Immunological Characterization of Plasmodium Falciparum Malaria in Central Sudan Using GLURP
Munsoor Mohammed Munsoor1, *, Khalda Mirghani Hamza1, Abeer Eltuhami2, Ibrahim M. El-Hassan2
1College of Medical Laboratory Science, Sudan University of Science and Technology, Khartoum, Sudan
2Institute of Endemic Diseases, Faculty of Medicine, University of Khartoum, Khartoum, Sudan
Abstract
Introduction: Antigen-specific antibody-mediated immune responses play an important role in natural protection against clinical malaria, but conflicting estimates of this association have emerged from immuno-epidemiological studies in different geographical settings. This study was aimed at assessing in a standardized manner the relationship between the antibody responses to the most promising malaria vaccine candidate antigens, glutamate rich protein (GLURP) and protection from clinical malaria, in a cohort of Gazira inhabitants, central Sudan. Methods: Standardized ELISA protocols were used to measure IgG and IgG subclass levels to GLURP antigen in plasma samples from 133 central Sudan inhabitant aged 1to 80 years in a hospital based cross-sectional study. The levels of antibodies were related to the severity of malaria as indicated by anemia and parasitemia. The ELISA protocol follows AIA criteria to permit comparisons of results with those achieved in nearby African continents. Results: In the patients with parasitemia <5000 parasite/µl and normal hemoglobin, the results indicate high titres of the above antibodies to the three fragments of GLURP (RO, R1 and R2), although R2 and R0 encountered with significantly higher concentrations of the antibodies compared to patients with high parasitemia and anemia. The results also showed that IgG3 was presented with high concentration followed by IgG1 suggesting their protective role and their major participation in acquisition of immunity to malaria. The results also re-enforce the on going announcement that any future malaria vaccine should include GLURP fragments notably GLURP-R2 and GLURP-R0. Conclusion: The present study provided and presented here the first evaluation of humoral responses to recombinant GLURP in individuals living in Central Part of Sudan. This work also showed the gradual acquisition of immunity to malaria among individuals living in Gazira State in central Sudan. Finally, the study has confirmed the importance of antibodies to GLURP in reducing the risk of clinical malaria in Central Sudan, thus substantiating its potential as a malaria vaccine candidate.
Keywords
Malaria Immunity, Plasmodium Falciparum GLURP, Malaria in Central Sudan
Received: March 23, 2015
Accepted: April 10, 2015
Published on line: April 19, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CCBY-N C license .http://creativecommons.org/licenses/by-nc/4.0/
1. Background
Malaria remains one of the most important causes of morbidity and mortality in the world. Current methods of control are only partially effective and, therefore, the development of a vaccine which can provide a high degree of protection is a priority. Antibody-mediated immune responses to malaria antigens are known to be involved in protection against disease [1-4], but the antigens that induce protective antibodies have not been conclusively identified. Immuno-epidemiological studies from different laboratories have sometimes yielded conflicting results [5]. There is need to study immune responses to potential malaria vaccine in different epidemiological settings in endemic areas in order to determine the possible protection role of these antigens. Samples for the present study were retrospectively obtained from patients who attended Madani adult and pediatric hospitals complaining from malaria like symptoms, this region is considered as a mesoendemic malaria region and the highest transmission usually follows the rainy season which extended from July to October each year. The antigen selected for this study was the Glutamate Rich Protein (GLURP), this antigen was thought to induce protective antibody responses through various mechanisms [7-11]. Vaccines incorporating this antigen are currently in clinical trials and are described in detail else where [11]. It is likely that a future malaria vaccine will comprise multiple rather than single antigen and it is, therefore, useful to study natural immune responses to multiple malaria antigens in relation to incidence of malaria in a more standardized way. In this study, the standardized AIA ELISA procedures [6], were used to assess the relationship between anemia induced by clinical malaria and naturally acquired isotype and IgG subclass responses to the leading malaria vaccine candidate antigen, GLURP in Sudanese individuals living in Gazira. Ethical clearance for this work has been approved by the ethical committee of the institute of endemic diseases, faculty of medicine, University of Khartoum.
