Waste Mediated Synthesis of Alginate/Ag Beads for Tertiary Water Treatment
Padmanaban Sivakumar1, Kamalakkannan Anbarasu2, Kanniyan Pandian3, Sahadevan Renganathan4, Pandian Sivakumar4, 5, *
1Research and Development Division, Chennai Petroleum Corporation Limited, Chennai, Tamilnadu, India
2Quality Control Department, Tamilnadu Co-operative Milk Producers Federation Limited, Aavin, Sholinganallur, Chennai, Tamilnadu, India
3Department of Inorganic Chemistry, University of Madras, Chennai, Tamilnadu, India
4Department of Chemical Engineering, Anna University, Chennai, Tamilnadu, India
5Department of Petrochemical Engineering, RVS college of Engineering and Technology, Coimbatore, Tamilnadu, India
Abstract
Reuse of waste bio source from food industrial waste. In this study, concentrated dairy effluent was utilized to synthesis silver nano particles (AgNPs), which is used to prepare alginate composite micro beads were for slow delivery of silver. These beads were then used to disinfect dairy tertiary treated water. Initially, AgNPs were synthesized and characterized using Ultra Violet-visible spectrum (UV-Vis), X-Ray Diffraction (XRD) and High Resolution Transmission Electron Microscope (HRTEM). The composite beads were made from AgNPs and alginate. The physical structure and silver content were determined using optical microscope and Inductively Coupled Plasma Optical Emission Spectroscopy (ICPOES), respectively. Packed bed column study was carried to know the inactivation kinetics on reduction of total microbial load and coliform were carried out in tertiary treated water. This study confirms that these beads were effective in disinfecting water, which was mainly due to encapsulated and relatively slow release of AgNPs.
Keywords
Dairy Effluent, Silver Nanomaterial, Synthesis: Alginate-Ag Composite, Inactivation Kinetics
Received: August 10, 2015
Accepted: August 23, 2015
Published online: September 2, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Pathogenic microorganisms present in potable water sources pose major threats to public health. With increasing demands for higher quality water to fulfill ever-increasing public and industrial needs, the generation of pathogen-free uncontaminated water with limited byproducts has gained significant attention [1]. About 1.8 million people, most of them are children; die every year from diarrheal diseases. Nano scale materials have received increasing interest as alternative disinfectants due to their high interfacial reactivity and unique physicochemical properties [2]. Several factories generate large quantities of waste water. Removal of contaminants and reuse of the purified water would provide substantial reductions in waste to industry [3]. Water treatment is an important problem in recent years due to water loss and low quality of distribution networks and transport cost of water. An alternative source for decrease the water demand is necessary. Reuse of waste water is a better alternative way to compensate the water shortage problem [4,5]. Directly adding antimicrobial agents to food or into packaging materials during food processing [6]. This is considered as an effective means of controlling microbial contaminants in food and extending the shelf life of fresh produce and meat. In recent years, inorganic antimicrobial agents such as metals and metal oxides have received increasing attention in food applications [7]. Unlike conventional chemical disinfectants, these antimicrobial nanomaterial are not strong oxidants and are relatively inert in water. Therefore, they are not expected to produce harmful disinfectant by products. If properly incorporated into treatment processes, they have the potential to replace or enhance conventional disinfection methods [8]. It is proposed that functional nanomaterial with antimicrobial properties can be used to build a small-scale systems to decrease the water shortage. Testing for all individual pathogens is impractical and expensive. Total coliform counts react to the environment and treatment process in a manner and degree similar to pathogens. Due to this association, the bacterial safety of drinking water is monitored by testing for total coliform bacteria.
Dairy effluents are formed in several ways by washing milk containers and through many processes by products. These effluents are considered as a waste and drain out from the dairy industry [9]. The wastes containing some organic compounds are having property of chemical reaction with metals. The waste containing fatty acids is used for the production of biodiesel [10]. Biodiesel production proven that the effluent can be used for the chemical reaction.
Nanomaterial is having unique physical properties and chemical nature by increasing the surface to volume ratio [11]. Metal nanoparticles of Ag, Au and Pt are having antimicrobial property and less toxic than other metals and metal oxide nanoparticles. With the rapid development of nanotechnology, applications have been extended further and now silver is the engineered nanomaterial most commonly used in consumer products [12,13]. Coliform organism treated with ultrasonic irradiation for short time periods before Ag nanoparticle treatment at low concentration, enhances antibacterial effect. Then in future combination of both maybe the best option for treatment of wastewater [14].
