Neuroprotective Effect of Ellagic Acid Against Chronically Scopolamine Induced Alzheimer's Type Memory and Cognitive Dysfunctions: Possible Behavioural and Biochemical Evidences
Ramandeep Kaur, Shaba Parveen, Sidharth Mehan*, Deepa Khanna, Sanjeev Kalra
Department of Pharmacology, Rajendra Institute of Tech & Sciences, Sirsa, Haryana, India
Abstract
Adjudge the neuroprotective ability of Ellagic acid (EA) as a constructive herbal drug to impede cholinergic dysfunctions and oxidative stress in Alzheimer’s disease (AD) in chronically administered scopolamine induced Alzheimer’s type dementia in rats. Alzheimer’s type dementia was induced by chronically administered intraperitoneal injection of scopolamine (0.7 mg/kg) to rats for period of 7 days. EA (25 mg/kg and 50 mg/kg) and Donepezil (0.5 mg/kg) were administrated to rats orally daily for a period of 13 days. Memory-related behavioral parameters were evaluated using the elevated plus maze (EPM) for 2 days and morris water maze (MWM) for 5 days. At the end of protocol schedulei.e day 14, biochemical parameters were estimated like AChE, MDA, GSH, catalase and SOD to evaluate the neuroprotective action of EA via AChE inhibition and antioxidant activity. Chronically injected scopolamine treatment increased the transfer latency in EPM, escape latency time and shortened time spent in the target quadrant in MWM; these effects were reversed by EA. Scopolamine-mediated changes in malondialdehyde (MDA) and AChE activity were significantly attenuated by EA in rats. Recovery of antioxidant capacities, including reduced glutathione (GSH) content, and the activities of SOD and catalase was also evident in EA treated rats. The present findings sufficiently encourage that EA has a major role in the neuroprotection in chronically injected Scopolamine induced Alzheimer type dementia. The EA can be used as an effectual herbal treatment to prevent cholinergic dysfunctions and oxidative stress associated with Alzheimer type dementia.
Keywords
Neuroinflammation, Oxidative Stress, Acetylcholinesterase, Polyphenols, Ellagic Acid
Received: March 24, 2015
Accepted: April 23, 2015
Published online: May 28, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Alzheimer’s disease (AD) is a severe neurodegenerative disorder that gradually results in loss of memory and impairment of cognitive functions in the elderly.1-5 In 2014, an estimated 5.2 million people of all ages have AD in U.S. This includes an estimated 5 million people age 65 and older and approximately 200,000 individuals under age 65 who have younger-onset Alzheimer’s.6 The pathological features of AD include extracellular amyloid deposition and intra-neuronal neurofibrillary tangles (NFTs) of hyperphosphorylated microtubule-associated tau protein.7-9 The deposition of amyloid plaques is the primary event that leads to an inflammatory reaction, NFTs formation, and ultimately cause neuronal death.10-12 The mechanisms of neuronal cell loss in AD have not yet been fully elucidated, but increased oxidative stress and inflammation are considered important mediators of neuronal damage in AD.13-17
Many naturally occurring compounds have been proposed as potential therapies to slow or prevent the progression of AD, mostly by acting as antioxidants,18-24 but also with some direct anti-amyloid actions.18,23,25-30 Recent studies have suggested the positive effects of dietary antioxidants as an aid in potentially reducing somatic cell and neuronal damage by free radicals.18-21,31-34 The beneficial health effects of plant-derived products have been largely attributed to polyphenolic compounds, as well as vitamins, minerals and dietary fibers.18,19,35
Ellagic acid (EA), a non flavonoid polyphenol, plays an essential role in explaining the sensory properties of fruits, food and beverages which exhibit this phyto-constituent. 36-40 EA has been well proven to contain anti-oxidant, 41-46 anti-inflammatory, 47-51 anti-proliferative, 52-56 antidiabetic57-59 and cardioprotective60,61properties.