2. Materials and Methods
2.1. The Study Area
This study was cross-sectional hospital based study carried out in Madani, capital of Gazira state that located in the central part of Sudan. The city is located at latitude14.4 N and longitude 35.5 E. Plasmodium falciparum is the predominant parasite species, accounting for more than 90% of malaria parasite. Anopheles arabiensis is the main mosquito vector (12-13).
2.2. Study Population
Individuals recruited for the present study, were permanent residents of the city and surrounding villages who came to Medani paediatric and adult university teaching hospitals complaining from malaria symptoms. Clinical malaria is defined when blood film for malaria showed positive result and the measured body temperature was found to be ≥37.5°C(12).
2.3. Parasitological Examinations
Thin and thick blood films: Thin and thick blood films were prepared from blood samples collected from the individuals recruited for the study and processed according to the procedure reported earlier(14)
2.4. Hemoglobin Level
Hemoglobin level was estimated by Drabkin’s method as described by Dacie and Lewis(15).
Enzyme-linked immuno sorbent assay (ELISA): To ensure quality control, ELISA tests were done according to the criteria set by the African immuno-assays (AIA) and following the standard operating procedures that were kindly mailed to our laboratory by Daniel Dodoo (16).
2.5. Antibody Quantification
For antibody quantification, each assay includes a calibration (standard) curve obtained with a polyclonal reference IgG (1.0mg/ml). For the standard curve, serial dilutions (3-fold) of standard IgG proteins are coated directly to duplicate wells in columns 1 and 2 using 100µl/well. Concentrations of standard IgG are 1000; 333.3; 111.1; 37.0;12.3; 4.1; 1.4;0.5 ng/ml.
Negative controls.
The pools of negative samples have been selected from a panel of blood samples from Danish blood donors never exposed to malaria.
2.6. Analysis of the Data
Data were analyzed using SPSS version11.5 (Stata Corporation, Texas, USA; http://www.stata.com). The age dependence of antibody responses was analyzed by use of Kendall’s-tau-b, Pearson’s and Spearman's rank-order correlations. The mean matched-pairs test (Chi-square) was used to compare R0-GLURP IgG levels between samples. Frequencies of various variants were computed cross-tabulation, explore and percentages and means were compared by anova. The Mann-Whitney test was used to test R0-GLURP IgG class and subclasses levels between females and males. Univariate and multivariate models were fitted to estimate to what extent R0-GLURP IgG class and subclass levels could be attributed to protection against high parasite density, anaemia in individuals from the high transmission area. Differences were considered statistically significant if the 95% confidence interval was not overlapping or P ≤0.05 and p ≤.0.010 in some cases.
3. Results
3.1. Patient Characteristics
Based on the WHO criteria for clinical anemia, the Plasmodium faciparum patients were classified into severe anemic, mild anemic and patient with normal haemoglobin level (lookinghealthy) with Hb values of ≤ 7gm/dl, 8-11gm/dl and ≥12gm/dl respectively. Patients are also categorized into 4 age groups. Group 1 (0-5years) has 70 (52.65%) individuals, group 2 (6-10 years) has 40 (30.75%) individuals, group 3 (11-15 years) has 15 (11.50%) individuals and group 4 (>15years) has 8 (6.01%) individuals.
3.2. Parasitemia
Malaria parasitemia ranges from 900-16000 parasite per µl blood. The results show that the children below 5 years of age harbouring the most higher value of parasitemia (more than 15000 parasite/µl blood) with a frequency of 41 and constitutes 31.66% of the total samples studied (133). This is followed by age group 2, 3, and 4 with a mean parasitemia of 1123, 6826, and 5745 parasite/µl blood respectively (tables1).
