Nitric Oxide and Interleukin-6 Production in Patients with Transient Cerebral Microcirculatory Disturbances
Maria N. Starodubtseva1, *, Natallia V. Halinouskaya2, Nadezhda M. Halubykh3, Evgenii V. Voropaev3, Vasiliy B. Smychek4
1Department of Medical and Biological Physics, Gomel State Medical University, Gomel, Belarus
2Department of Neurology and Neurosurgery, Gomel State Medical University, Gomel, Belarus
3Research Laboratory, Gomel State Medical University, Gomel, Belarus
4National Science and Practice Centre of Medical Assessment and Rehabilitation, Gorodische, Belarus
Abstract
Reactive nitrogen species are well known to be important participants of cerebral stroke development. Low concentration of NO is produced by constitutive NO-synthase under physiological conditions, whereas high concentration of NO is produced by inducible NO-synthase of the stimulus-activated cells of the immune system and endothelium. Based on the concept of two cell states (resting and activated) with different NO production rates we have analyzed NO metabolite (NOx) concentration in plasma of patients with transient cerebral microcirculatory disturbances (TCMD) simultaneously with the levels of inflammatory markers (superoxide dismutase activity and concentration of interleukins (IL-6 and IL-8) and C-reactive protein) and studied the effect of a standard 10-day therapeutic course on them. The sample of patients with TCMD included 24 patients with transient ischemic attack (TIA) and 19 patients with acute hypertensive encephalopathy (AHE). The NOx concentration was measured by the modified Griess method. The presence of two groups with low and high NOx concentration in plasma was detected for the studied sample both in the beginning and in the end of patient’s hospital stay (high NOx plasma samples were found in 18% of cases on Day 1 and 27% of cases on Day 10). The majority of subjects with high plasma NOx concentration on Day 1 were AHE patients (63%) and on Day 10 were TIA patients (78%), correspondingly, The high NOx plasma samples were associated with low superoxide dismutase activity (p=0.045) and increased IL-6 level (p=0.003). Our study’s results suggest that the analysis of NOx concentration changes during a therapeutic course is useful for improving the diagnosis and future prognosis of patients with TCMD.
Keywords
Stroke, Cerebral Hypertension, Transient Ischemic Attack, Peroxynitrite, Nitrite/Nitrate Ions, Interleukin-6, C-Reactive Protein
Received: July 7, 2015
Accepted: July 26, 2015
Published online: August 6, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Stroke is one of the leading causes of death and acquired adult disability in industrialized countries (Kernan et al., 2014; van Rooy and Pretorius, 2015). A transient ischemic attack (TIA) early defined as an episode of neurologic dysfunction caused by focal cerebral ischemia with complete recovery within 24 hours has been classified as ischemic stroke since 2009 (Kernan et al., 2014; Gupta et al., 2014). Transient disturbances of cerebral blood microcirculation can take place in the result of the loss of blood flow through the small vessels via ether their occlusion with emboli as in case of TIA or vasoparalysis caused by cerebral hypertension. The second form of cerebrovascular pathology is known as acute cerebral vascular insufficiency or acute hypertensive encephalopathy (AHE). AHE clinically manifests as headache, nausea, vomiting, confusion, in some cases, transient hemiparesis, aphasia, hemihypestesia. The main mechanism of these cerebral pathologies is based on the phenomenon of cerebral ischemic/reperfusion injury (CIRI). In the considered case, the extent of cell dysfunction, injury and death is influenced by both magnitude and duration of ischemia. Reactive oxygen and nitrogen species (ROS/RNS) are important participants of CIRI pathways (Carden and Granger, 2000). During ischemia and after reperfusion concentration of ROS/RNS in cells and tissues increases (Szabo, 2000; Pacher et al., 2007; Jiang et al., 2014). Depending on their concentration, ROS/RNS can be regulators of cellular pathways as well as cytotoxic agents that exacerbate ischemic injury (Carden and Granger, 2000). ROS/RNS at high concentration in the brain are produced basically by the activated cells of the immune system (neutrophils, macrophages, lymphocytes), glia, neurons and endothelium. They can regulate all the functional responses of the cells including generation of numerous cytokines and other molecules considered as "inflammatory markers" (Jickling et al., 2012). Determination of RNS role in the development of cerebrovascular pathology is one of current problems of brain science. RNS include nitrogen monoxide (NO) and its reactive derivatives among them nitrogen dioxide (NO2) and peroxynitrite (ONOO¯, HOONO). The quasi-stable products of RNS metabolism in tissues are nitrite and nitrate ions (NOx) (Pacher et al., 2007).