Neuroprotection can be a property of EA as it prevents both neuro-oxidation and neuroinflammation. 62-68 Moreover, by in-vitro studies it was observed that EA inhibits β-secretase (BACE1), thus inhibiting Aβ-fibrillation and decrease AChE activity. 4,69-71Recent studies suggested that glucose metabolism is affected during AD. 72-75The EA stimulated GLUT4 translocation primary factor responsible for insulin induced glucose uptake and maintain glucose homeostasis. 76,77The EA also shows modulation of monoaminergic system (serotonergic and noradrenergic systems) and GABAnergic system.78-80Cognitive impairment in AD patients correlates with disturbance in various neurotransmitters, as the ratio of excitatory-inhibitory neurotransmitter levels disturb, cytotoxic damage to neurons and glia occurs and norepinephrine and serotonin levels declined.81-91 Further, Gamma-amino butyric acid (GABA) increases the formation of soluble receptor for advanced glycation end products (RAGE) and decreases the levels of full-length RAGE, lowering the Aβ uptake and inflammatory mediated reactions.92,93
Scopolamine, an antimuscarinic agent, competitively antagonizes the effect of acetylcholine on the muscarinic receptors by occupying postsynaptic receptor sites with high affinity and increases AChE activity in the cortex and hippocampus. 94-103 Scopolamine abolishes cerebral blood flow due to cholinergic hypofunction. 104-107Scopolamine additionally triggers ROS, inducing free radical injury and an increase in a scopolamine-treated group brain MDA levels and deterioration in antioxidant status. 108-112 Scopolamine induces neuro-inflammation by promoting high level of oxidative stress and pro inflammatory cytokines in the hippocampus. 113-119Scopolamine is proven to increase levels of APP and Tau. Chronic administration of scopolamine led to marked histopathological alterations in the cerebral cortex, including neuronal degeneration.30,120-122 Scopolamine administration has been used both in healthy human volunteers and in animals as a model of dementia to determine the effectiveness of potential new therapeutic agents for Alzheimer's disease123-128 (Fig.1).
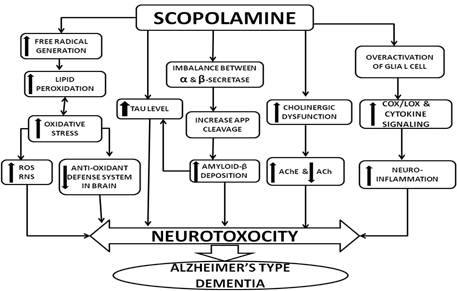
Fig. 1. Scopolamine induced experimental model of Alzheimer’s type dementia
Donepezil, a reversible inhibitor of AChE, is neuroprotective due to not only activation of cholinergic transmission but also by reducing the amount of the toxic form of amyloid β fibrils. 129-136 Donepezil ameliorated the scopolamine induced memory impairment by reducing AChE activity and oxidative stress and restoring cerebral circulation. 137-143With this background, EA might show neuroprotection via inhibiting neuronal dysfunctions. There is major requirement to determine therapeutic potential for EA in cases of AD with suitable behavioral and biochemical markers. This research was an attempt to investigate the neuroprotective effect of EA, potential of doses for the treatment of Alzheimer’s disease.
2. Material and Method
2.1. Chemicals
EA was purchased from Yucca Interprises, Mumbai, India and suspended in saline solution. Scopolamine hydrochloride was purchased from Sigma–Aldrich, St, Louis, MO, USA. Donepezil was obtained from Ranbaxy Pvt. Limited, Mumbai, India and both scopolamine and doenpezil were dissolved in saline solution. All reagents used in this study were of analytical grade and high purity.
2.2. Animals
Male Wistar rats (weighing 220-250 g, aged 8-10 months) obtained from the Animal House of the Institute were employed in the studies. The animals were kept in polyacrylic cages with wire mesh top and soft bedding. They were kept under standard husbandry conditions of 12h reverse light cycle with food and water ad libitum, maintained at 22±2oC. The experimental protocol was approved by Institutional Animal Ethics Committee (IAEC) as per the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Government of India (RITS/IAEC/2013/01/01). Animals were acclimatized to laboratory conditions prior to experimentation.