Table 1. Mean parasitemia among anemic and normal subjects. Anemic subjects have statistically high parasitemia (p<0.01).
| Subjects | Number | MeanParasitemia | Pvalue |
| Anemic subjects (Hb <7-11gm/dl) | 31 | 11560 | |
| Healthy subjects (Hb ≥12gm/dl) | 102 | 11007 | 0.004 |
3.3. ELISAResults
The results of the present study show that, the IgG antibodies directed against the three GLURP regions show high level in malarious patients with normal Hb level compared with those levels found in severe and mild anemic patients (tables 2 and 3).
Table 2. Means of IgG antibodies (ng/l) directed against GLURPR0, R1 and R2 among study population.
| Anemia status | AntiR0 | AntiR1 | AntiR2 |
| Severe (Hb <7gm/dl) | 55668.2000 | 4336.5600 | 51588.3333 |
| Mild (Hb7-11gm/dl) | 35887.1596 | 5338.1383 | 58650.0366 |
| No anemia (Hb >12gm/dl) | 77805.3571 | 7498.9286 | 81225.4286 |
Table 3. Means of IgG subclasses (IgG1 and IgG3) antibodies (ng/l) directed against GLURPR0, R1 and R2 among study population.
| Anemia status | R0/IG1 | R1/IG1 | R2/IG1 | R0/IG3 | R1/IG3 |
| Severe (Hb <7gm/dl) | 27349.3600 | 20083.0000 | 37918.4400 | 27590.9600 | 18106.4800 |
| Mild (Hb7-11gm/dl) | 23338.5319 | 17817.5957 | 39463.1915 | 25036.2021 | 10916.1702 |
| No anemia (Hb >12gm/dl) | 31026.5000 | 34228.2857 | 47725.8571 | 38576.2857 | 48949.2143 |
Relation between R0-GLURP IgG levels and Plasmodium-associated anemia in Medani area (high- malaria transmission area).
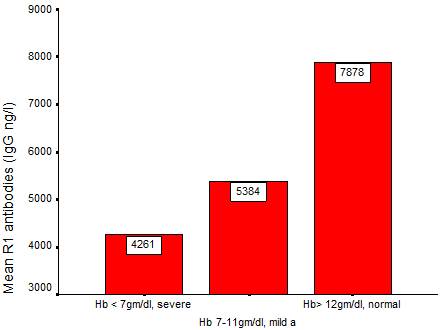
Fig. 1. Mean IgG antibodies directed against GLURP-R1 protien among anemia pateints.
To access the association between anti-GLURP antibodies and protection against malaria–related anemia, the mean antibodies were plotted against the anemia level. The data obtained showed that there were negative relation between the two parameters in a way that when one parameter increases the other decreases. Furthermore, the higher the prevalence of the severe anemia, the lowest the mean of the anti-GLURP antibodies and the lower the age of the subjects (fig.1).
4. Discussion
The goal of this of this study was to evaluate the antibody response induced by Plasmodium falciparum glutamate-rich protein (GLURP) in naturally exposed individuals from Medani region (Gazira State, Central Sudan). All antibodies (IgG, IgG1 and IgG3) tested against GLURP in this work show high concentration in the plasma of anemic patients compared to the concentrations of these antibodies in normal healthy individuals. The concentrations of these antibodies are variables and found to be dependent on a number of factors such as antigentype, age of patients and parasitemia (fig.1, tables 1, 2 and 3). The antibody of higher concentration found to be IgG, followed by IgG1 and IgG3 (tables 2 and 3). Several immuno-epidemiological studies using sera and clinical data from various sites have consistently identified high anti-R0-GLURP immunoglobulin G (IgG) levels as significant predictors of protection against high levels of parasitaemia, and febrile malaria episodes (17). The protective antibodies are thought to elicit antibody dependent cytotoxic inhibition (ADCI) (18 and 19) through binding to the surfaces of merozoites. The differences in antibody levels between malaria protected and non-protected individuals living in different regions of sub-Saharan Africa have been reported (20 and 21). This includes individuals naturally protected by life-long exposure to infection such as immune adults and those carrying protective mutations, such as hemoglobinopathies (21). The present study provided and presented here the first evaluation of humoral responses to recombinant GLURP in individuals living in Central Part of Sudan. The prevalence of GLURP