In this investigation, concentrated dairy effluent was used for the synthesis of AgNPs. The synthesized AgNPs was incorporated with alginate beads for disinfecting microorganism present in the treated water. Colloidal silver has been accepted by the United States Environmental Protection bureau as a disinfectant for clinical and health centers. Colloidal silver at low ppm concentration has been found effective in killing numerous pathogens [15-17].
However, easy agglomeration of AgNPs is due to the larger surface area and higher surface energy is a main problem. Stabilize and prolong release of AgNPs using polymers is one of the viable method for utilization. Calcium alginate is a high molecular weight biocompatible copolymer which composed of mannuronate and guluronate monomers Alginate widely used as a biopolymer for, wound dressings, transplanted cells, drug delivery and many other biomedical applications [18]. Formation of AgNPs incorporated alginate hydrogels solve these issues and enhances the antimicrobial activity.
Herein, we have reported a green and waste reuse method for synthesize of silver nanoparticle and simple method for incorporation of silver nanoparticles in the alginate beads. The synthesized materials were used for improve the quality of waste water with slow release and less toxic effect within the toxic limit as stated by World Health Organization. The results shown are effective for water treatment, better than earlier methods of synthesis and applications.
2. Materials and Methods
2.1. Materials
Concentrated milk dairy effluent pooled sample and tertiary treated water were collected from Aavin dairy industry, Solinganallur, India. AgNO3 (ACS grade), Double deionized water, NaOH, Whatmann No.1 filter paper (25 micron) were purchased from Aldrich Chemicals India. Sodium alginate with average molecular weight and calcium chloride were obtained from Merck, Mumbai, India. Tris (hydroxymethyl) amino methane-phosphate buffer was purchased from Sigma Aldrich, Mumbai, India. Casein was prepared in 50 mM Tris buffer and pH adjusted using NaOH. Centrifuge filters molecular weight cut off value of 100 kDa (Merck, Mumbai, India).
2.2. Method
Dairy effluents have an alkaline pH value (6.6-12.2) and are rich in organic matters [19]. The collected milk dairy sample was taken in a 500 mL beaker. Sample was heated to 40º C and diluted glacial acetic acid was added slowly into the sample till reach the isoelectric point of pH 4.6 to separate the casein from milk waste. Casein was separated and settled down in the beaker. The separated casein was filtered and pressed out the water using whatman filter paper then it was allowed to air dry. Dried powder was suspended (100 mL, 1%) in Tris-buffer with an adjusted pH of 13. It was quickly added to a boiling aqueous solution of 1 mM AgNO3 (250 mL,) under vigorous stirring. The color of the reaction mixture changes from colorless to yellow within 10 min. The synthesized nanomaterial was then filtered to remove un-reactant casein and impurities using centrifuge filters and then stored at room temperature for further analysis. The final residue was redispersed in a 50 mL ultra-pure and 3 % (w/v) of sodium alginate was added. The mixture was stirred for 1 h in a magnetic stirrer for complete dissolution. Calcium chloride 2 % (w/v) was taken in 500 mL beaker. The mixture was filled in a 50 mL burette and then added drop by drop in calcium chloride solution. The final beads hardened for 1 h in calcium chloride solution.
2.3. Instrumentation
UV-vis absorption spectra were recorded with a Perkin Elmer Lambda 25 instrument in the spectral range of 200 to 1100 nm. The structure of the samples was characterized by XRD data were collected with a shimadzu XD-D1 diffractrometer using Cu Ka (λ = 1.54 Aº) radiation. The samples were scanned in the 2θ range of 10 to 90˚. The chemical composition and morphology of the samples were captured using HRTEM JEOL 3010.The morphology of Alginate/AgNPs composite beads was captured using an optical microscope (BX51TF, Olympus) equipped with digital camera. Silver concentration in beads and water were analyzedin ICPOES Perkin Elmer Optima 5300 DV.
2.4. Standard Plate Count (SPC) & Coliform
Water containing SPC and coliform was calculated by the plate count method. Samples were taken at different time intervals, agar was used to enumerate the total count of were mesophlic aerobic bacteria, and coliform numbers were determined using violet red bile agar plates incubated at 37 °C for 48 h. The above-mentioned experiments were repeated again at different time intervals for microbial inactivation kinetics [9]. Different weight of Alginate/Ag micro beads with different concentration 0.1g to 10.g were added into tertiary water for determine the minimum concentration of Ag used for microbial control. Packed bed reactor experiment was carried to apply the synthesized material to industrial scale microbial inactivation.
