Squamous Cell Carcinoma Arising in a Radicular Cyst: Case Report
Parviz Deihimy1, Neda Karghahi2, Saeedeh Khalesi3, *
1Torabinejad Dental Research Center, Department of Oral and Maxillofacial Pathology, Scholar Dentistry, Isfahan University of Medical Sciences, Isfahan, Iran
2Dental Implants Research Center, Department of Oral and Maxillofacial Pathology, Scholar Dentistry, Isfahan University of Medical Sciences, Isfahan, Iran
3Department of Oral and Maxillofacial Pathology, School of Dentistry, Shahid Sadoughi University of Medical sciences, Yazd, Iran
Abstract
Introduction: Primary intraosseous squamous cell carcinoma (PIOSCC) is a rare malignant lesion arising within the jawbones, without initial connection with the oral mucosa. This malignant lesion develops from odontogenic epithelium or odontogenic cysts or tumors. Case Report: In this case report, an intraosseous squamous cell carcinoma arising from an odontogenic cyst in a 60-year-old male patient is presented. Conclusion: This case report demonstrates the importance of clinician’s knowledge of malignant potential of odontogenic cyst and the accurate and timely management and treatment of these lesions.
Keywords
Squamous Cell Carcinoma, Radicular Cyst
Received: April 6, 2015 / Accepted: April 19, 2015 / Published online: May 11, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Primary intraosseous squamous cell carcinoma (PIOSCC) is a rare malignant lesion arising within the jawbones, without initial connection with the oral mucosa. This malignant lesion develops from odontogenic epithelium or odontogenic cysts or tumors (1). Malignant change in the epithelial lining of odontogenic cysts is a very rare, but these changes have been described in the literature (2-5).
According to the World Health Organization classification (1992), malignant variants of odontogenic epithelial tumors, which are PIOSCC and malignant changes in odontogenic cysts, are classified as odontogenic carcinomas (6). Furthermore, PIOSCC is subcategorized into 3 types including solid type, SCC derived from karatocystic odontogenic tumor, and SCC derived from the lining of the lining of odontogenic cysts (1). Subsequently, Waldron and Mutoe was changed the classification odontogenic carcinoma (Table 1) (7). Most frequency of odontogenic cyst has been associated with odontogenic carcinomas, including residual cysts, dentigerous cysts, calcifying odontogenic cysts, and lateral periodontal cysts (8). For diagnosis of PISCC require to some criteria including the absence of ulceration on the overlaying mucosa and the exclusion of distant metastasis from primary malignant tumor detection by physical and radiographic examination (9). Furthermore, the detection of early dysplastic change of benign epithelial lining of an odontogenic cyst to squamous cell carcinoma is necessary for the diagnosis of PIOSCC arising in odontogenic cyst (3).
Table 1. Waldron and Mustoe’s classification of odontogenic carcinoma (7).
| Type 1 | PIOSCC ex odontogenic cyst |
| Type 2A | Malignant ameloblastoma |
| Type 2B | Ameloblastic carcinoma arising de novo, ex ameloblastoma, or ex ameloblastoma |
| Type 3 | PIOSCC arising de novo |
| Type 4 | Intraosseous mucoepidermoid carcinoma |
In this report, we present a case of squamous cell carcinoma arising in a radicular cyst, and demonstrate the clinical, radiological, and histological features of this lesion.
2. Report of a Case
A 60-year-old male was referred to the Isfahan University of Medical Sciences, Faculty of Dentistry, Department of Oral and Maxillofacial surgery on 17 December, 2014 with a chief complaint of mobility of mandibular anterior teeth for two month duration. He had not a past history of tobacco or alcohol use and no traumatic injuries. But, there was a history of increase blood pressure and taking Terazosin. According to the patient statement, there was no sign of pain, sensation disorders, pus discharge, and bleeding in the involved region.
Extra oral examination showed normal, but the bilateral submental and submandibular lymph nodes was palpable, firm, mobile, and mild tender. Examination of the intra oral cavity revealed a mild buccal and lingual cortical expansion between mandibular left canine to mandibular right first premolar with normal overlying mucosa. Furthermore, severe mobility of mandibular right and left central incisors was observed. It should be noted, that our patient had not proper oral hygiene. A few amount of brownish liquid aspiration in a fine needle biopsy.
The panoramic X-ray revealed a well-demarcated, multilacular cyst like radiolucent lesion between the left canine and right second premolar that involved the roots of the neighboring teeth (Figure 1). Furthermore, left and right mandibular lateral and central incisors had root canal therapy, and composite dental restorations.
