Pituitary Apoplexy Development During Bilateral Knee Replacement Surgery: Cause of Prolonged Altered Sensorium in the Postoperative Period
Guru Dutta Satyarthee1, *, V. S. Mehta2
1Department of Neurosurgery, All India Institute of Medical sciences, New Delhi, India
2Paras Institute of Neurosciences, Gurgaon, India
Abstract
Authors reports a case of pituitary apoplexy, which occurred in the incidental pituitary adenoma during bilateral knee replacement surgery. She developed right eye ptosis, headache and vomiting with alteration of sensorium in the immediate postoperative period with diminution of vision in the both eyes. Which was detected in the immediate postoperative period and CT and MRI scan showed a giant sellar-suprasellar lesion suggestive of pituitary adenoma with apoplexy. She underwent successful transsphenoidal surgery. Altered blood with necrotic pituitary adenoma was decompressed. Following surgery, her headache completely subsided, vision improved but right eye ptosis continued to persist during early follow-up period. To the best of knowledge current case is the first case in the western literature, who developed pituitary apoplexy during bilateral knee replacement surgery.
Keywords
Pituitary Apoplexy, Knee Replacement Surgery, Incidental Pituitary Adenoma, Precipitating Factors for Apoplexy
Received: August 18, 2015
Accepted: August 28, 2015
Published online: September 2, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Pituitary apoplexy is a clinical syndrome characterized by sudden onset severe headache, commonly associated with vomiting, signs of meningeal irritability, ophathalmoplegia and visual deterioration. 13Apoplexy mostly occurs spontaneously, although in some cases may have predisposing factors including endocrine manipulation, trauma, cerebral angiography, bromocriptine therapy,radiation therapy of pituitary adenoma, lumbar puncture, and in the immediate postoperative period. 5, 6, 7, 8, 11
2. Case Report
A 66 – year - old female underwent bilateral knee replacement surgery at our Institute for bilateral knee joint osteoarthritis. Her previous medical history included diabetes mellitus, which was controlled with oral hypoglycemic agents and knee joint osteoarthritis. Immediately after surgery, she presented with severe headache with repeated episodes of vomiting and altered sensorium. Examination revealed stable vital signs. Neurological examination revealed right sided third nerve paresis with bilateral visual acuity of perception of hand movement only. She had bitemporal field defect on bedside confrontation test, however, rest of the neurological examination was normal. The hematological and biochemical profile was normal and urine examination showed absent ketone bodies. Endocrinologist opinion was sought for monitoring of diabetes. The cranial CT scan revealed a giant pituitary adenoma, with heterogeneous areas of hyperdensity suggestive of recent haemorrhage (fig-1), on contrast administration showing fair enhancement. MRI brain, sagittal view on T1 weighted image revealed presence of giant mass in the sella with large suprasellar extension reaching up to the floor of third ventricle, causing obstructive hydrocephalus, showing scattered areas of hyperintense signal suggestive of pituitary apoplexy (Fig-2), and axial T2 weighted image (Fig-3) suggestive of recent bleed in the pituitary adenoma following pituitary apoplexy. There was also associated mild hydrocephalus. She underwent decompression of pituitary adenoma after adequate control of blood sugar level with insulin therapy. During surgery there was soft suckable grey mass with altered blood. Post-operative, computed tomography scan of head, axial section image in the post-operative period showing evidence of craniotomy and small residual admixed with blood was present in the operative cavity with subsidence of hydrocephalus. (fig-4) Histopathology revealed chromophobe pituitary adenoma with evidence of necrosis and haemorrhage. Immunohistochemistry revealed negative reactivity for growth hormone as well as prolactin. She showed improvement in the vision in the immediate postoperative period. Her vision improved to 6/18 in the right eye and 6/24 in the left eye. Her ptosis was still persisting at the last follow up after 3 months following surgery, with well controlled diabetes on lesser dosage of oral hypoglycemic agent.
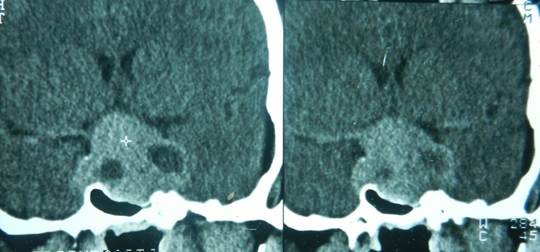
Fig. 1. Non-contrast computed tomography scan of head, coronal reconstruction showing heterogeneous areas with areas of hyperdensity suggestive of apopleptic pituitary lesion.