2.3. Drug Administration
EA was administered by oral (p.o.) route in dose of 25 mg/kg and 50 mg/kg. Scopolamine was administered by intraperitoneal (i.p.) route in dose of 0.7 mg/kg. Donepezil was administered by oral (p.o.) route in dose of 0.5 mg/kg.
Six groups (each group consist six rats) were employed in the present study. (i) Group1-Normal Control (ii) Group2-Scopolamine Control (0.7mg/kg,i.p.) (iii) Group3-EA Perse (50mg/kg, p.o.)25mg/kg, p.o.+ Scopolamine (0.7mg/kg, i.p.) (vi) Group6-EA 50mg/kg, p.o.+ Scopolamine (0.7mg/kg, i.p.). After a 5-day habituation period, rats were given EA (25 and 50 mg/kg, p.o.) and Donepezil (0.5 mg/kg, p.o.) for total of 13 days. EA alone was treated for 6 days and then scopolamine (0.7 mg/kg, i.p.) was administered together with EA for another 7 days. Rats underwent locomotor activity (LMA) for 2 days i.e. 6th day and 13thday, MWM test for 5 days i.e. 7th day to 11th day. The day after completion of morris water maze (MWM), the elevated plus maze (EPM) was conducted for 2 days i.e. 12th to 13th day. The day after EPM, the rats were sacrificed and biochemical parameters were estimated (Fig. 2).
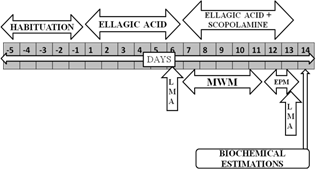
Fig. 2. Protocol schedule to determine the neuroprotective effect of Ellagic Acid in scopolamine induced Alzheimer’ type memory and cognitive dysfunctions
2.4. Elevated Plus Maze
Elevated plus maze (EPM) served as the behavioral model (where in the stimulus existed outside the body) to evaluate learning and memory in rats. It consists of two opened arms (50 cm*10cm) and two covered arms (50cm*40cm*10cm). The arms were extended from central platform (10cm * 10 cm), and the maze was kept elevated to a height of 50cm from the floor. The EPM was conducted for 2 days i.e. 12th to 13th day of protocol schedule. Each animal was kept at the end of an open arm, facing away from the central platform on 12th day. Transfer Latency (TL), which was taken as the time taken by the animal to move into any one of the covered arms with all its four legs, recorded on 12th day i.e. acquisition trial144. If the rat did not enter into one of the covered arms within 120s then it was gently pushed into one of the two covered arms and thetransfer latencywas assigned as 120s. The rats were allowed to explore the maze for 10s and then were returned to its home cage. TL was again examined 24hr after the first trial on 13th day of protocol schedule i.e. retention latency.
2.5. Spatial Navigation Task in Morris Water Maze
Morris water maze employed in the present study was a model to evaluate spatial learning and memory. Escape from water itself acts as motivation and eliminates the use of other motivational stimuli such as food and water deprivation. Water provides uniform environment and eliminates interference due to olfactory clues. 145 Animals were trained to swim to a platform in a circular pool (180 cm diameter*60 cm) located in a sound attenuated dark test room. The pool was filled with water (28±2°C) to a depth of 40 cm. A movable circular platform, 9cm in diameter and mounted on a column, was placed in the pool 2 cm below the water level for escape latency time (ELT), while during time spent in the target quadrant (TSTQ) the platform was removed. Four equally spaced locations around the edge of the pool (N, S, E, and W) were used to divide the pool into 4 quadrants and one of them is used as start point, which was same during all trials. The pool was filled with opaque water to prevent visibility of the platform in the pool. The escape platform was placed in the middle of one of the random quadrants of the pool and kept in the same position throughout the experiments. Animals received a training session consisting of day 7 to 10 and ELT was recorded. ELT defined as the time taken by the animal to locate the hidden platform. ELT was noted as an index of learning. Each animal was subjected to single trial for four consecutive days (starting form 7th day of EA administration to 10th day), during which they were allowed to escape on the hidden platform and to remain there for 20 s. If the rats failed to find the platform within 120 s, it was guided gently onto the platform and allowed to remain there for 20 s.