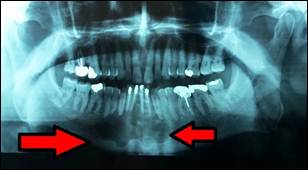
Figure 1. A panoramic X-ray shows a radiolucent lesion with ill-defined borders between the root of left canine and right second premolar.
Surgical removal of the lesion with left and right mandibular central incisors was done under general anesthesia (Figure 2). During the operation, a very aggressive lesion with perforated buccal cortical bone was noted.
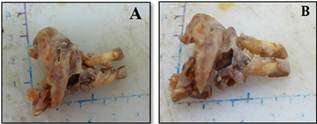
Figure 2. Macroscopic view of the surgical specimen.
Histopathological analysis of the hematoxylin/eosin-stained material showed a cystic lesion with nonkeratinized stratified squamous epithelium lining with different thickness. But, some area of histopathological view, degenerative changes with ballooning cells and in some area dysplastic features were observed. The islands of anaplastic squamous cells with marked nuclear pleomorphism, hyperchromatism and formation of keratin pearls and individual cell keratinizations were seen in connective tissue wall of cystic lesion (Figure 3).
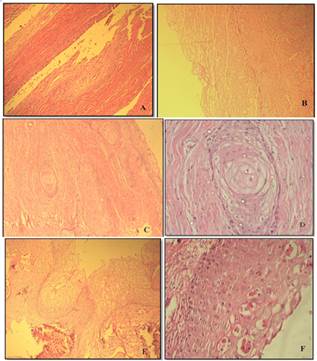
Figure 3. Microscopic appearance of specimen. A, Low-power view of cystic lesion that superiorly the cyst wall was lined by typical, nonkeratinized starified squamous epithelium and inferiorly including thickened cyst wall and connective tissue wall (original magnification х100). B, Degenerative changes with ballooning epithelial cells (original magnification х100). C, In place the epithelium was dysplastic and islands of squamous cell carcinoma infiltrated the fibrous cyst wall (original magnification х100). D, The formation of epithelial pearl in islands of dysplastic epithelium in fibrous cyst wall (original magnification х400). E, Different thickness of epithelial cyst wall with ballooning changes (original magnification х100). F, The formation of individual cell keratinization in epithelial cyst wall (original magnification х400).
The connective tissue wall of this cyst showed moderate chronic inflammatory infiltrate that chiefly consisted of lymphocytes and plasma cells. Furthermore, in many areas of connective tissue wall observed the basophilic bodies similar foreign bodies that caused foreign body inflammatory reactions. These basophilic bodies may be due to release sealer of root apex in during the root canal therapy. Also, giant cells and clestrol clefts was observed around these (Figure 4).
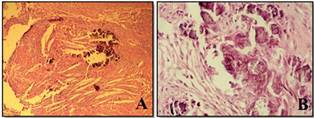
Figure 4. A, Foreign body inflammatory reactions and clestrol clefts around these (original magnification х100). B, The basophilic bodies similar foreign bodies (original magnification х400).
Finally, Primary intraosseous squamous cell carcinoma (PIOSCC) arising from a radicular cyst was diagnosed. The squamous cell carcinoma of this lesion was well differentiated.
The patient underwent partial resection of the mandible with 1-cm healthy bone margins. The patient was free from disease at 1 year of follow-up. To date, recurrence or metastasis has not occurred and follow-up visits are continuing.
3. Discussion
Only bone that contains epithelial tissue is jaw bones (10). Primary squamous cell carcinoma of the jaw bone is extremely rare with a prevalence of 0.3% to 3%. Most of this neoplasm is arising the epithelial lining of odontogenic cysts (4). Detection of the total number of reported cases of PIOSCC ex radicular cyst is very difficult, due to lack of follow-up and missing there. According to most studies, PIOSCC among the cancers that occur in the mouth is very rare. Gardner’s criteria for diagnosis of primary squamous cell carcinoma in odontogenic cysts including the following: 1) observation to histopathological changes from benign epithelial lining of odontogenic cyst to invasive SCC, 2) absence of dysplastic and carcinomatous changes in overlying oral mucosa, and 3) absence of carcinoma in the adjacent structures (11,12).