The aim of the present study was to determine the concentration of NOx and inflammatory markers such as interleukin-6 (IL-6), interleukin-8 (IL-8), and C-reactive protein (CRP) in plasma of patients with transitory cerebral microcirculation disturbances, and investigate the effect of a standard 10-day therapeutic course on them.
2. Research Significance
On analyzing the groups of patients with transient cerebral microcirculatory disturbances (transient ischemic attack or acute hypertensive encephalopathy) with low and high concentration of nitrite and nitrate ions (NOx) in plasma in the beginning and in the end of patient’s hospital stay, we revealed that the plasma NOx concentration dynamics was dependent on diagnosis. The high plasma NOx concentrations were associated with high rate of peroxynitrite formation in patients due to low superoxide dismutase activity and increased IL-6 level in plasma. Determination of the time dependence of plasma NOx concentration can be useful for improving the diagnosis and future prognosis of patients with transient cerebral microcirculatory disturbances.
3. Methods
Patient selection. The studied population included 44 patients admitted to the Stroke Unit of the Gomel Regional Veteran’s Hospital: 24 patients with TIA (11 women, 13 men; mean age 66.7±2.0 years (CI)) and 19 patients with AHE (14 women and 5 men, mean age 60.6±2.7 years (CI)) without visible changes in the brain structure on MRA and CT images. Neurological deficit of TIA patients on Day 1 of their hospital stay was presented by hemiparesis (46.8%), asymmetry of nasolabial folds (72.3%), instability in the Romberg position (76.6%), asymmetry of tendon reflexes, Babinski symptom (57.4%), and hypoesthesia (34.0%). TIA subjects had a mean ABCD2 score of 4.9 (4-6) (Me (IQR)). For patients with AHE, hemiparesis (14.0%), hypoesthesia (37.2%), static and kinetic insufficiency (74.1%), asymmetry of nasolabial folds (25.6%), asymmetry of tendon reflexes and Babinski symptom (25.6%) were characteristic. All the patients were on the diet with restriction of water and vegetables with high content of nitrate. They were not allowed nitrate-containing medications or permitted to smoke during the study period. The patients enrolled in the present study were examined and treated according to the Protocols of diagnosis and treatment approved by the Ministry of Health of the Republic of Belarus. The study was approved by the Ethic Committee of the Gomel State Medical University.
Blood plasma samples and biochemical measurements. Venous blood samples from the patients were taken at 8:00 AM on Day 1 and Day 10 of their hospital stay. The concentration of IL-8, IL-6, and CRP were measured in EDTA-anticoagulated plasma using enzyme-linked immunosorbent assays (ELISA) according to the manufacturer’s instructions ("Vector-Best", Russia). Normal ranges for IL-8, IL-6 and CRP were 0-10.0 pg/ml; 0-5.0 pg/ml; and 0-10.0 g/l, respectively. Concentration of nitrite/nitrate ions in the samples was determined by the method based on the Griess reaction. A total of 300 ml plasma was diluted with 600 ml 96% ethanol, incubated during 1 hour (20°C) and centrifugated at 18000g (4°C) for 2 min. The supernatant (500 ml) was mixed with 200 ml of the Griess reagent mixture: 2 volumes of VCl3 (8 g/l in 0.1 M HCl), 1 volume of 0.1% N-naphthyl-ethylenediamine in deionized water and 1 volume of 0.35% 4,4’-diaminodiphenylsulfone in 2M HCl) and kept at 37°C for 30 min. Absorbance (A) of the supernatants was measured at 550 nm with Sunrise photometer (Tecan). NOx concentration was determined from a calibration curve plotted using sodium nitrite standards. Superoxide dismutase (SOD) activity was measured using the adrenaline auto-oxidation method (Sirota et al., 2000). Briefly, 0.1 ml adrenaline hydrochloride was added to 2 ml of 0.2 M carbonate buffer (рН 10.55), and the absorbance of the mixture was recorded as a function of time at 347 nm with Solar RV1251 spectrophotometer (Belarus). The rate of adrenaline auto-oxidation was determined in the absence (v0=dA0/dt) and in the presence (v=dA/dt) of 2 ml of blood plasma. SOD activity was calculated using formula (in activity units, a.u): SOD activity=[1-(v/v0)]×100.