specific IgG was markedly age-dependent. This is indicated by the positive correlation found between IgG1 directed against R1and IgG3 directed against R0 (fig. 1). However, this finding is comparable to those described in other African populations (15). Acquisition of antibodies to R0 and R2 sequences have been reported to be most frequent and with higher concentration compared to that related to R1 region (6). The present results were consistent with GLURP immune responses reported in other part of Africa (7 and 22). However, the results of the present study pointed to variable efficacy among the GLURP antigens. This was assessed by comparing the means of antibodies (IgG, IgG1 and IgG3) directed against the three sequences of GLURP. Among the IgG antibodies, there are variation with IgG1 and IgG3 are most prevalent and with the high concentration (table 2 and 3). Regarding the GLURP antigen, R0 and R2 antigens were encountered with high level of IgG antibodies and R1 was encountered with significantly decreased level of antibodies (tables 2 and 3).These observations are comparable with those reported in former studies and support the incorporation of R0 and R2 GLURP subunits in future GLURP vaccine formulations. In contrast with former reports, the acquisition of antibodies to the central repeat region, R1, was age-dependent (22). The results of the present and former studies suggest that, cytophilic IgG1and IgG3 antibodies against GLURP-R2 and R0-GLURP may contribute to the control of parasite multiplication and reduction in febrile malaria incidence in children living in an area of intense malaria transmission(6).
Acknowledgement
The authors are grateful for Dr. Daniel Dodoo for his great cooperation and Emailing of the AIA.
References
- Cohen S, McGregoria, Carringtons: (1961). Gamma-globulin and acquired immunity to human malaria. Nature 192:733-737.
- Cohen S, Butcher GA: (1971). Serum antibody in acquired malarial immunity. Trans R Soc Trop Med Hyg 65:125-135.
- Good MF: (1991). To wards the development of the ideal malaria vaccine. Adecade of progress in a difficult field. Med J Aust 154:284-289.
- Jepsen MP, Jogdand PS, Singh SK, Esen M, Christiansen M, Issifou S, Hounkpatin AB, Ateba-Ngoa U, Kremsner PG, Dziegiel MH, Olesen-Larsen S, Jepsen S, Mordmüller B, Theisen M. (2013). The malaria vaccine candidate GMZ2 elicits functional antibodies in individuals from malaria endemic and non-endemic areas. J Infect Dis. 208(3):479-88
- Soe S, Theisen M, Roussilhon C, Aye KS, Druilhe P: (2004). Association between protection against clinical malaria and antibodies to merozoite surface antigens in an area of hyperendemicity in Myanmar: complementarity between responses to merozoite surface protein 3 and the 220-kilodalton glutamate-rich protein. Infect Immun 72:247-252.
- Lusingu JP, Vestergaard LS, Alifrangis M, Mmbando BP, Theisen M, Kitua AY, Lemnge MM, Theander TG: (2005). Cytophilic antibodies to P. Falciparum glutamate rich protein are associated with malaria protection in an area of holoendemic transmission.Mala rJ.,4:48.
- Nebie I, Diarra A, Ouedraogo A, SoulamaI, Bougouma EC, Tiono AB, Konate AT, Chilengi R, Theisen M, Dodoo D, Remarque E, Bosomprah S, Milligan P, Sirima SB. (2008) Humoral responses to P. falciparum blood-stage antigens and association with incidence of clinical malaria in children living in an area of seasonal malaria transmission in Burkina Faso, West Africa. Infect Immun 76:759-766
- Meraldi V, Nebie I, Tiono AB, Diallo D, Sanogo E, Theisen M, Druilhe P, Corradin G, Moret R, Sirima BS: (2004). Natural antibody response to P. Falciparum Exp-1, MSP-3 and GLURP long synthetic peptides and association with protection. Parasite Immunol 26:265-272.