The mean age of patients in PIOSCC ex odontogenic cysts is 60.2 years, and occurs in a wide age group, ranging from 1.3 to 90 years. A male predication is noted, that the male/female ratio is 2.2:1. The male predominance for the PIOSCC patients is not specific. But, this result may be reflects the higher prevalence of odontogenic cysts in men (13,14). In this report, a 60-year-old man with PIOSCC arising of radicular cyst was presented, which is similar to the other studies (3,4,14).
According to some studies, PIOSCC arising odontogenic cyst the mandible are affected more commonly than maxilla (14,15). Furthermore, the posterior region of the mandible and the anterior region of the maxilla are more commonly involved (3,4). We observed the involvement of anterior mandibular region in our case.
In most studies have been showed that the clinical features in affected patients were jaw swelling that in early stage is painless, but in late stage become painful (16). Furthermore, perforation of buccal and /or lingual (or palatal) cortical plate of mandible or maxilla, adherence of the cyst lining to the bony cavity, and chronic sinus tract were common clinical features. According to studies, sensory disturbance in involved area is rare. This finding without a history of trauma is an alarming sign for diagnosis of malignant lesion such as PIOSCC(3,14). In our case, perforation of buccal cortex and mobility of involved teeth were observed, but there was no pain, sensory disturbance and chronic discharging sinus tract in the affected area.
Radiographically, a PIOSCC ex odontogenic cyst usually has a unilocular noncorticated radiolucency with well-defined or ill-defined margins. The odontogenic cyst with jagged or irregular margins that has indistinct borders should be considered as malignant changes. Especially, if in occlusal radiographs show the invasion and destruction by the lesion and erosion of the buccal and lingual plates (3,4,17). In this report, similar radiographic findings were observed by us.
Histopathologically, PIOSCC ex odontogenic cysts represent well or moderately differentiated squamous cell carcinoma arising the cystic epithelial lining (8,14). In this study, histopathological assessment showed a well differentiated keratinizing carcinoma which developed from a radicular cyst.
According to more studies, the residual or radicular cyst is the most common cyst associated with PIOSCC (3,4,8). Causes of the malignant transformation of odontogenic cyst lining are not known. But, the main stimulus factor to malignant changes is long term chronic inflammation with or without consideration of genetic cofactors (3,14).
The relation between chronic inflammation and carcinogenesis has been proved, but the exact mechanisms have not been determined (18). However, three main mechanisms in this regard are stated: (1)the formation of reactive oxygen and nitrogen species by phagocytes in chronic inflammation has been observed. Superoxide anion, hydrogen peroxide, and hydroxyl radical are the major species produced by the inflammatory cells, which these products can combine with nitric oxide to from other reactive nitrogen intermediates. These productions have the ability to damage DNA, proteins, cell membrane, and modulate enzyme activities and gene expression, therefore, Can act as carcinogenesis. Furthermore, the chronic inflammations promote apoptosis of normal cells, leading to compensatory proliferation of the remaining cells and increases the tissue stem cells and therefore are subject to DNA mutations and malignant transformation. Furthermore, in chronic inflammation responses produced cytokines that increase the growth of transformed cells. On the other hand, according to some studies expression of enzyme cyclooxygenase-2 causes the production of prostaglandins, leading to malignant changes (19). (2) Directly malignant transformation by inserting active oncogens in to the host genome, inhibiting tumor suppressor genes and stimulating mitosis have been observed in chronic inflammation. (3) Chronic inflammation may induce immunosuppression with reduced immunosurveillance (20).
According to studies, metastasis of PIOSCC is rare, but is an important prognostic factor and survival rate (5,14). The prognosis of PIOSCC ex odontogenic cyst is better than PIOSCC de novo (3). In our study, the patient has not lymph nodes metastasis.
The best treatment method for PIOSCC determined by the extent of the carcinoma. In the patients with carcinoma in situ or located intramural within the cyst, surgical removal is the choice treatment and the patient should be follow up. Additional therapy such as bone resection, radiotherapy, and chemotherapy even neck dissection should be planned in the patients with positive margins of the tumor and involved surrounding bone (14,21).
This tumor is very rare. The diagnosis of this neoplasm have been done by pathologic evaluation after cyst enucleation. This topic have been clear the importance of careful examination of the clinical, radiographical and histopathological features. We present the unusual case of PIOSCC ex radicular cyst without pain or sensation disorder and pus discharge. Furthermore, foreign body reaction associated with dysplastic epithelial of cystic wall would confuse the pathologist for best diagnosis.
