Superparamagnetic Manganese Ferrite Nanoparticles: Synthesis and Magnetic Properties
Kisan Zipare1, Jyoti Dhumal1, Sushil Bandgar1, Vikas Mathe2,
Guruling Shahane1, *
1Department of Electronics, DBF Dayanand College of Arts & Science, Solapur, Maharashtra, India
2Department of Physics, Savitribai Phule Pune University, Pune, Maharashtra, India
Abstract
In this paper, manganese ferrite nanoparticles of controlled size were synthesized using chemical co-precipitation method and characterized for its structural and magnetic properties. The method is relatively simple, low cost and their particle size can be easily controlled. The X-ray diffraction pattern confirms the synthesis of single crystalline phase of MnFe2O4 nanoparticles with lattice parameter 8.5075Å. From TEM analysis it is seen that the sample contains nanoparticles of almost spherical in shape with the average particle size of 5 nm. The HRTEM image of the nanoparticles indicates the single crystalline nature which implies that high quality uniform manganese ferrite is obtained with our facile wet chemical method. Spectroscopic study implies that oleic acid is coated on the surface of the MnFe2O4 nanoparticles. The magnetic measurements show superparamagnetic nature of the sample. The saturation magnetization is 69emu/g. The superparamagnetic nature with high saturation magnetization makes these nanoparticles suitable for the synthesis of ferrofluid.
Keywords
Manganese Ferrite, Structural Characterization, Magnetic Properties
Received: August 21, 2015
Accepted: August 29, 2015
Published online: September 2, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Nanostructures of magnetic transition oxides with spinel structure MFe2O4 (M = Mn, Fe, Co, Ni, etc.) possess unique magnetic properties like moderate magnetization, high coercivity, single domain effects, superparamagnetism, spin filtering, etc. which leads to impressive industrial and biological applications [1]. As an important member of the ferrite family, MnFe2O4 (MFO) has attracted noteworthy research interest due to its mesmerizing magnetic and electromagnetic properties. MnFe2O4 is partially inverse spinel so that the about 80% of Mn2+ ions are located at tetrahedral (A) site while only 20% of them are located at octahedral (B) site [2]. Nanoparticles, nanostructures, and thin films of MnFe2O4 show diverse properties, such as, high anisotropy constant, size-dependent saturation magnetization, super spin glass state, superparamagnetism, and high Curie temperature. These properties of MFO result in many attractive applications such as magnetic recording, microwave, MRI contrast agent, ferrofluid, site specific drug delivery, magnetic tunnel junction-based sensor, photocatalytic, gas sensor and absorbent material for hot-gas. Recently, it was found that MnFe2O4 magnetic nano particles were effectively used an excellent adsorbent for the removal of the azo dye Acid Red B from water [3-6].
The properties of manganese ferrite highly depend on the composition, morphology and size, which are strongly connected with the preparation conditions. Many preparation technologies such as hydrothermal, combustion route, co-precipitation, sol-gel, mechano-chemical, solvo-thermal and reverse micelle have been developed to prepare the single-domain MnFe2O4 nanoparticles [1, 2, 5-10]. Co-precipitation is an attractive method of producing nanoferrites because of increased homogeneity, purity and reactivity. The method is relatively simple, low cost and their particle size can be tuned by optimizing the reaction parameters such as temperature, pH and ionic strength imposed by non-complexing salt [10].
Generally, MFO nanoparticles grown by conventional methods have low saturation magnetization due to dead layer effect. Hence, there is still the demand to synthesize uniform spherical nanoparticles of MnFe2O4 with high saturation magnetizations, low coercivities, and low remanent magnetizations for their possible applications in the synthesis of stable ferrofluid. Herein, we report an economical and efficient chemical co-precipitation method of large-scale synthesis of superparamagnetic Mn-ferrite nanoparticles with controlled size and high saturation magnetization.
2. Experimental Details
Mn ferrite nanoparticles were prepared by a facile chemical co-precipitation method [11,12]. For synthesis, equimolar solutions of AR grade MnCl2 and FeCl3 were mixed in their stoichiometric ratio and homogenized at room temperature. Oleic acid was used as surfactant to prevent agglomeration of particles. The pH of the solution was adjusted by adding 1M NaOH solution. The mixture was then heated at 80oC for about one hour. The particles were washed several times with de-ionized water to remove unwanted residual of salts and then dried at 35°C.