3. Result and Discussion
3.1. UV-Visible Spectroscopy Result
The change in color occurred in the solution from colorless to yellow confirm the formation of silver nanoparticles. UV-Vis result shows the broad peak in the range of 400-500 nm wavelength confirms the AgNPs formed from the silver nitrate by reduction and capping of casein. Casein present in waste water has high organic load (high concentrations of COD and BOD) making it too polluted for its direct disposal on land. Separation of casein from the waste water minimizes BOD & COD problems [20]. After separation of solids by adding mild acid and centrifuged, the separated casein was reacted with silver nitrate to form Silver nano was shown in UV-Vis spectrum (Fig.1). The tyrosine and histidine of casein may be responsible for the reduction of ionic silver into silver nano. Most of carboxyl group containing molecules reduction potential was improved at higher pH and most of the hydrosols of gold and silver are produced in alkaline salts of carboxylic acids. The optimum pH for the formation of silver nanoparticles was found to be pH-13 and for further characterization and applications of nanomaterial was prepared at this pH [21]. Milk effluent acting as both reducing and capping agents.
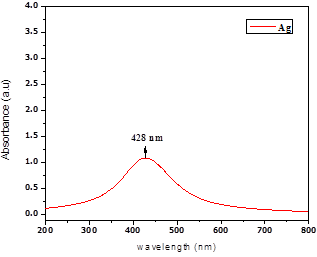
Fig. 1. UV-visible absorption spectra of silver nano after removal of dispesrsion solids in dairy Effluent.
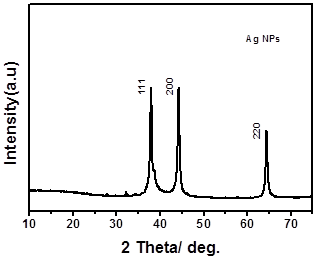
Fig. 2. XRD spectra of synthesized silver nano.
3.2. XRD & HRTEM
Crystalline nature of synthesized silver nano was confirmed from X-ray diffraction peak values at 38, 42, and 64. These values are corresponding to the planes of [111], [200] and [220] which is confirms the formation of silver nanoparticles (Fig. 2). These values explain the silver nano having face centrecubic crystal lattice structure [22]. Surface morphology of synthesized Silver nano material was confirmed through HRTEM image. The resultant image shows that the synthesized materials having different shapes like trigonal, sphere and rod shape materials, which was confirmed through image result (Fig.3). This different shape of material formation might be on reducing agent, pH and reaction condition.

Fig. 3. HRTEM image of silver nano with various size and shape.
3.3. Alginate/Ag Optical Microscopy and UV-Visible Results
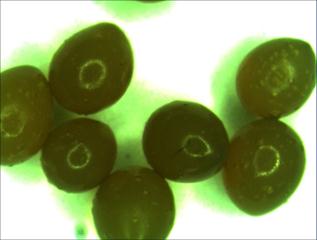
Fig. 4. Microscopic image of Alginate/Ag composite beads.
Synthesized Alginate/AgNPs composite beads were viewed through optical microscope having nearly spherical shape was shown in photos (Fig. 4). The beads were then immersed into the deionized water for identify the silver leaching in water. The immersed water was analyzed through UV-Vis spectrum. Deionized water was taken as a control and beads immersed deionized water was taken as the sample after removed the beads. Silver leaching in deionized water after addition of Alginate/AgNPs beads was confirmed through UV-Vis absorbance peak (Fig. 5).
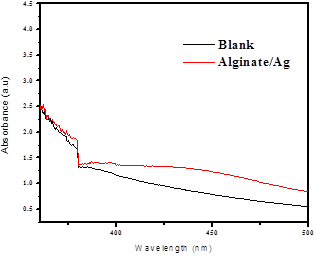
Fig. 5. UV-visible specrtrum results of Alginate/nano Ag composite Beads immersed in water and blank deionized water.