On fifth day (i.e., 11th day of EA administration) the platform was removed. Rats were placed in water maze and allowed to explore the maze for 120 s. Time spent in three quadrants, that is, Q1, Q2 and Q3 was recorded and TSTQ in search of the missing platform provided as an index of retrieval. Care was taken not to disturb the relative location of water maze with respect to other objects in the laboratory.
2.6. Assessment of Locomotor Activity
Gross behavioral activity was assessed by digital actophotometer on 6th day and 13th day of protocol schedule to rule out any interference in locomotor activity by drugs which may affect the process of learning and memory, in before and after of MWM task. Each animal was observed over a period of 5 min in a square (30 cm) closed arena equipped with infrared light-sensitive photocells and values expressed as counts per 5 min. 146 The beams in the actophotometer, cut by the animal, were taken as measure of movements. The apparatus was placed in a darkened, sound-attenuated and ventilated testing room.
2.7. Preparation of Brain Homogenate
On 14th day of protocol schedule, Animals were sacrificed by decapitation, brains removed and rinsed with ice cold isotonic saline solution. Brain tissue samples were then homogenized with 10 times (w/v) ice cold 0.1M phosphate buffer (pH 7.4). The homogenate was centrifuged at 10,000 x g for 15min, supernatant was separated and aliquots were used for biochemical estimations. 146
2.8. Protein Estimation
The protein content was measured by using Agappe protein estimation kit (Biuret method).
2.8.1. Estimation of Acetylcholinesterase Levels
The quantitative measurement of AChE activity in brain was performed according to the method described by Ellman et al. (1961). 147 The enzymatic activity in the supernatant was expressed as nmol per mg protein.
2.8.2. Estimation of Malondialdehyde
The quantitative measurement of MDA – end product of lipid peroxidation - in brain homogenate was performed according to the method of Wills (1966). 148The concentration of MDA was expressed as nmol per mg protein.
2.8.3. Estimation of Reduced Glutathione
GSH in brain was estimated according to the method described by Ellman et al. (1959). 149 The concentration of glutathione in the supernatant expressed as µmol per mg protein.
2.8.4. Estimation of Superoxide Dismutase Activity
SOD activity was measured according to the method described by Misra and Frodvich (1972). 150The activity of SOD was expressed as % activity.
2.8.5. Estimation of Catalase Activity
Catalase activity was measured by the method of Aebi (1974). 151 The activity of catalase was expressed as % activity.
2.9. Statistical Analysis
All the results and data were expressed as mean±standard deviation. Data was analyzed using two way ANOVA followed by Post hoc test bonferroni and one way ANOVA followed by Post hoc test tukey’s multi-comparison test. P<0.05 was considered as statistically significant.
3. Results
3.1. Effect of Ellagic Acid on Rats in Elevated Plus Maze
On 12th day of protocol schedule, acquisition latency was recorded. Retention was observed as transfer latency (TL) on 13th day to evaluate learning and memory in rats using EPM. On 12th and 13th day Scopolamine administered rats showed remarkable increase (113±9.380 and 106.5±11.148 sec) in TL, when compared to normal (64±4.242 and 36.833±6.765 sec) and EAperse rats (63.333±10.385 and 32.833±3.311 sec). During experiment, EAperse administration did not reveal any change, when compared to normal rats in TL. Donepezil, a well established standard drug for AD considerably decrease (65.5±13.003 and 21.666±5.085 sec) TL, when compared to Scopolamine managed rats and reversed the memory impairment induced by Scopolamine. Administration of EA at the dose of 25 mg/kg, p.o. exhibit notable decrease (72.00±8.049 and 39.333±6.186 sec) in TL, when compared to Scopolamine treated rats. EA (50 mg/kg, p.o.) administration also decrease (69.333±8.041 and 25.333±3.881 sec) TL, when differentiate to Scopolamine handled rats and there were expressively variation was found in between treatment doses of EA 25 & 50 mg/kg, p.o. indicating improved retention memory (Fig. 3).