Fig. 2. Magnetic resonance image of brain T1W image, sagittal section showing mass in the sella with suprasellar extension reaching up to the floor of third ventricle, showing scattered areas of hyperintensity suggestive of pituitary apoplexy.

Fig. 3. Magnetic resonance image of brain T2W image, axial section showing mass in the sella showing areas of hyperintensity suggestive of pituitary apoplexy comparing to brain.

Fig. 4. Non-contrast computed tomography scan of head, axial section image in the post-operative period showing evidence of craniotomy and small residual mixed with blood in the operative cavity with subsidence of hydrocephalus.
3. Discussion
Pituitary apoplexy is a well-known clinical syndrome. 1-4, 6-8 Pituitary tumour is the intracranial neoplasm having bleeding frequency of 5.4 times higher than any other primary intracranial neoplasm.1The incidence of apoplectic presentation of pituitary adenoma varies in literature between 15 - 27.7 %. 2, 3, 12 It is a cascade of complex clinical events, occurring following fulminant expansion of a pituitary tumor by haemorrhage, infarction or combination of two, which involves tumour and neighbouring pituitary gland.4 There is a range of presentation of pituitary apoplexy, which may vary from sub-clinical infarction observed during the surgery or on preoperative imaging to full blown apoplexy that mimics subarachnoid haemorrhage.3Although most cases occur spontaneously, there are some apparent predisposing factors such as bromocriptine therapy,5head trauma, 6, 19 cerebral angiography7, endocrine manipulation, 8 coughing and sneezing,9radiation therapy of pituitary tumours10, 11 lumbar puncture, 13pneumocephalography13, and increased intracranial pressure.13 Weisberg et al11reported in a series of 14 cases of pituitary apoplexy, out of which eight cases developed apoplexy either during the course of radiotherapy or later on after completion of the treatment. All these cases were receiving radiotherapy as primary mode of treatment. Kato- et al 15reported occurrence of pituitary apoplexy in a known case of acromegaly, while undergoing subtotal thyroidectomy for large goiter. In a case report, Lennon et al 18 reported 51-year-old male, who underwent total hip replacement under spinal anesthesia developed headache, 3rd nerve paresis and electrolyte disturbance. On imaging study pituitary apoplexy was diagnosed. Cummina et al 16 reported delayed development of hypopituitarism without any clinical evidence of pituitary apoplexy. Patient was evaluated with neuro -imaging revealed pituitary apoplexy. The disturbances of CSF have been reported up to eight days after surgery. Cummina’s - al 16further concluded that the arterial hypotension during the coronary bypass surgery might be a precipitating factor for the delayed occurrence of pituitary apoplexy. Sugita 17 reported a case report of pituitary apoplexy in diabetic patient undergoing treatment for myocardial infarction. They postulated possibility of raised blood pressure by contrast agent might lead to permeability changes in pituitary portal circulation, with pre-existing diabetes mellitus resulting in vasculopathy which in turn may trigger apoplexy. Our patients was also known diabetic for last nine years on oral hypo-glycaemic agent. She had very severe headache along with repeated episodes of vomiting and progressive decline in visual acuity and ptosis with diabetes insipidus. Presence of focal neurological deficit, and alteration in sensorium, electrolyte imbalance and subtle presentation of pan-hypopituitarism led to suspicion of pituitary apoplexy and imaging revealed incidental pituitary adenoma with apoplexy. Perhaps this is the first case report of pituitary apoplexy, which occurred during the surgery in a diabetic patient who was harbouring asymptomatic pituitary adenoma.
Goel et al 14reported fatal pituitary apoplexy, in the residual tumour, after partial decompression following transcranial surgical approach in two cases. Rovit et al 4 reported higher incidence of apoplexy in patients in acromegalic or ACTH producing adenoma. Weisberg et al 11 also noticed higher incidence in acromegalic after radiotherapy. However, our case had a homophobe pituitary adenoma. Goel et al 14reported sudden release of feeding tumour vessel from internal carotid artery, compromise of venous drainage, tumour manipulation, swelling and compression of hypophyseal artery at diaphragma sella resulting in ischemia and secondary tumour necrosis leading t pituitary apoplexy. Ischemic necrosis of adenomatous tissue, compression of superior hypophyseal arteries against diaphgrama sella and intrinsic vasculopathy of pituitary tumour has been suggested.