4. Conclusions
The PIOSCC arising the odontogenic cyst is associated mainly with the residual/radicular cyst. Long standing chronic inflammation might be an important factor in the etiopathogenesis of PIOSCC ex odontogenic cyst, Therefore, because reduce the rate of malignant transformation of benign odontogenic cysts, the accurate and timely management and treatment of these lesions should be suggested.
References
- Eversole LR, Siar CH, vander Waal I: Primary intraosseous squamous cell carcinoma, in Barnes L, EvesonJW, Reichart P, Sidransky D (eds): World Health Organization Classification of Tumours: Pathology and Genetics of Head and Neck Tumours. Lyon, France, IARC, 2005, pp 290-291.
- Swinson BD, Jerjes W, Thomas GJ.Squamous cell carcinoma arising in a residual odontogenic cyst: case report.J Oral Maxillofac Surg 2005; 63: 1231-3.
- Jain M,Mittal S,Gupta DK. Primary intraosseous squamous cell carcinoma arising in odontogenic cysts: an insight in pathogenesis.J Oral Maxillofac Surg2013; 71: e7-14.
- Bereket C, Bekçioğlu B, Koyuncu M, Şener İ, Kandemir B, Türer A.Intraosseous carcinoma arising from an odontogenic cyst: a case report.Oral Surg Oral Med Oral Pathol Oral Radiol 2013; 116: e445-9.
- Adachi M,Inagaki T,Ehara Y,Azuma M,Kurenuma A,Motohashi M,etal. Primary intraosseous carcinoma arising from an odontogenic cyst: A case report.Oncol Lett2014; 8: 1265-1268.
- Kramer IRH, Pindborg JJ, Shear M. The WHO histological typing of odontogenic tumors. A commentary on the second edition. Cancer 1992; 70: 2988-94.
- Waldron CA, Mustoe TA. Primary intraosseous carcinoma of the mandible with probable origin in an odontogenic cyst. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1989;67:716-24.
- Chaisuparat R, Coletti D, Kolokythas A, Ord RA, Nikitakis NG. Primary intraosseous odontogenic carcinoma arising in an odontogenic cyst or de novo: a clinicopathologic study of six new cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2006; 101: 194-200.
- Sukegawa S,Matsuzaki H,Katase N,Kanno T,Mandai T,Takahashi Y, et al. Primary intraosseous squamous cell carcinoma of the maxilla possibly arising from an infected residual cyst: A case report.Oncol Lett2015;9:131-135.
- Murillo-Cortes J, Etayo-Perez A, Sebastian-Lopez C, Martino-Gorbea R, Rodriguez-Cortel JM. Primary intraosseous carcinoma arising in a mandibular cyst. Med Oral 2002;7:370-4.
- Gardner AF: A survey of odontogenic cysts and their relationship to squamous cell carcinoma.Dent J1975;41:161-7.
- Yasuoka T, Yonemoto K, Kato Y, Tatematsu N.Squamous cell carcinoma arising in a dentigerous cyst.J Oral Maxillofac Surg 2000;58:900-5.
- Bodner L,Manor E,Shear M,van der Waal I. Primary intraosseous squamous cell carcinoma arising in an odontogenic cyst: a clinicopathologic analysis of 116 reported cases.J Oral Pathol Med2011; 40:733-8.
- Bodner L, Manor E. Cystic lesions of the jaws – a review and analysis of 269 cases. Eur J Plastic Surg 2010; 33: 277–82.
- Zwetyenga N, Pinsolle J, Rivel J, Majoufre-Lefebvre C, Faucher A, Pinsolle V. Primary intraosseous carcinoma of the jaws. Arch Otolaryngol Head Neck Surg 2001;127:794-7.
- Müller S, Waldron CA: Primary intraosseous squamous carcinoma. Report of two cases.Int J Oral Maxillofac Surg1991; 20:362-5.
- KaffeI,Ardekian L,Peled M,Machtey E,Laufer D. Radiological features of primary intra-osseous carcinoma of the jaws: Analysis of the literature and report of a new case.Dento maxillofac Radiol1998;27:209-14.
- Karin M, Greten FR. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol 2005; 5: 749–59.
- DuBois RN. Cyclooxygenase-2 and colorectal cancer.Prog Exp Tumor Res2003; 37:124-37.
- Hold GL, El-Omar ME. Genetic aspects of inflammation and cancer. Biochem J 2008; 410: 225–35.
- Thomas G, Pandey M, Mathew A, et al. Primary intraosseous carcinoma of the jaws; pooled analysis of world literature and report of two new cases. Int J Oral Maxillofac Surg 2001; 30: 349–55.