3.4. SPC & Coliform
Table 1. Tertiary treated water with same volume against different concentration of Alginate/Ag.
| S.NO | Weight of Alginate/Ag (g) | Volume of Tertiary water (ml) | SPC | Coliform |
| 1 | 0 | 50 | 300 | 330 |
| 2 | 10 | 50 | 45 | 0 |
| 3 | 9 | 50 | 60 | 0 |
| 4 | 8 | 50 | 70 | 0 |
| 5 | 7 | 50 | 70 | 0 |
| 6 | 6 | 50 | 70 | 0 |
| 7 | 5 | 50 | 80 | 0 |
| 8 | 4 | 50 | 80 | 0 |
| 9 | 3 | 50 | 80 | 0 |
| 10 | 2 | 50 | 90 | 0 |
| 11 | 1 | 50 | 90 | 0 |
Table 2. Tertiary treated water with same volume against minimum weight of Alginate/Ag.
| S. NO | Weight of Alginate/Ag (g) | Volume of Tertiary water (ml) | SPC (counts) | Coliform (counts) |
| 1 | 1 | 50 | 90 | 0 |
| 2 | 0.9 | 50 | 110 | 0 |
| 3 | 0.8 | 50 | 110 | 2 |
| 4 | 0.7 | 50 | 115 | 3 |
| 5 | 0.6 | 50 | 115 | 3 |
| 6 | 0.5 | 50 | 115 | 3 |
| 7 | 0.4 | 50 | 125 | 4 |
| 8 | 0.3 | 50 | 125 | 5 |
| 9 | 0.2 | 50 | 130 | 5 |
| 10 | 0.1 | 50 | 130 | 5 |
Coliforms and SPC present in the water was observed at different weight ratio. Weight of 1 to 10 g of beads totally reduces the coliform to zero counts in 50 mL tertiary treated water. SPC count was decreasing from 1 to 10g was shown in table.1. Further decreasing the concentration of silver nano by adding Alginate/ag micro beads from 1g to 0.1g for same 50 mL water. Decreasing the amount of silver nano, inactivation of SPC and Coliform was less shown in table. 2. The Tertiary treated undisinfected water experimental and control counts of spc and coliform with a constant weight of 1g in 50 mL water was shown in table. 3. Reason for maintaining a predominance of hypochlorous acid during treatment has to do with the fact that pathogen surfaces carry a natural negative electrical charge. These surfaces are more readily penetrated by the uncharged, electrically neutral hypochlorous acid than the negatively charged hypochlorite ion. The same reason follows here bulk positive charged Ag+ having less penetrating power than neutral Ag0. The neutral silver nano can easily penetrate on smaller in size and larger surface area with very fast inactivation. Alginate/Ag composite beads were used to kill the micro-organisms shown better results. There is no need of replace for long term and energy consumes biodegradable biopolymer material.
Table 3. Tertiary treated water experimental and control counts of SPC and Coliform.
| Time | Coliform (Control) | Coliform (Experiment) | SPC (Control) | SPC (Experiment) |
| 0 | 330 | 330 | 300 | 300 |
| 1 | 330 | 310 | 300 | 210 |
| 2 | 330 | 300 | 300 | 210 |
| 3 | 330 | 270 | 300 | 190 |
| 4 | 330 | 230 | 300 | 160 |
| 5 | 330 | 170 | 300 | 140 |
| 6 | 330 | 100 | 300 | 120 |
| 7 | 330 | 0 | 300 | 110 |
| 8 | 330 | 0 | 300 | 95 |
| 9 | 330 | 0 | 300 | 95 |
| 10 | 330 | 0 | 300 | 90 |
3.5. Inactivation Studies of Packed Bed Reactor
The experimental setup consists of a liquid storage tank from which a centrifugal pump pumps water. The flow rate of water is measured using a Rotameter. A schematic diagram of experimental setup is shown in the Figure 6. A vertical perspex column, 1.6 m tall with internal diameter of 5.4x10-2 m and outer diameter of 6x10-2m is used, which consists of three sections liquid distributor section, test section and liquid collection section. The liquid distributor is located at the bottom of the test section and designed in such a manner that uniform distribution of the liquid can be maintained in the column. The liquid inlet pipe of 2.5 x10-2m I.D. is located centrally in this section. Test section is filled with micro beads. The outlet of the test section is at a height of 1.5 m and has a mesh attached to it in order to retain the beads. There are two pressure tapings provided at the top and the bottom of the test section which are connected to the manometer for pressure drop measurement. The liquid velocity is kept constant till a steady state is attained and samples were taken. The same procedure is repeated for different liquid velocities.