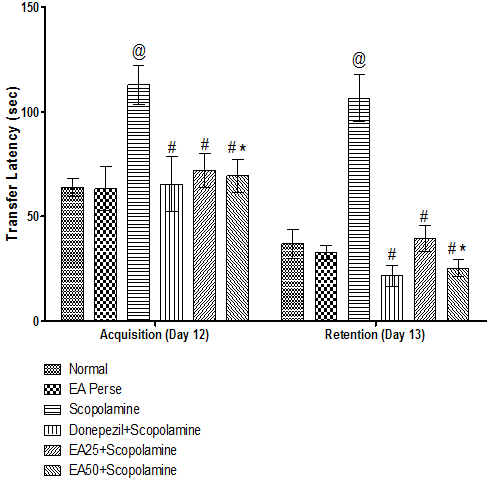
Fig. 3. Effect of Ellagic acid on Transfer latency of rats using Elevated Plus Maze
3.2. Effect of Ellagic Acid on Rats in Spatial Navigation Task Using Morris Water Maze
On 7th to 10th day of 14 day protocol schedule, escape latency time (ELT) was observed. On 7th day, there were no significant changes observed in Scopolamine (94.33±13.125 sec) treated rats, when compared to normal (89±9.859 sec) and EAperse governed (86.33±13.937 sec)rats.EAperse administration did not show any significant change when compared to normal rats. Moreover, Donepezil treated rats did not show any considerable changes (88±9.033 sec), when compared to Scopolamine responded rats. In the treatment groups, administration of EA did not confirm notable changes (96.33±10.053; 88.66±10.689 sec) in ELT at 25 and 50 mg/kg, p.o. when compared to Scopolamine treated rats. There were no changes found in ELT between treatment doses of EA 25 & 50 mg/kg, p.o.
Comparison data of 8th day, 9th day and 10th day ELT in MWM, showed that Scopolamine administered rats manifest remarkable increase (92±8.173, 85.33±12.75 and 83.33±8.664 sec) in ELT, when collate to normal (76.33±7.840, 29.16±7.808 and 15.33±3.723 sec) and EAperse (67.33±5.645, 29.33±8.710 and 15±2.898 sec) rats. EAperse administration did not show any significant difference, when compared to normal rats during ELT. Donepezil served rats outstandingly decreased (51±10.158, 26.16±6.40 and 10.83±4.622 sec) ELT when compared to Scopolamine dosed rats. EA at 25 mg/kg, p.o. proved remarkable decreased (79±10.807, 60.83±8.658 and 38.16±9.703 sec) in the ELT, when compared to Scopolamine employed rats. EA at the dose 50 mg/kg, p.o. significantly decreased (65.33±11.707, 43±9.838 and 24.5±8.312 sec) the ELT, when compared to Scopolamine and EA 25 mg/kg, p.o. treated rats, indicating remarkable improvement in learning (Fig. 4).
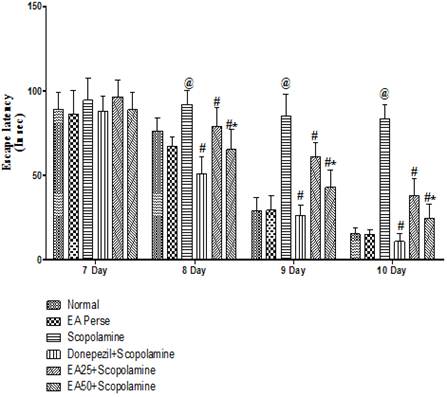
Fig. 4. Effect of Ellagic acid on Escape latency time of rats on 7th day to 10th day using Morris Water Maze
Values were mean ± SD, @ p<0.05 as compared to Normal & EA perse, # p<0.05 as compared to Scopolamine, * p<0.05 as compared to EA 25 + Scopolamine.
