the antimicrobial activity of chitosan and its derivatives against most economic plant pathogens have been investigated [5, 6, 7, 8]. Therefore, these compounds are considered as useful pesticides in the control of plant diseases.
The locust, S. gregaria is considered one of the most harmful pests to different cultivated crops in Egypt. Its economic importance comes from attacking many cultivated crops, vegetables and even trees, feeding on it and causing great losses in quantity and quality of the attacked crop. In some cases, thousands of cultivated hectares may be attacked by the swarms of grasshoppers leaving it as a divested desert. The economic injury of S. gregaria in Egypt had been documented by [9]. Egypt, from 4500 years ago [10, 11, 12]. More than 300 species of locusts and grasshoppers are known to exist in the African continent FAO (1995), but fortunately only a few of them are major pests; most are sedentary, inhabiting a rather confined area throughout their life cycle. [13,14] showed that range of mortality was between 64-85% based on the end point data after using the bioinsecticdes and the nano materials against the locust under laboratory and semifield conditions.
The aim of this work is to evaluate the nano chitosan against the desert locust S. gregaria under laboratory and semi field conditions.
2. Materials and Methods
2.1. Materials
The raw materials used for ion exchange preparation are:
* The Chitosan(CS) used in this study was purchased from HAS HMRZEL laboratories LTD (Netherlands)
* The acrylic acid was purchased from Sd. Finc. Chem. Limited (Laboratory grade for synthesis) and was freshly distilled under reduced pressure to eliminate any inhibitors.
* Both initiators (ammonium persulfate and sodium bisulfite) and other chemicals were of analytical reagent and used as received
2.2. Insect Rearing
S. gregaria was reared under laboratory condition for several generations on semi-artificial diet as mentioned by [10].
2.3. Preparation of Nano- Chitosan
Chitosan Nanoparticles were synthesized by h y- drolyzing titanium tetra isopropoxide in a mixture of 1:1 anhydrous ethanol and water. 9 ml of titanium tetra isopropoxide is mixed with 41ml of anhydrous ethanol (A). 1:1 ethanol and water mixture is prepared. (B) Solution A is added in drop wise to solute ion B and stirred vigorously for 2hrs. At room temperature hydrolysis and condensation are performed, using 1M sulphuric acid and stirred for 2 hrs. Then the ageing was undertaken for 12hrs. The gel was transferred into an autoclave and tightly closed, and the mixture was subjected to hydrothermal treatment at 353K for 24hrs. After filtration the solid residue was washed thoroughly with water and ethanol mixture, dried at 373K in an oven and calcined at 773K.
2.4. Nanoencapsulation
The Nanoencapsulation is a process through which a chemical is slowly but efficiently released to the particular host for insect pests control. "Release mechanisms include dissolution, biodegradation, diffusion and osmotic pressure with specific pH" [15]. Encapsulated of the Chitosan nano-emulsion is prepared by high-pressure homogenization of 2.5% surfactant and 100% glycerol, to create stable droplets which that that increase the retention of the oil and cause a slow release of the nano materials. The release rate depends upon the protection time; consequently a decrease in release rate can prolong insect pests protection time [16].
2.5. Efficacy of Chitosan Against the Target Insect Pests
The insecticide Chitosan were tested at the 6 concentrations: 6 mg, 5mg, 4mg, 3mg, 2mg, 1mg. The insecticide, prepared 6 concentrations (prepared according [17] Percentages of mortality were calculated according to Abbott’s formula [18], while the LC50 values was calculated throughout probit analysis [19]. The experiment was carried out under laboratory conditions at 26±2°C and 60-70% RH
2.6. Statistical Analysis
Data obtained was statistical analysed using Duncan's multiple range test according to [20].