- Baumann A, Magris MM, Urbaez ML, Vivas-Martinez S, DuránR, Nieves T, Esen M, Mordmüller BG, Theisen M, Avilan L, Metzger WG (2012). Naturally acquired immune responses to malaria vaccine candidate antigens MSP3 and GLURP in Guahibo and Piaroa indigenous communities of the Venezuelan Amazon. Malar J.11:46.
- Hermsen CC, Verhage DF, Telgt DS, Teelen K, Bousema JT, Roestenberg M, Bolad A, Berzins K, Corradin G, Leroy O, Theisen M, Sauerwein RW: (2007). Glutamate-rich protein (GLURP) induces antibodies that inhibit invitro growth of P. Falciparum in a phase 1 malaria vaccine trial. Vaccine 25:2930-2940.
- Duru KC, Thomas BN (2014). Genetic Diversity and Allelic Frequency of Glutamate-Rich Protein (GLURP) in Plasmodium falciparum Isolates from Sub-Saharan Africa. Microbiol Insights.7:35-9.
- UN Millennium Project (2005). Coming to Grips with Malaria in the New Millennium. Task Force on HIV/AIDS, Malaria, TB, and Access to Essential Medicines, Working Group on Malaria. World Malaria Report 2005. Geneva. RBM/WHO/UNICEF, 2005.
- Haridi, A.M. (1972). Partial exophily of Anopheles gambiiae species B in the Khashm Elgirba area in eastern Sudan. Bulltin of World health Organization. 46:39-46.
- Gilles, H.M. (1993). The malaria parasites. In: Gilles, H.M. and Warrell, D.A. (ED.) Bruce Chwatts 'Essential Malariology, 3rdEdition. 13-34. Little brown & Co., USA
- Dacie, J.V, Lewis M., (1994). Practical Hematology, 8thed. Livingstone, London.
- Dodoo D., TheisenM , Kurtzhals JA, Akanmori BD, Koram K.A, Jepsen S, Nkrumah FK, Theander TG, Hviid L. (2000). Naturally acquired antibodies to the glutamate-rich protein are associated with protection against P. Falciparum malaria. J. Infect. Dis. 181,1202–1205.
- Dziegie lM, Borre MB, Jepsen S, Hogh B, Petersen E, Vuust J. (1991). Recombinant P. Falciparum glutamate rich protein; purification and use in enzyme-linked immune sorbent assay. Am J Trop Med Hyg. 44:306-313.
- Hogh B, Petersen E, Dziegie lM, David K, Hanson A, Borre M, Holm A, Vuust J, Jepsen S. (1992): Antibodies to a recombinant glutamate rich P. Falciparum protein: evidence for protection of individuals living in a holoendemic area of Liberia. Am J Trop Med Hyg .46:307-313.
- Theisen M, Roeffen W, Singh SK, Andersen G, Amoah L, vandeVegte-Bolmer M, Arens T, Tiendrebeogo RW, Jones S, Bousema T, Adu B, Dziegiel MH, Christiansen M, Sauerwein R. (2014). Amulti-stage malaria vaccine candidate targeting both transmission and a sexual parasite life-cycle stages. Vaccine. 7;32(22):2623-30.
- Ellman R, Maxwell C, Finch R, Shayo D. (1998). Malaria and anaemia at different altitudes in the Muheza district of Tanzania: childhood morbidity in relation to level of exposure to infection. Ann Trop Med Parasitol, 92:741-753.
- Cavanagh, D.R., Dodoo, D., Hviid L., KurtzhalsJA, Theander G., Akanmori, BD, Polley S, Conway DJ, KoramK, McBride JS. (2004). Antibodies to the N-terminal block 2 of P. Falciparum merozoite surface protein 1 are associated with protection against clinical malaria. Infect. Immun. 72,6492–6502.
- Dodoo D, Aikins A, Asamoah K, Lamptey H, Remarque E, Milligan P, Bosomprah S, Chilengi R, Osei Y, Akanmori B and Theisen M. (2008). Cohort study of the association of antibody levels to AMA1, MSP119, MSP3 and GLURP with protection from clinical malaria in Ghanaian children. Malaria Journal 7:142