Biousse et al. 20 concluded multiple factors act as precipitating factors of pituitary classified into four categories. First group: reduced blood flow to pituitary gland resulting from fluctuations in blood pressure like, hypotensive event cardiac surgery or lumbar laminectomy, are associated with pituitary apoplexy as also even transient rise in the intracranial pressure with resultant reduction in blood flow of the pituitary gland during sneezing and coughing. Second group is sudden rise in the blood flow to pituitary is also considered triggering factor. Diabetes or chronic hypertension also causes degenerative changes microvasculature. However, diabetic keto - acidosis and hypertension may precipitate apoplexy Next group is pituitary gland stimulation by excessive oestrogen level by intake of pills, pregnancy, or bromocriptine therapy. 20 Fourth group comprises anticoagulation therapy as anticoagulant drug administration, thrombolytic agents, or reduced platlet count is also associated with apoplexy. However, our case was a known case of a diabetes mellitus with uneventful perioperative period with un-associated any hypotensive or hypoxaemic event, however, a combination of factors are responsible for apoplexy in the current case.
4. Conclusion
Every patient with history of intense headache, fever, altered sensorium, electrolyte disturbances, focal neurological deficits including worsening of visual acuity and early feature of pan-hypopituitarism a possibility of pituitary apoplexy must be entertained and appropriate early radiological investigation must be carried out. Surgical decompression preferably through transsphenoidal approach is advisable. The tumour decompression should be carried out as emergency procedure, as it can control further injury and also helps in preservation of vision and can prevent its associated morbidity.
References
- Wakai S, Yamakawa K, Manaka S, et al. Spontaneous intracranial hemorrhage caused by brain tumor: its incidence and clinical significance. Neurosurg 1982; 10: 437-444.
- Cardoso ER, Peterson EW. Pituitary Apoplexy –a review. Neurosurg 1984; 14: 363-373.
- Anderson JR, Anton N, Burnet N, et al. Neurology of the pituitary gland. J Neurol Neurosurg Psychiatry 1999; 66: 703-721.
- Rovit RL, Fien JM. Pituitary apoplexy: a review and reappraisal. J Neurosurg 1972; 37: 280-288.
- Mc Gregor AM, Scalon MF, Hall K, et al. Reduction in size of tumor by bromocriptine therapy. N England J Med 1979; 300: 291-193.
- Lopez IA. Pituitary apoplexy. J Oslo City Hospital 1970; 20: 17-27.
- Stiemle R, Royeer J, Opperman A, et al. Hematome post-angiographique dans un adenoma de I’hypophyse: cecite et troubles oculomoteurs regressant après intervention d’urgence Neurochirurgie 1974; 20: 599-608.
- Masago A, Ueda Y, Kanai H, Nagai et al. Pituitary apoplexy after pituitary function test: A report of two cases and review of the literature. Surg Neurol 1995; 43:158-165.
- Dawson BH, Kothandram P. Acute massive infarction of pituitary adenomas. A study of five patients. J Neurosurg 1972; 37: 275-279.
- Onseti ST, Wisniewski T, Post KD. Clinical versus sub clinical pituitary apoplexy: presentation, surgical management, and outcome in 21 patients. Neurosurg 1990; 26: 980-986.
- Weisberg LA. Pituitary apoplexy: association of degenerative changes in pituitary adenoma with radiotherapy and detection by cerebral computed tomography. Ame J Med 1977; 63:109-115.
- Motta LACR, Mello PA, Lacerda CM, et al. Pituitary apoplexy: clinical course, endocrine evaluations and treatment analysis. J of Neurosci 1999; 43:25-36.
- David NJ, Gargano FP, Glaser JS. Pituitary apoplexy in clinical perspective. Neuroophthalmol 1975; 8: 140-165.
- Goel A, Deogaonkar M, Desai K. Fatal postoperative pituitary apoplexy: its causes and management Brit J Neurosurg 1995; 9:37-40.
- Powell M. Recovery of vision following transsphenoidal surgery for pituitary adenomas. Brit J Neurosurg 1995; 9:367-373.
- Bills DC, Meyer FB, Laws Jr ER, et al. A retrospective analysis of pituitary apoplexy. Neurosurgery, 1993; 33:602-609.
- Sugita S, Hirohaya M, Tokutomi T, et al. case of pituitary apoplexy in a child. Surg Neurol 1995; 4:154-157.
- Mohr G, Hardy J. Hemorrhage, necrosis and apoplexy in pituitary adenoma. Surg Neurol 1982; 18:181-189.
- Ebersold MJ, Laws ER, Scheithauer BW, et al. apoplexy treated by transsphenoidal surgery, A clinicopathological and immunohistocytochemical study. J Neurosurg. 1983; 58: 315-320.
- Biousse V,Newman NJ,Oyesiku NMPrecipitating factors in pituitary apoplexyJ Neurol Neurosurg Psychiatry.2001 Oct;71(4):542-5.