Efficacy of tested nano- Chitosan applied alone on the mean number of deposited eggs of target insects for conducting the combination tests with Chitosan (0.5 g/kg of grains). The Chitosan alone were used at rate (1.0 g/kg) of grains. Four replicates of 100 g grains for each treatment were used. Each replicate was treated individually with treatments and then shaken manually for 1 min to achieve equal distribution of the dust in the entire formulation quantity and was placed in glass jar. Four replicates jar containing untreated grain served as control. Subsequently, one paired of newly emerged adults were introduced into each jar. The number of deposited eggs on treated or untreated grains/female was counted. The data was analyzed using analysis of variance (ANOVA), where significant differences between the treatments were observed. Mean values were significantly separated by using the least significant difference (LSD) test at 5% level [21].
3. Results and Discussion
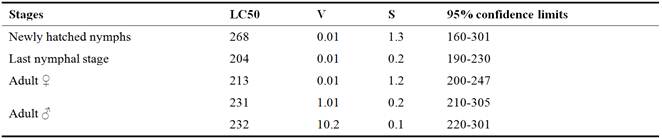
Table 1 show the effect of Chitosanon the target insect pest S. gregaria under laboratory conditions, the LC50 of the newly hatched is recorded, 278, 244 233, 247 and 241 ppm for newly hatched, nymphs, Last nymphal stage Adult ♀ and Adult ♂ respectively.
Table 1. Effect of Chitosan against Schistocerca gregaria under laboratory conditions.
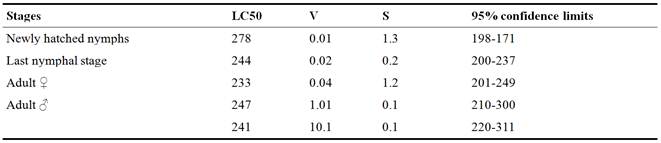
Table 2, show that the under laboratory conditions, the LC50s of locust S. gregaria obtained 268,204,213,231, and 132 ppm for Newly hatched, nymph, Last nymphal stage, Adult ♀ and Adult ♂, respectively after treated with nano-Chitosan (Table 2).
Table 2. Effect of nano-Chitosan against Schistocerca gregaria under laboratory conditions.
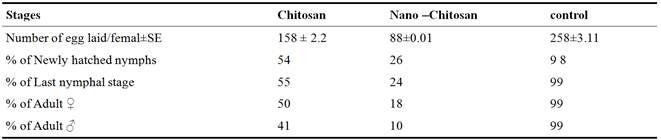
Under laboratory conditions, the Number of egg laid/femal±SE recorded, 158 ± 2.2 and 88±0.01 eggs/ female after treated with Chitosan and nano- Chitosanas compared to 258±3.11 eggs/ female in the control (Table 2). The % of Adult ♀ and % of Adult ♂ significantly decreased to 18 and 10 after nano-Chitosantreatments as compared to 99 and 99% in the control (Table 3).
Table 3. Effect of Chitosanand nano-Chitosan against Schistocerca gregaria under laboratory conditions.
Also, under semi field conditions, the number of S. gregaria were significantly decreased after the Chitosanand nano-Chitosantreatment, the number of infestations with S. gregaria decreased to 29 ±3.6 and 8 ±1.1 individuals after 20 days of treatments (Table 4).
Table 4. Effect of Chitosan against Schistocerca gregaria under semi field conditions.
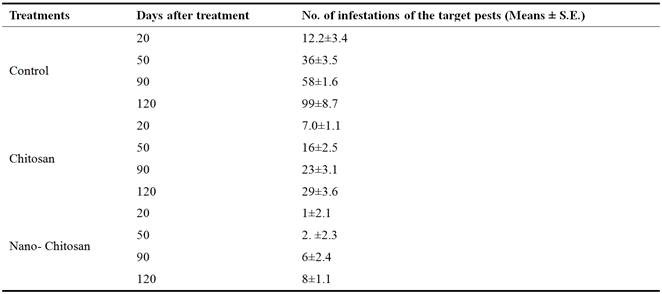
Figure 1, show that, the infestation percent significantly decreased especially after nano-chitosan treatments. Figure 2 show that, the nano Chitosanprticles by scanning electron microscopy. Figure 2 a and b show that the nano Chitosanat 50 and 100 nanometer.