Statistical analysis. The fit of data distribution to a normal distribution was checked by Shapiro-Wilk’s W test. The NOx concentration distribution was fitted to the curve that represents a sum of two Gaussian functions (R2 is a correlation coefficient for the fitting). The data in the table and figures are represented as median and the limits of the interquartile range: Me (IQR). The comparisons between the samples were performed by Mann-Whitney U test.
4. Results and Discussion
|
а |
b |
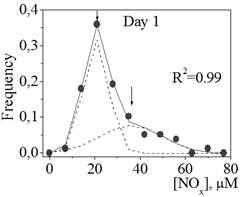
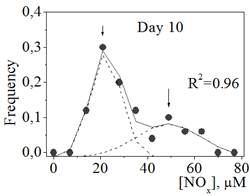
Figure 1. The distribution of NOx concentration in blood plasma samples of patients with transitory cerebral microcirculatory disturbances in the beginning (Day 1, a) and in the end (Day 10, b) of their hospital stay.
Circuits represent the experimental points; lines represent the results of fitting the distribution to the sum of two Gaussian functions drawn as dot lines.
Figure 1 represents the distributions of NOx concentration in the blood plasma samples of patients with transitory cerebral microcirculatory disturbances in the beginning (Day 1) and in the end (Day 10) of their hospital stay. The distributions are not normal (non-Gaussian distributions with positive skewness, Sapiro-Wilk test: W=0.89, p<0.05; W=0.92, p<0.05 for Day 1 and Day 10, correspondingly). The distributions were fitted with the sum of two Gaussian functions with high correlation coefficients (Figure 1, a and b), which corresponded to the presence of two different groups in the general population: group A with low NOx concentration in plasma (about 20 mM) and group B with high NOx concentration in plasma (about 40-50 mM). For groups A and B on Day 1 we did not found a statistically significant difference (difference test) in the frequency of occurrence of such concomitant pathologies as type 2 diabetes, arterial hypertension, myocardial infarction, coronary heart disease, cardiosclerosis, circulatory insufficiency, urolithiasis, and cilliary arrhythmia.
Because of our controlling patient’s nitrate/nitrite intake, we assumed that the changes in the concentration of nitrate/nitrite ions in plasma had endogenous base and related to the different states of the immune systems and endothelium: the state of activation and relative rest. In the activated state, the immune and endothelial cells produce high concentrations of NO via the activation and expression nNOS and iNOS (Szabo, 2003; Pacher et al., 2007). In the relative rest state, NO is synthesized mostly by eNOS at a ten-fold lower rate comparing to the NO production rate for iNOS. NO synthesis with high rate is accompanied, as a rule, by the high production level of reactive oxygen species (ROS) and is related to a low level of antioxidative defense in tissues. We have revealed the specific relationship between the NOx concentration and superoxide dismutase (SOD) activity in blood plasma. In general, concentration of nitrite/nitrate ions in plasma increases with decreasing the activity of SOD (Figure 2). All the plasma samples with SOD activity higher than 50 a.u. were the samples from group A (the state of stable relative rest). With decreasing SOD activity less than 45 a.u. the average NOx concentration and its variance were increased (the activated state). Therefore, the obtained results revealed high probability of peroxynitrite formation in tissues of an organism in a state with high NOx concentration in blood plasma (group B) (Kar and Kavdia, 2013). Peroxynitrite is a strong oxidizing agent modifying the structure of lipids, nucleic acids and proteins, which leads to functional and structural changes in cells and tissues (Szabo, 2003; Pacher et al., 2007). The mechanisms of the peroxynitrite reactions with biologically important molecules mostly involve a free radical formation stage. The rate of peroxynitrite production in an organism can achieve 50-100 μM per min (Szabo, 2003). The peroxynitrite effect on the cells and tissues depends on its concentration (Starodubtseva et al., 2010). Peroxynitrite at low concentrations serves as a signal molecule of the different pathways (brain activity, pain signaling, vascular tone regulation and others). At high concentration, peroxynitrite causes mostly cytotoxic effect. Reactive nitrogen species (RNS), including NO and peroxynitrite, play important roles in the process of cerebral ischemia-reperfusion injury. RNS trigger numerous molecular cascades, lead to the disruption of the blood brain barrier and exacerbate brain damage (Chen et al., 2013).