The X-ray diffraction patterns of the samples were recorded on Rigaku make powder X-ray diffractometer (Model- XRG 2KW) in the 2θ range from 20° to 80° at 40 kV, 30 mA using CuKα radiation (λ=1.54059 Å). The surface morphology and compositional analysis of the films was recorded on SEM (JEOL JSM 6360) scanning electron microscope operating at 20 kV. The shape and microstructure of the samples were analyzed by using transmission electron microscope (JEOL JEM-200CX). The magnetization measurements were carried out by vibrating sample magnetometer VSM Lake Shore Model 7307. The EPR measurements were carried out by using X-band Bruker EPR spectrometer (Model-A300).
3. Results and Discussion
The X-ray diffraction pattern of as-synthesized sample is shown in Fig. 1. The diffraction pattern provides a clear evidence for the formation of ferrite phase. The ‘d’ values and intensities of observed diffraction peaks match with the single crystalline spinel form of the MnFe2O4 (JCPDS Card No. 074-2403). The broad XRD line indicates that the ferrite particles are of nano size. The lattice parameter is calculated and found to be 8.5075Å. The value of lattice parameter is in good agreement with the standard JCPD value. The crystallite size of each composition was then determined from the full width at half maximum intensity for (311) plain using Scherrer’s formula:
D = 0.89 l /b cos q (1)
where, D is the crystallite size, l is wavelength of X-ray, b is full width at half maximum (FWHM) measured in radians and q is the Bragg angle. The average crystallite size is 4.8 nm.

Fig. 1. XRD pattern of MnFe2O4 nanoparticles.
Compositional analysis of the samples without oleic acid coating was done by energy dispersive X-ray spectroscopy technique. The EDX technique supplies the effective atomic concentration of different constituents on top surface layers of the solid investigated. The energy dispersive X-ray spectra of MnFe2O4 nanoparticles (Fig. 2) show the peaks of Mn, Fe and O along with the C substrate peak. It is observed that the atomic percent of Mn, Fe and O are 12.97, 21.51 and 54.70 respectively. The other minor peaks might be due to the impurities in the starting materials.
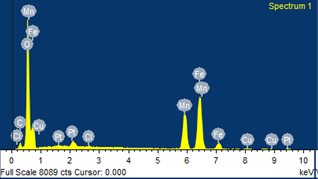
Fig. 2. EDX spectrum of MnFe2O4 nanoparticles.
The surface morphology of the samples was studied with scanning electron microscope. The SEM images of MnFe2O4 nanoparticles with different magnification are shown in Fig. 3. It is seen that sample consists of nanoparticles agglomerated together to form large grains. The TEM image of as-synthesized MnFe2O4 nanoparticles and the corresponding selected area electron diffraction (SAED) pattern are shown in Fig 4a. The sample contains nanoparticles of almost spherical in shape in the range 3nm-8nm with the average particle size of 5 nm. The observed particle size is in good agreement with the XRD value. The SAED pattern (Fig. 4a inset) provides the d-spacing consistent with those obtained from XRD. The high resolution TEM (HRTEM) image of the nanoparticles is shown in Fig. 4b. The lattice fringes of the core indicates the single crystalline nature with lattice spacing 0.26nm which corresponds to (311) lattice plane. These observations imply that high quality uniform phase pure manganese ferrite is obtained with our facile wet chemical method.


Fig. 3. (a, b) SEM images of MnFe2O4 nanoparticles with different magnification.
Formation of the spinel MnFe2O4 nanoferrite was further supported by FTIR analysis. Fig. 5 shows the FTIR spectrum of oleic acid coated Fe3O4 nanoparticles recorded at room temperature. The spectra show two main absorption bands below 1000 cm−1 which is a common feature of ferrites. The characteristic peaks at 449cm-1and 573cm-1 which corresponds to intrinsic stretching vibrations of the metal-oxygen at the octahedral and tetrahedral sites, respectively. Peak at 1122 cm-1 can be attributed to the vibrations of –CH. Peak at 1384 cm-1 is assigned to the vibrations of C=C in oleic acid. The strong peaks at 1636 cm-1 and 3423cm-1 can be assigned to stretching vibrations of –CH2 and –CH3, respectively. The spectroscopic studies confirm that oleic acid is coated on the surface of MnFe2O4 nanoparticles [13, 14].
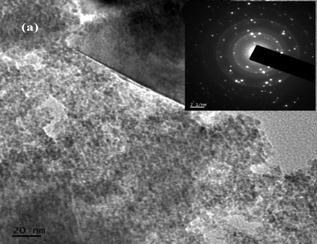
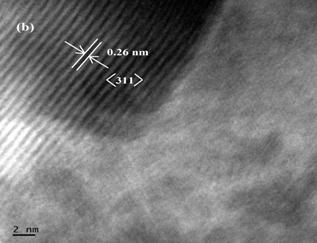
Fig. 4. (a) TEM image with SAED pattern and (b) HRTEM image of MnFe2O4 nanoparticles.