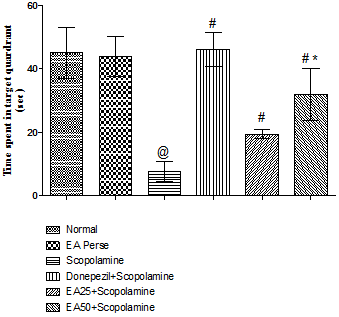
Fig. 5. Effect of Ellagic acid on time spent in target quadrant of rats using Morris Water Maze
Values were mean±SD, @ p<0.05 as compared to Normal & EA perse, # p<0.05 as compared to Scopolamine, * p<0.05 as compared to EA 25 + Scopolamine
On 11th day of protocol schedule TSTQ was performed. Time spent in target quadrant (TSTQ) in search of missing platform provided as an index of retrieval. Scopolamine treated rats showed remarkable decrease (7.667±3.077 sec) in TSTQ when compared to normal (45.17±8.060 sec) and EAperse treated (43.83±6.242 sec) rats. In perse group of EA, there were no changes during TSTQ when compared to normal group. Further, Donepezil served rats improved (46.17±5.345 sec) memory when compared to Scopolamine treated rats. EA (25 mg/kg, p.o.) administration showed remarkable increase (19.50±1.517 sec) in TSTQ when compared to Scopolamine treated rats. EA (50 mg/kg, p.o.) administration indicated improvement (32.00±8.149 sec) in memory function when compared with Scopolamine governed rats. Moreover, markedly difference was also observed in between treatment doses of EA(Fig. 5).
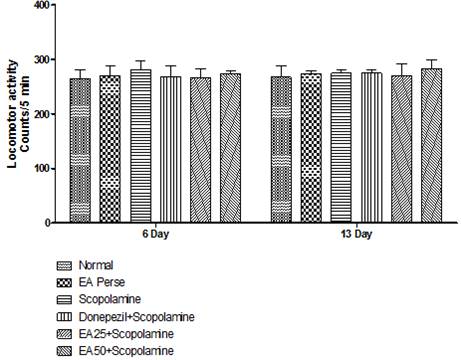
3.3. Effect of Ellagic Acid on Rats in Locomotor Activity
On 6th day and 13th day of protocol schedule, locomotor activity was observed to rule out any interference in locomotor activity by treatment drugs. Scopolamine employed rats did not reveal any significant changes (281.333±15.318 and 274.833±5.344) in locomotor activity when compared to normal (263.833±17.474 and 274.5±21.314) and EAperse (270.666±18.250 and 274.5±4.764) rats. EAperse administration also did not show any considerable change in locomotor activity at 50 mg/kg, p.o. when compared to normal rats. Donepezil treated also showed trivially changes (267.5±21.314 and 274.833±5.344) when compared to Scopolamine treated rats. EA 25 mg/kg, p.o. (266.833±15.458 and 270.833±20.692) and 50 mg/kg, p.o. (274.5±4.764 and 283.5±16.208) administration did not showed any notable changes in locomotor activity of rats when differentiate to Scopolamine treated rats, indicating there were no effect on locomotor activity (Fig. 6).
3.4. Effect of Ellagic Acid on Acetylcholinesterase Levels
Prolongation of availability of acetylcholine has been used to enhancing cholinergic function. This prolongation may be achieved by inhibiting AChE. Scopolamine administered rats significantly increased (415.0±19.62) the AChE level when compared to normal (136.8±4.956) and EAperse (137.2±4.167) rats. EAperse administration did not show any appreciable changes in AChE level at the dose of 50 mg/kg, p.o. when compared to normal rats. Donepezil treated rats appreciably decreased (231.0±7.668) the AChE level in contrast to Scopolamine dosed rats. EA (25 mg/kg, p.o.) showed remarkably diminished the AChE level (360.8±15.96) when compared to Scopolamine rats. Administration of EA (50 mg/kg, p.o.) significantly reduced (311.7±17.63) the AChE level when compared to Scopolamine employed rats. Moreover, there were expressive distinction was present in between treatment doses of EA(Fig.7).