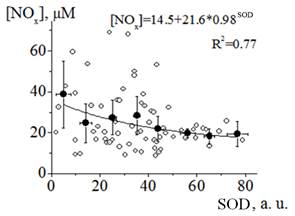
Figure 2. Relationship between NOx concentration and SOD activity (SOD) in plasma samples of patients with transitory cerebral microcirculatory disturbances.
Experimental points are represented by open circuits, black circuits represent the results of averaging values within the range of 10 a. u. of SOD activity and the black line shows the result of fitting the obtained dependence to an asymptotic function (the formula is written in the figure).
For the further analysis, we divided the studied population into the groups with low and high NOx concentration in blood plasma, samples A and B, correspondingly. The boundary value of NOx concentration for the group separation was admitted as 33 mM. The errors of wrong classification of groups with low and high NOx concentrations in plasma were 1% and 41% for the plasma samples taken on Day 1 and 5% and 12% for Day 10, correspondingly. On comparing inflammatory marker levels in A and B samples, we revealed the significant difference between SOD activities (p=0.045) and concentrations of IL-6 (p=0.003) for these two groups. Meanwhile, if the decreasing in SOD activity with transition of a patient from group A to group B was only about 20%, the increase in the concentration of IL-6 was more than two-fold (figure 3). Therefore, in the state associated with high rate production of peroxynitrite, the concentration of pro-inflammatory cytokines IL-6 is increased. This fact is accord with literature data that pointed to peroxynitrite-induced dose-dependent release of pro-inflammatory cytokines including IL-6, for example, by human monocytes (Matata and Galinanes, 2002).
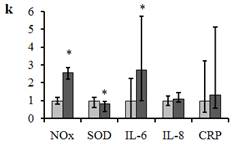
Figure 3. Relative change (k) in the inflammatory marker levels for the samples with low and high [NOx] (samples A and B, correspondingly).
Parameter k calculated as the ration between inflammatory marker levels for A and B samples (for example, k=[IL-6]B/[IL-6]A for IL-6). Samples A (n=78) and B (n=22) are denoted in light gray and in dark gray correspondingly. Data are presented as Me (IQR). *p<0.05 versus sample A.
For the analysis of changes in inflammatory marker levels in plasma of our patients during the period of a standard therapeutic course, we have considered four groups: group with low values of NOx concentration in plasma in both the beginning and end of investigation (group AA); group with increasing the values of NOx concentration (group AB); group with high values of NOx concentration in plasma on both Day 1 and Day 10 (group BB); group with decreasing the values of NOx concentration (group BA). The averaged concentrations of the inflammatory markers for all the groups are represented in Table 1. The group AA with relatively stable NOx concentrations during the hospital stay had the stable relative high level of SOD activity and stable low levels of IL-6, IL-8 and CRP. This group includes the comparable numbers of TIA and AHE patients. The groups AB and BA with either increasing or decreasing NOx concentration in plasma during the hospital stay showed, correspondingly, the decrease/increase in SOD activity level. The decrease in SOD activity for group BA versus group AA on Day 1 was significant (p<0.05, table). This group consists of only AHE patients. If in the beginning of a therapeutic course, the high production of peroxynitrite took place, in the course end all the studied inflammatory marker levels were within the ranges of group AA. Group AB is characterized by increase in IL-6 level (p<0.05) during the hospital stay. In scant group BB, at high level of IL-6 we had revealed the significant increase in the average concentration of CRP on Day 10 (p<0.05). These two groups mostly consist of TIA patients (75%). Because of the increase of NOx level or maintenance of high NOx level with simultaneously increasing IL-6 or CRP levels during a standard therapeutic course, the patients from these groups have a high risk of stroke development in near future (Whiteley at el., 2009; Cieślak et al., 2013).