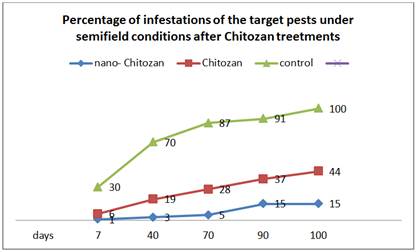
Fig. 1. Percentage of infestations of the target pests under semifield conditions after Chitosantreetments.

a. Sacnnining electron microscopy of nano Chitosanits size (50 nanometer)

b. Sacnnining electron microscopy of nano Chitosanits size (100 nanometer)
Fig. 2. Scanning electron microscopy, the nano chitosan particles 50 nano meter and b. 100 nanometer.
The obtained by [22] who, reported that, under laboratory conditions, the LC50s, were significantly decreased when the adult female of grasshopper Hetiracris littoralis treated with nano-destruxin and reached to 153X104spores/ml. Also, Under semi-field conditions, the percentage of infestations of H. littoralis significantly decreased to 1.0±0.3, 3±0.1, 5±3.0 and 10±2.9 individuals after treated with nano-destruxin in 20, 50, 90 and 120 days, respectively as compared to 15.2±2.9, 39±3.5, 66±9.6 and 98±6.6 individuals in the control. [23] found LC50s of the locust S. gregaria after treatment with destruxin, 210 X 104, 221 X 104, 250 X 104 spores/ml, of newly hatched nymphs last nymphal stage and adult stage., respectively The effect of nano-destruxin against S. gregaria under semi-field conditions show that after 20 days, the infestations number were significantly decreased to 2.2±1.2, as compared to 2.4±5.3, and 12.2±2.2 individuals after treated with destruxin and in the control. [13, 14] reported that. Desert locust Schistocerca gregaria bioassayed by using the leaves containing early stages larvae and the data were recorded after 1, 2, 3 and 4 days after treatment. Results showed that range of mortality was between 84-65% based on the end point data. The minimum of three days to achieve 60% mortality was proved by probit analysis of time-mortality responses. They found that, the range of mortality was between 88-65% based on the end point data. The minimum of three days to achieve 50% mortality was proved by probit analysis of time-mortality responses. The same results obtained by [24, 24, 25, 26, 27]. [28,28,30,21,32] found the insecticidal activity the nano-chitosan (CS-g-PAA) showed highest effect against the three insect of soybean. As the means number of eggs deposited /female were significantly decreased. Under laboratory and semifield condition, Aphis gossypii were significantly decreased to 20.9±9.1 and 28.9±9.2 eggs/female respectively as compared to 97.3±4.9 and 90.3±4.9 eggs/female in the control, respectively. The same trends were also observed against Callosobruchus maculatus. [33, 34, 35, 36, 37, 38] found that the nano insecticides of Imidacloprid and fungi strains cases a higher mortality for insect infestations. Our results agree with [39, 40, 41, 42]. [43, 27, 23], who find that the nano pesticide decrease the infestation percentage of different pests. [39, 40, 41, 42] reported that the usage of nano bioinsecticides, have the solvent characteristic in water which decrease the insecticide amounts due to to their very tiny size and its capability to infect the insect bodies and easier to enter the pest body. Also [43, 27, 23] tolled that, the nano particles not block the sprayer macin opining. [44, 45, 46, 47, 40, 37, 38] mentioned that, the nano-pesticides have a valuable effect in insect control, with a safe method than the chemicals. Said that the nano materials use the carrying methods which not pollute the environments and give the required results [43, 27, 23].
4. Conclusion
The usage of nano materials of chitosan which characterized by the very mall size which cusses the efficacy increase of the nano material which causes rapidly effect on the insect pests and accelerate the entrance to the insect body
Acknowledgments
This research was supported by Agric. Department, National Research Centre, and Cairo, Egypt. Project No (10120601 ).