Values were mean±SD
Fig. 6. Effect of Ellagic acid on locomotor activity of rats using actophotometer.
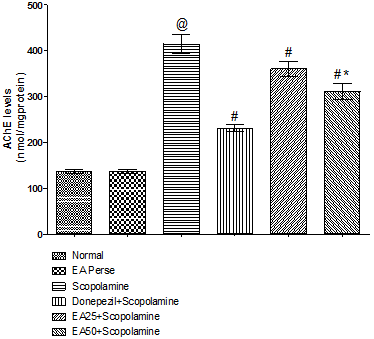
Fig. 7. Effect of Ellagic acid on Acetylcholinesterase levels.
Values were mean±SD, @ p<0.05 as compared to Normal & EA perse, # p<0.05 as compared to Scopolamine, * p<0.05 as compared to EA 25 + Scopolamine
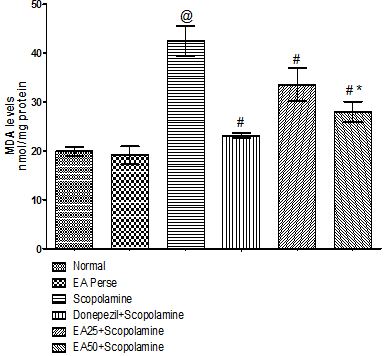
Fig. 8. Effect of Ellagic acid on Malondialdehyde levels
Values were mean±SD, @ p<0.05 as compared to normal & EA perse, # p<0.05 as compared to Scopolamine, * p<0.05 as compared to EA 25 + Scopolamine.
3.5. Effect of Ellagic Acid on Malondialdehyde Levels
MDA is an indicator of lipid peroxidation. Scopolamine administration increased (42.50±3.082) the MDA level when compared to normal (19.88±0.960) and EAperse (19.15±1.841) rats. Further, EAperse administration did not show any considerable changes in MDA levels when compared to normal rats. Donepezil appreciably decreased (23.12±0.511) the MDA level when compared to Scopolamine managed rats. EA (25 mg/kg, p.o.) administration showed remarkably decrease (33.57±3.347) in MDA level when compared to Scopolamine treated rats. EA administered rats at the dose of 50 mg/kg, p.o significantly decreased (27.97±2.089) in MDA level when compared to Scopolamine and EA 25 mg/kg, p.o. treated rats(Fig. 8).
3.6. Effect of Ellagic Acid on Reduced Glutathione Levels
Reduced GSH is a marker of cellular antioxidant and provide protection against oxidative stress. Scopolamine governed rats remarkably decreased (2.067±0.417) the GSH level when compared to normal (9.833±0.776) and EAperse treated (9.733±0.799) rats. EAperse administration did not show any considerable changes in GSH levels in contrast to normal rats. Donepezil outstandingly increase (7.767±0.361) the GSH levels when compared to Scopolamine treated rats. EA (25 mg/kg, p.o.) administration exhibited remarkable increase (5.250±0.575) in GSH level when compared to Scopolamine treated rats. EA (50 mg/kg, p.o.) showed significantly increase (6.317±0.386) in GSH level when compared to Scopolamine treated rats. Moreover, in between treatment doses of EA, there were significance difference was present (Fig. 9).