Table 1. Concentration of NOx and inflammatory markers in plasma samples of the studied groups.
| Group | AA | AB | ||
| n | 26 | 9 | ||
| Age | 59.5 (50.0; 73.0) | 67.0 (66.0; 77.0) | ||
| Day | 1 | 10 | 1 | 10 |
| [NOx] | 16.9 (11.9;20.5) | 20.3 (15.1;24.3) | 23.6 (16.5;29.3) | 48.8 (48.0;54.2) |
| SOD | 37.8 (23.0;59.5) | 40.2 (26.5;46.6) | 36.8 (25.2;42.2) | 34.2 (8.8;34.3) |
| CRP | 8.7 (5.3;32.7) | 15.6 (6.1;27.7) | 8.83 (8.19;55.23) | 12.10 (8.91; 35.61) |
| IL-6 | 0.41 (0.00;1.06) | 0.49 (0.00;1.36) | 1.38 (0.53;1.58) | 2.10* (1.05;4.03) |
| IL-8 | 5.81 (5.00;7.49) | 6.53 (4.27;7.42) | 5.66 (4.16;6.99) | 6.56 (5.29;8.24) |
| Diagnosis | 15 TIA, 11 AHE | 6 TIA, 3 AHE | ||
Table 1. Continuous.
| Group | BB | BA | ||
| n | 3 | 5 | ||
| Age | 81.0 (68.0;89.0) | 62.0 (55.0; 71.5) | ||
| Day | 1 | 10 | 1 | 10 |
| [NOx] | 53.2 (34.6;53.5) | 43.8 (33.1; 45.5) | 42.7 (41.4; 43.5) | 21,0 (18,0; 23,1) |
| SOD | 37.2 (15.0;43.4) | 38.6 (18.6; 40.0) | 23.1* (11.3; 30.7) | 33,3 (26,5; 43,0) |
| CRP | 44.61 (7.67;61.47) | 310.17*# (78.77; 318.78) | 8.97 (6.16; 24.28) | 8,12 (3,25; 84,76) |
| IL-6 | 1.38 (0.49; 3.41) | 3.41# (0.08; 67.39) | 1.05 (0.57; 1.14) | 1,05 (0,70; 1,38) |
| IL-8 | 8.58 (5.06; 9.91) | 6.21 (5.87; 27.72) | 5.23 (3.49; 5.75) | 5,12 (4,11; 17,41) |
| Diagnosis | 3 TIA | 5 AHE | ||
n is a sample size, [NOx] is concentration of nitrite/nitrate ions in mM, SOD is activity of superoxide dismutase in a. u., CRP is concentration of C-reactive proteins in mg/l, IL-6 and IL-8 are concentrations of interleukins 6 and 8 in pg/l. Data are presented as Me(IQR).
*р<0.05 versus group AA (Day 10), #р<0.05 versus group AB, Mann-Whitney U t-test.
5. Summary and Conclusions
We have investigated how NOx concentration was distributed in the plasma samples of patients with transitory cerebral microcirculatory disturbances, and studied its dynamics during their hospital stay. Taking into consideration our control of nitrite and nitrate intake with food, water, medicaments and tobacco smoke, we suggested that a human organism could be in one of two possible states with low or high NOx concentration in blood that related to the states of the relative rest and activation of the endothelium/immune system cells. In case of transient cerebral microcirculatory disturbances, 30-40% of the patients could be in the second state (in the activated state). According the obtained data, the state with high NOx concentration in blood plasma is characterized by the decreased level of the tissue antioxidative defense. That fact means that in the considered state there are conditions for formation of highly reactive nitrogen species, peroxynitrite. The standard therapeutic course for the patients with transitory cerebral microcirculatory disturbances did not results in returning the patients back to the stable relative rest state of the endothelium/immune system in 30% of cases (groups AB and BB) and led to further activation of those system with an inhibition of the cellular antioxidative enzymes and enhancement of immune system responses. We suppose that the patients with high NOx plasma concentration in the end of their therapeutic course have a high risk of the stroke development in near future.