The magnetization curve of Mn-ferrite is shown in Fig. 6. The magnetization curve demonstrates superparamagnetic behavior of the sample with zero remanence and coercivity. The particles have superparamagnetic behavior when KV˂kT, where, K is anisotropy constant, V is volume, k is Boltzmann constant and T is temperature. The superparamagnetism of these nanoparticles can be attributed to their small crystallite size, which makes it easier for them to be thermally activated to overcome the magnetic anisotropy [15]. Rafique et al. have calculated the critical diameter for MnFe2O4 particle at T=300 K and found to be 42.9nm, below which the particle possess superparamagnetism [1]. Gnanaprakash et al also reported that the particle size limit for MnFe2O4 particle is 42nm [10]. The size of the synthesized MnFe2O4 particles is about 5 nm which is well below the critical size limit showing the superparamagnetic behavior of the sample. It is seen that saturation magnetization for MnFe2O4 is 69 emu/g. The observed value of saturation magnetization is small as compared to the bulk value (80 emu/g) but large as compared to other reported values [4, 16]. The smaller value of saturation magnetization is due to lattice defects, weaker magnetic super-exchange interactions between A-sites and B-sites, and existence of a magnetically inert layer at the surface of the nanoparticles. Due their broken bonds, iron ions contained in superficial layer present a random orientation of their magnetic moments, thereby leading to a decrease of the total magnetization of the nanoparticles [17].
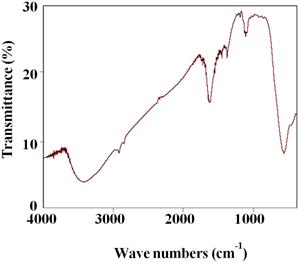
Fig. 5. FTIR spectrum of MnFe2O4 sample.
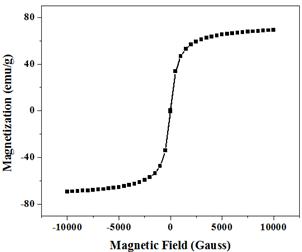
Fig. 6. Magnetization curve of MnFe2O4 nanoparticles.
In order to understand the information about the spin related phenomena the electron paramagnetic resonance (EPR) measurements have been carried out on the sample. The EPR spectrum of MnFe2O4 sample was recorded at ambient temperature and is shown in Fig. 7. From figure it is seen that the EPR spectrum is narrow, which is the characteristic property of superparamagnetic materials [15]. The spectrum was analyzed using Lorentzian distribution function to obtain the values of various parameters such as resonant magnetic field (Hr), peak-to-peak line width (DHPP) and g-value. The values of magnetic field at resonance (Hr) and resonance line width (ΔHpp) are 3367 G and 195 G, respectively. The g-value, which is a constant of proportionality between the frequency and the field, is a function of the molecular motion, the paramagnetic properties and the symmetry of ions. The value of g is calculated by the relation:
g = hn / bH (2)
where, h is a Plank constant, n is the microwave frequency, b is Bohr magneton and H is the magnetic field at the resonance. The value of g is found to be 2.06. The smaller magnitudes of line width and g-value in the present study indicate that super-exchange interaction between magnetic ion and oxygen ion is the main factor that determines EPR resonance parameters [15].
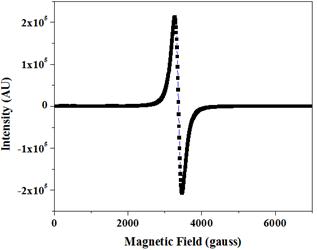
Fig. 7. EPR spectrum of MnFe2O4 nanoparticles.
4. Conclusion
A chemical co-precipitation method is used to synthesize size-controlled synthesis of Mn-ferrite nanoparticles. The method is relatively simple, low cost and their particle size can be easily controlled. The X-ray diffraction pattern confirms the synthesis of single crystalline phase of MnFe2O4 nanoparticles with lattice parameter 8.5075Å. From TEM analysis it is seen that the sample contains nanoparticles of almost spherical in shape with the average particle size of 5 nm. The HRTEM image of the nanoparticles indicates the single crystalline nature which implies that high quality uniform manganese ferrite is obtained with our facile wet chemical method. Spectroscopic study implies that oleic acid is coated on the surface of the MnFe2O4 nanoparticles. The magnetic measurements show superparamagnetic nature of the sample. The saturation magnetization is 69emu/g. The superparamagnetic nature with high saturation magnetization makes these nanoparticles suitable for the synthesis of ferrofluid.