References
- Bhuiyan, K.A. and Fakir G. A. (1993). Prevalence of seed-borne infection of Colletotrichum dematium var. truncatum on soybean and its control both in- vitro and in- vivo with different seed dressing fungicides. Thai J. Agric. Sci., 26: 1-10.
- Harris, C.A., Renfrew M.J. and M.W. Woolridge (2001),. Assessing the risk of pesticide residues to consumers: recent and future developments". Food Add and Cont., 18: 1124-1129.
- El Ghaouth, A. and Wilson C. L. (1995). Biologically-based technologies for the control of postharvest diseases," Postharvest News and Information, 6: pp. 5N–11N.
- Jo, Y.K., Kim B.H. and Jung G. (2009). Antifungal activity of silver ions and nanoparticles on phytopathogenic fungi". Plant Dis., 93:1037-1043.
- Rabea, E. I. Badawy M. E. I., Steurbaut W. and Stevens C.V. (2009). In- vitro assessment of N-(benzyl) chitosan derivatives against some plant pathogenic bacteria and fungi," European Polymer J., 45(1):237–245.
- Badawy, M. E. I. (2010). Structure and antimicrobial activity relationship of quaternary N-alkyl chitosan derivatives against some plant pathogens, J. of Appl. Polymer Sci, 117(2): 960–969.
- Kaur, P., Thakur R. and Choudhary A. (2012). An in-vitro study of antifungal activity of silver/chitosan nanoformulations against important seed borne pathogens. Int. J. of Scientific & Technology Res., 1(6): 83-86.
- El-Mohamedy, R.S., Abdel-Kareem F., Jaboun-Khiareddine H. and Daami-Remadi M. (2014). Chitosan and Trichoderma harzianum as fungicide alternatives for controlling Fusarium crown and root rot of tomato. Tunisian J. of Plant Prot., 9: 31-43.
- El-Shazly, M. M. 2000. Laboratory studies on the effect of selected host plant species on certain life table statistics of the grasshopper Heteracris littoralis (Rambur) (Orthoptera, Acrididae). J. Egypt. Ger. Soc. Zool. 33 (E): 65-79.
- Amer, M.M.; T.I. El-Sayed; H.K. Bakheit; S.A. Moustafa and Yasmin A. El-Sayed (2008). Pathogenicity and Genetic Variability of Five entomopathogenic fungi against Spodoptera littoralis. Res. J. Agric. and Biolog. Sci., 4(5): 354-367.
- Uvarov, B.P. (1966). Grasshoppers and Locusts. A Handbook of General Acridology. Vol. 1. Cambridge University Press, Cambridge, UK.
- Uvarov, B.P. (1977). Grasshoppers and Locusts. A Handbook of General Acridology. Vol. 2. Centre for Overseas Pest Research, London, UK
- Sabbour, M.M. (2013).Evaluating toxicity of extracted destruxin from Metarhizium anisopliae againstthe desert locustSchistocerca gregariain Egypt. J.Egypt. Acad. Environ. Develop., 14(1): 35-41.
- Sabbour, M.M. (2013).Evaluating toxicity of extracted destruxin from Metarhizium anisopliae againstthe grasshopperHetiracris littoralisin Egypt. J. Egypt. Acad. Environ. Develop., 14(1): 29-34.
- Vidyalakshmi, R., Bhakyaraj and Subhasree, R.S. 2009. Encapsulation the future of probiotics-A review. Adv.Biol. Res. 3(3-4): 96-103.
- Sakulk, Nuchuchua, U., Uawongyart, Puttipipatkhachorn,N., Soottitantawat and Ruktanonchai, U. 2009. Characterization and mosquito repellent activity of citronella oil nano emulsion. Int. J. Pharm. 372: 105-111.