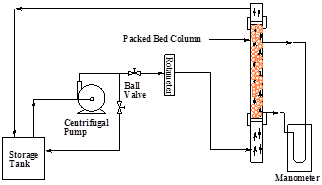
Fig. 6. Packed bed reactor for tertiary water treatment.
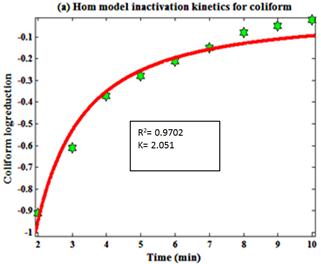
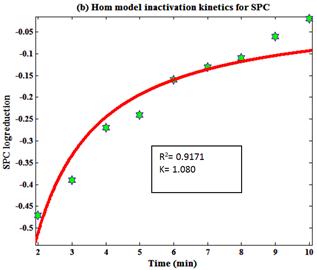
Fig. 7. Hom model inactivation kinetics for tertiary treated water containing (a) Coliform and (b) SPC.
3.6. ICPOES Results
Silver leaching and amount of silver ion concentration in tertiary water was further confirmed from the ICPOES analysis. Amount of silver ions present in tertiary water was 30ppm (15 µg L-1 = 0.000015g) in 500 mL water. This result shows that silver ion concentration present in the water is not exceed than WHO limit of 100µgL-1[2]. The increasing liquid velocity decreases the number of coliform and SPC in water due to increasing contact time of water with Alginate/Ag beads.
3.7. Inactivation Kinetics for Disinfection of Tertiary Treated Water
Hom model
Log N/N0 = -k Ca Tb
Where N is the number of microbial population at inactivation time and N0 is the initial number of microbial population. K is the reaction constant, C is the concentration, T is the time in min, a and b are variables. Hom model kinetics was used to identify the inactivation of microbial population. Hom model inactivation kinetics gives the different sign of R2 values (fig.7) for SPC and Coliform. Graphical representation of kinetics was plotted using MATLAB 7.8.0.347 as shown in (Fig.7). Experimental plot for coliformand SPC fit with a Hom model curve is shown in (Fig.7).
3.8. Microbial Inactivation Percentage in Tertiary Treated Water
(BC - BT) 100/BC
BC initial control microbial concentration and BT the microbial concentration in time.
Below chart explain the percentage of inactivation of coliform and SPC. Initial time of 1 min 6% of coliform and SPC were reduced by adding Alginate/Ag micro beads. At 10 min 100% coliform and 70% SPC were killed. When the time increase amount of microbial inactivation gets increase on reaction with silver nano is shown in (Fig.8).
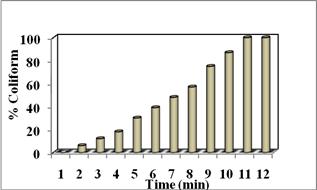
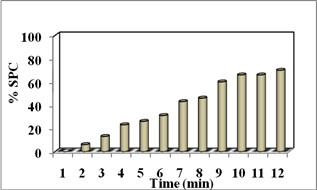
Fig. 8. Inactivation percentage of tertiary treated water containing (a) Coliform and (b) SPC.
4. Conclusion
Dairy waste containing casein proteins was used to produce eco-friendly and highly stable silver nanoparticles without using any additional reducing agent. Tertiary treated undisinfected water was effectively purified from pathogens to zero level. Small amount of material was enough to kill the organism present in the large quantity of water was confirmed. This method will be an effective technique in utilizing and reuse of waste in the field of nanotechnology. A Greener, reusable, slow delivery and less toxic Alginate/Ag nanocomposite beads were synthesized and used for control the micro-organism present in tertiary undisinfected water. This composite will be a good antimicrobial material in water treatment.
References
- Xie. Y. (2003). "Disinfection Byproducts in Drinking Water: Formation, Analysis, and Control", Boca Raton, FL, CRC Press.
- Siew. L. L, Anthony. G.F, Teik. T.L, William. B.K, Yen. N.L, Xin. L and Xiao. H. (2013). "Superabsorbent Cryogels Decorated with Silver Nanoparticles as a Novel Water Technology for Point-of-Use Disinfection", Environ. Sci. Technol, 47, 9363−9371.
- Nora. S, Mamadou. S.D. (2005). "Nanomaterials and water purification: Opportunities and challenges", Journal of Nanoparticle Research, 7, 331–342.