List of Notations, Definitions, and Symbols
Transitory ischemic attack, TIA; acute hypertensive encephalopathy, AHE; cerebral ischemic/reperfusion injury, CIRI; reactive oxygen species, ROS; reactive nitrogen species, RNS; interleukin-6, IL-6; interleukin-8, IL-8; C-reactive protein, CRP; interquartile range, IQR; superoxide dismutase, SOD; arbitrary units, a.u., confidence interval, CI.
Acknowledgments
Financial support from the State Scientific Research Program "Medicine and Pharmacy" (Belarus) and the Belarusian Republican Foundation for Fundamental Research (M13-081) is gratefully acknowledged.
References
- Carden DL, Granger DN: Pathophysiology of ischemia-reperfusion injury. J Pathol 2000, 190:255-266.
- Chen X, Chen H, Xu M, Shen J: Targeting reactive nitrogen species: a promising therapeutic strategy for cerebral ischemia-reperfusion injury. Acta Pharmacologica Sinica 2013, 34: 67-77.
- Cieślak M, Wojtczak A, Cieślak M: Relationship between the induction of inflammatory processes and infectious diseases in patients with ischemic stroke. Acta Biochimica Polonica 2013, 60(3): 345–349.
- Gupta HV, Farrell AM, Mittal MK: Transient ischemic attacks: predictability of future ischemic stroke or transient ischemic attack events. Ther Clin Risk Manag 2014, 10:27-35.
- Jiang Z, Li C, Arrick DM, Yang S, Baluna AE, Sun H: Role of nitric oxide synthases in early blood-brain barrier disruption following transient focal cerebral ischemia. PLoS ONE 2014, 9(3):e93134.
- Jickling GC, Zhan X, Stamova B, Ander BP, Tian Y, Liu D, Sison SM, Verro P,Johnston SC, Sharp FR.: Ischemic transient neurological events identified by immune response to cerebral ischemia. Stroke 2012, 43:1006-1012.
- Kar S, Kavdia M: Endothelial NO and O2− production rates differentially regulate oxidative, nitroxidative, and nitrosative stress in the microcirculation. Free Rad Biol Med 2013 63 (2013) 161-174.
- Kernan WN, Ovbiagele B, Black HR, Bravata DM, Chimowitz MI, Ezekowitz MD, Fang MC, Fisher M, Furie KL, Heck DV, Johnston SC, Kasner SE, Kittner SJ, Mitchell PH, Rich MW, Richardson D, Schwamm LH, Wilson JA; American Heart Association Stroke Council, Council on Cardiovascular and Stroke Nursing, Council on Clinical Cardiology, and Council on Peripheral Vascular Disease: Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2014, 45:2160-2236.
- Matata BM, Galinanes M: Peroxynitrite is an essential component of cytokines production mechanism in human monocytes through modulation of nuclear factor-kappaB DNA binding activity. J Biol Chem 2002, 277(3): 2330-2335.
- Pacher P, Beckman JS, Liaudet L: Nitric oxide and peroxynitrite in health and disease. Physiol Rev 2007, 87:315-424.
- Sirota TV, Lange NV, Kosiakova NL, Vanichkin AV, Kondrashova MN: New application of adrenalin oxidation reaction for estimation of pro-anti-oxidant properties of bioactive substances. Curr Topics Biophys 2000, 24(2): 185-189.
- Starodubtseva MN, Kavalenka AI, Petrenyov DR, Cherenkevich SN: Effects of peroxynitrite on morphology and functions of neutrophils. Handbook of free radicals: formation, types and effects. еd.: D. Kozyrev, V. Slutsky. Hauppauge, New York: Nova Science Publishers Inc 2010, 478-503.
- Szabo C: Multiple pathways of peroxynitrite cytotoxicity. Toxicol. Lett 2003, 140-141:105-112.
- Van Rooy MJ, Pretorius E: Metabolic syndrome, platelet activation and the development of transient ischemic attack or thromboembolic stroke. Thromb Res 2015, 135(3):434-442.
- Whiteley W, Jackson C, Lewis S, Lowe G, Rumley A, Sandercock P, Wardlaw J, Dennis M, Sudlow C: Inflammatory markers and poor outcome after stroke: a prospective cohort study and systematic review of interleukin-6. PLoS Med 2009, 6(9): e1000145.