- Sameh, A. Moustafa; Ahmed, E. Abd El-Mageed; Mostafa, M. El-Metwally and Nabil, M. Ghanim (2009). Efficacy of Spinosad, Lufenuron and Malathion against olive fruit fly, Bactrocera oleae (Gmelin) (Diptera: Tephritidae) Egypt. Acad. J. biolog. Sci., 2 (2): 171-178.
- Abbott, W.W. (1925). A method of computing the effectiveness of an insecticide. J. Econ. Entomol 18: 265-267.
- Finney, D.J. (1971). Probit Analysis, Cambridge: Cambridge University Press.
- Snedecor, G.W. and Cochran W.G. (1980). Statistical Methods, 7th ed. Ames (IA): Iowa State University Press.
- Shovan, M., Bhuiyan K. A., Sultana N., Begum j. A. and Pervez Z. (2008). Prevalence of fungi associated with soybean seeds and pathogenicity tests of the major seed-borne pathogens. Int. J. Sustain. Crop Prod. 3(4):24-33.
- Sabbour, M.M.2014a.Evaluating toxicity of extracted nano -Destruxin againstthe desert locustSchistocerca gregariain Egypt. J. Egypt. Acad. Environ. Develop. 15(2): 9-17.
- Sabbour, M.M.2014b. Evaluating Toxicity of nano-Extracted Destruxin from Metarhizium anisopliae Againstthe grasshopperHetiracris littoralisin Egypt. J.Egypt. Acad. Environ. Develop. 15(2): 1-7.
- Sabbour M.M1 and S.M. Singer.2015.Control of locust Schistocerca gregaria (Orthoptera: Acrididae) by using imidaclorprid. International Journal of Scientific & Engineering Research, Volume 6, Issue 10, October-2015. 243-247.
- Sabbour M.M and S.M. Singer. 2015.Control of Locust Schistocerca gregaria (Orthoptera: Acrididae) by Using Imidaclorprid. International Journal of Science and Research (IJSR) ISSN (Online): 2319-7064.
- Sabbour, M.M.2014.Evaluating toxicity of extracted nano -Destruxin againstthe desert locustSchistocerca gregariain Egypt. J. Egypt. Acad. Environ. Develop. 15(2): 9-17.
- Sabbour M.M. 2013e.Evaluating toxicity of extracted destruxin from Metarhizium anisopliae againstthe desert locustSchistocerca gregariain Egypt. J.Egypt. Acad. Environ. Develop. 14(1): 35-41.
- Sabbour, M.M. and Shadia E. Abed El-Aziz (2002). Efficacy of some botanical oils formulated with microbial agents against the cotton leafworm and greasy cutworm attaching cotton plants. Bull. Ent. Soc. Egypt. ser. 28, 2001-2002: 135-151.
- Sabbour, M.M. and Shadia, E. Abd-El-Aziz (2010). Efficacy of some bioinsecticides against Bruchidius incarnatus (BOH.) (Coleoptera: Bruchidae) Infestation during storage. J. Plant Prot. Res. 50 (1): 28-34.
- Sabbour, M.M. and sahab, A.F. (2005). Efficacy of some microbial control agents against cabbage pests in Egypt. Pak. J. Biol. Sci. 8: 1351-1356.
- Sabbour, M.M. and sahab, A.F. (2007). Efficacy of some microbial control agents against Agrotis ipsilon and Heliothis armigera in Egypt. Bull. N.R.C. Egypt. 13.
- Sabbour, M.M 2015. Efficacy of nano-extracted destruxin from Metarhizium anisopliae against red flour beetle, Tribolium castaneum and confused flour beetle, Tribolium confusum (Coleoptera: Tenebrionidae), under laboratory and store conditionsIntegrated Protection of Stored Products. IOBC-WPRS Bulletin Vol. 111, 2015. pp. 361-367.