- Sivakumar. P, Sivakumar. P, Anbarasu. K, Pandian. K, Renganathan. S. (2013). "Synthesis of silver nano-rods from food industrial waste and its application on improving keeping quality of milk", Ind. Eng. Chem. Res, 52, 17676−17681,
- Haas C.N. (2000). "Disinfection in the twenty-first century", J. Am. Water. Works. Assoc. 92, 72-74.
- Li. Q, Mahendra S, Lyon. D.Y, Brunet. L, Liga. M.V, Li. D, Alvarez. P.J.J. (2008). "Antimicrobial nanomaterials for water disinfection and microbial control: Potential applications and implications", Water Research, 42, 4591-4602.
- National Research Council. (2006). "Drinking Water Distribution Systems: Assessing and Reducing Risks", Washington, DC: The National Academies Press.
- Yanping. X, Yiping. H, Peter. L.I, Tony. J, Xianming. S.(2011)."Antibacterial Activity and Mechanism of Action of Zinc Oxide Nanoparticles against Campylo bacter jejuni", Appl. Environ. Microbiol. 77, 2325-2331.
- Sivakumar. P, Bhagiyalakshmi. M. Renganathan. S. (2012). "Anaerobic treatment of spoiled milk from milk processing industry for energy recovery – A laboratory to pilot scale study", Fuel, 96, 482-486.
- Sivakumar. P, Anbarasu. K, Renganathan. S. (2011). "Bio-diesel production by alkali catalyzed transesterification of dairy waste scum", Fuel, 90, 147–151.
- Roduner. E. (2006). "Size matters: why nanomaterials are different", Chem. Soc. Rev. 35, 583-592.
- Catalina. M.J, Eric. M.V.H. (2010). "A review of the antibacterial effects of silver nanomaterials and potential implications for human healthand the environment", J. Nanopart. Res. 12, 1531–1551.
- Poernomo. G, Cong. G, Xianghua. S, Quanyuan. Z, Susanna. S.J.L. (2011). "Hallow fiber membrane decorated with Ag/MWNTS: Towards effective water disinfection and biofouling control", ACS Nano, 12, 10033–10040.
- Alt. V, Bechert. T, Steinrucke. P, Wagener. M, Seidel. P, Dingeldein. E, Domann. E, Schnettler. R. (2004). "An in vitro assessment of the antibacterial properties and cytotoxicity of nanoparticulate silver bone cement", Biomaterials, 25, 4383–4391.
- Lee. H.Y, Park. H.K, Lee. Y.M, Kim. K, Park. S.B.A. (2007). "Practical procedure for producing silver nanocoated fabric and its antibacterial evaluation for biomedical applications", Chem. Commun. 2959–2961.
- Augst. A.D, Kong. H.J, Mooney. D.J. (2006). "Alginate hydrogels as biomaterials", Macromol. Biosci, 6, 623.
- Seo. S.Y, Lee. G. H, Lee. S.G, Jung. S.Y, Lim. J.O, Choi. J.H. (2012). "Alginate-based composite sponge containing silver nanoparticles synthesized in situ", Carbohydr. Polym. 90, 109
- Liu. Y, Chen. S, Zhong. L, Wu. G. (2009). "Preparation of high-stable silver nanoparticle dispersion by using sodium alginate as a stabilizer under gamma radiation", Radiat. Phys. Chem. 78, 251.
- González. J.A, García. A, Rosemary. F. (1982). "Discharge properties of a dairy", j. agrochem. food tech.22, 501-510.
- Aspasia. A.C, Anastassios. G.S. (2012). "Biotechnological Utilization with a Focus on Anaerobic Treatment of Cheese Whey", Curr. Stat. Prosp. Energ. 5, 3492-3525.
- Sumaira. A, Azhar. Z.A, Christian. P, Syed. Z.H, Zafar. M.K, Pilar. R.G, Wolfgang. J. P, Irshad. H. (2013). "Protein-mediated synthesis, pH-induced reversible agglomeration, toxicity andcellular interaction of silver nanoparticles", Coll. Surf. B: Biointer. 102, 511– 518.
- Mingru Zhou, Zhiqiang Wei, HongxiaQiao, Lin Zhu, Hua Yang, and Tiandong Xia, Particle Size and Pore Structure Characterization of Silver Nanoparticles Prepared by Confined Arc Plasma, Journal of Nanomaterials, (2009) 2009:1-5.