Acknowledgements
The author (GSS) is thankful to the Science and Engineering Research Board, Government of India, New Delhi, for the financial assistance under the Project Grant SB/S2/CMP-06/2013. Thanks are also extended to Dr. R. P. Pant, National Physical Laboratory, New Delhi for timely help and useful discussions.
References
- Rafique M. Y., Li-Qing P., Javed Q., Iqbal M. Z., Hong-Mei Q., Farooq M. H. Zhen-Gang G. and Tanveer M., "Growth of monodisperse nanospheres of MnFe2O4 with enhanced magnetic and optical properties". Chin. Phys. B, 22 (10) (2013) 107101-107107.
- Deraz. N. M, Alarifi. A., "Controlled Synthesis, Physicochemical and Magnetic Properties of Nano-Crystalline Mn Ferrite System". Int. J. Electrochem. Sci., 7 (2012) 5534-5543.
- Carta D., Casula M. F., Floris P., Falqui A., Mountjoy G., Boni A., Sangragoria C. and Corrias A., "Synthesis and microstructure of manganese ferrite colloidal nanocrystals". Phys. Chem. Chem. Phys., 12 (2010) 5074-5083.
- Kim D. H., Nikles D. E. and Brazel C. S., "Synthesis and Characterization of Multifunctional Chitosan- MnFe2O4 Nanoparticles for Magnetic Hyperthermia and Drug Delivery". Materials, 3 (2010) 4051-4065.
- Mozaffari M., Behdadfar B., and Amighian J., "Preparation and Characterization of Manganese Ferrite Nanoparticles via Co-precipitation Method for Hyperthermia". Iranian J. Pharmaceutical Sciences, 4(2) (2008)115-118.
- Sam S. and Nesaraj A. S., "Preparation of MnFe2O4 Nanoceramic Particles by Soft Chemical Routes". Int. J. Appl. Sci. Eng., 9(4) (2011) 223-239.
- Kwon W. H., Kang J. Y., Lee J. G., Lee S. W., Chae K. P., "Synthesis and magnetic properties of Zn, Co and Ni substituted manganese ferrite powders by sol-gel method". J. Magnetics, 15(4) (2010) 159-164.
- Lazarevic Z. Z., Jovalekic C., Milutinovic A., Romcevic M. J. and Romcevic N. Z., "Preparation and Characterization of Nano Ferrites". Acta Physica Polonica A, 121 (3) (2012) 682-686.
- Scano A., Ennas G., Frongia F., Barbera A. L., Lopez-Quintela M. A., Marongiu G., Paschina G., Peddis D., Pilloni M. and Vazquez-Vazquez C., "Mn-ferrite nanoparticles via reverse microemulsions: synthesis and characterization". J. Nanopart. Res. 13 (2011) 3063-3073.
- Gnanaprakash G., Philip J. and Raj B., "Effect of divalent metal hydroxide solubility product on the size of ferrite nanoparticles". Materials Letters, 61 (2007) 4545-4548.
- Shahane G. S., Zipare K. V. and Pant R. P., "Synthesis and Characterization of Superparamagnetic Fe3O4 Nanoparticles for Ferrofluid Application". Magnetohydrodynamics, 49 (3-4) (2013) 317-321.
- Shahane G. S., Kumar A., Arora M., Pant R. P. and Lal K., "Synthesis and characterization of Ni-Zn ferrite nanoparticles".J. Magn. Magn. Mater. 322 (2010) 1015-1019.
- Venkataraju C. and Pulsingh R., "FTIR and EPR studies of Nickel Substituted Nanostructured Mn Zn Ferrite". J. Nanoscience, (2014) Article ID 815385, 5 pages. http://dx.doi.org/10.1155/2014/815385.
- Kanade K. G., Amalnerkar D. P., Potda H. S. and Kale B. B.,"Nanocrystalline Mn-Zn ferrite by novel oxalato-hydrazinated complex method". Mat. Chem. Phys., 117 (2009) 187-191.
- Chand M., Kumar A., Annveer, Kumar S., Shankar A. and Pant R. P., "Investigations on MnxZn1-xFe2O4 (x=0.1, 0.3 and 0.5) nanoparticles synthesized by sol-gel and co-precipitation methods". Ind. J. Eng. Mater. Sci., 18 (2011) 385-389.
- Sharma U. S., Sharma R. N. and Shah R., "Physical and Magnetic Properties of Manganese Ferrite Nanoparticles". Int. J. Eng. Res. Appl., 4(8) (2014) 14-17.
- Venkataraju C., Satishkumar G. and Shivkumar K., "Effect of cation distribution on the structural and magnetic properties of nickel substituted nanosized Mn-Zn ferrites prepared by co-precipitation". J. Magn. Magn. Mater. 322 (2010) 230-233.