- Sabbour, Magdaand MA Abdel-Raheem.2015. TOXICITY OF THE FUNGUS BEAUVERIA BASSIANA AND THREE OILS EXTRACT AGAINST SITOPHILUS GRANARIES UNDER LABORATORY AND STORE CONDITIONS. American J. of innovative research and applied sci. 251-256.
- Sabbour, Magdaand MA Abdel-Raheem.2015. Determinations the efficacy ofBeauveria brongniartiiand Nomuraea rileyi against the potato tuber moth Phthorimaea operculella (Zeller). The American Journal of Innovative Research and Applied Sciences.1(6):197-202
- Sabbour M. M.2015a. Laboratory and Store Efficacy of Nano-Extracted Destruxin from Metarhizium anisopliae Against Indian Meal Moth Plodia interpunctella (Lepidoptera-Pyralidae).Journal of Nanoscience and Nanoengineering. Vol. 1, No. 3, 2015, pp. 142-147. (http://www.openscienceonline.com/journal/ajbls).
- Sabbour M. M. 2015b.The Toxicity Effect of Nano Fungi Isaria fumosorosea and Metarhizium flavoviride against the Potato Tuber Moth, Phthorimaea operculella (Zeller). American Journal of Biology and Life Sciences. 3 (5):155-160.
- Sabbour, M.M. 2015. Nano-Chitosan Against Three Olive Pests Under Laboratory and Field ConditionsOpen Science Journal of Bioscience and Bioengineering2(5: 45-4990.
- Sabbour, M.M. 2015. Efficacy of some nano-Imidacloprid against red flour beetle Tribolium castaneum and confused flour beetle, Tribolium confusum (Coleoptera: Tenebrionidae) under laboratory and store conditions. Advances in Biochemistry & Biotechnology.1-13.
- Sabbour, M.M. and ShadiaEl-Sayed Abd-El-Aziz. 2015. Efficacy of some nano-diatomaceous earths against red flour beetle Tribolium castaneum and confused flour beetle, Tribolium confusum (Coleoptera: Tenebrionidae)under laboratory and store conditions. Bull. Env.Pharmacol. Life Sci., Vol 4 [7] June 2015: 54-59.
- Sahab, A. F.; Waly, A.I., Sabbour, M. M. and Lubna S. Nawar. 2015. Synthesis, antifungal and insecticidal potential of Chitosan (CS)-g-poly (acrylic acid) (PAA) nanoparticles against some seed borne fungi and insects of soybean. Vol.8, No.2, pp 589-598.
- Sahab, A.F. and Sabbour, M.M. (2011). Virulence of four entomo-pathogenic fungi on some cotton pests with especial reference to impact of some pesticides, nutritional and environmental factors on fungal growth. Egyp. J. Boil. Pest Cont., 21 (1): 61-67.
- Sabbour, M.M. 2015.Efficacy of Isaria fumosorosea and Metarhizium flavoviride against corn pests under laboratory and store conditions in Egypt.Journal of Global Agriculture and Ecology,: 2454-4205, Vol.: 5, Issue.: 1.
- Sabbour, M.M. 2015. A novel pathogenicity of nano- Beauveria bassiana and Metarhizeium anisoplae aginst Sitophius oryzae (Coleoptera: Curuliondae) under laboratory and store conditions. CONDITIONS. International Journal of Scientific & Engineering Research, Volume 6, Issue 12, December-2015.Vol.8, No.12 pp 121-129.
- Hussein M.M.Sabbour M.M. andSawsan Y. El-Faham. 2015.Adenine and Guanineapplication and its Effect on Salinity tolerant of Wheat plants and Pest infestations.International Journal of ChemTech Research. Vol.8, No.12 pp 121-129.
- Sabbour, M.M 2015. Efficacy of nano-extracted destruxin from Metarhizium anisopliae against Sitophilus oryzae (L.) (Coleoptera: Curculionidae) under laboratory and store conditions. Integrated Protection of Stored Products IOBC-WPRS Bulletin Vol. 111, 2015. pp. 369-375.