3.7. Effect of Ellagic Acid on Superoxide Dismutase Activity
SOD is an antioxidant enzyme, which plays a key role in detoxifying superoxide anions. Scopolamine administered rats significantly decreased (27.33±3.386) the SOD levels in brain homogenate when compared to normal (100.0±0.0) and EAperse (95.83±2.639) rats. EAperse administration did not reveal any considerable change in SOD activity when collated to normal rats. Donepezil expressively increase (82.00±3.950) SOD activity when compared to Scopolamine treated rats. In treatment group, EA (25 mg/kg, p.o.) administration showed remarkable increase (59.17±8.060) in SOD activity when compared to Scopolamine treated rats. EA (50 mg/kg, p.o.) administration showed significantly increase (71.33±4.033) in SOD activity when compared to Scopolamine treated rats and there were remarkably disparity was found in between EA treated groups (Fig. 10).
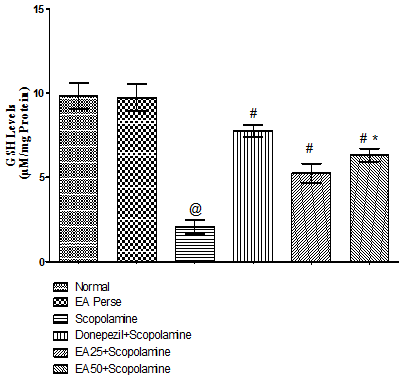
Fig. 9. Effect of Ellagic acid on reduced Glutathione levels
Values were mean±SD, @ p<0.05 as compared to Normal & EA perse, # p<0.05 as compared to Scopolamine, * p<0.05 as compared to EA 25 + Scopolamine.
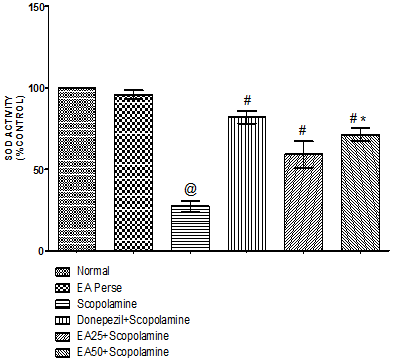
Fig. 10. Effect of Ellagic acid on Superoxide dismutase activity
Values were mean±SD, @ p<0.05 as compared to Normal & EA perse, # p<0.05 as compared to Scopolamine, * p<0.05 as compared to EA 25 + Scopolamine.
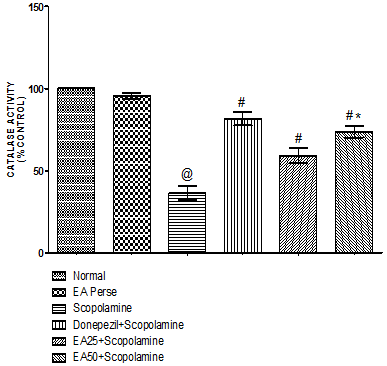
Fig. 11. Effect of Ellagic acid on catalase activity
Values were mean±SD, @ p<0.05 as compared to normal & EA perse, # p<0.05 as compared to Scopolamine, * p<0.05 as compared to EA 25 + Scopolamine.
3.8. Effect of Ellagic Acid on Catalase Activity
Catalase is also an antioxidant enzyme which has capability to detoxify oxidative free radicals. Scopolamine treated rats manifested remarkable decrease (36.50±4.461) in catalase activity in brain homogenate when differentiated to normal (100.0±0.0) and EAperse treated (95.50±1.871) rats. EAperse administration did not show any considerable changes in catalase activity when compared to normal rats. Donepezil significantly increase (81.67±4.033) in catalase activity when compared to Scopolamine treated (36.50±4.461) rats. EA (25 mg/kg, p.o.) remarkably increased (59.17±4.579) the catalase activity when compared to Scopolamine treated rats. EA (50 mg/kg, p.o.) administration exhibited significantly increase (73.67±3.559) in catalase activity when compared to Scopolamine and EA 25 mg/kg, p.o. treated rats (Fig. 11).
4. Discussion
Clinically AD is characterized by an insidious degradation of memory, associated with functional decline and neurobehavioral disturbances. 152,153Despite the availability of various treatment strategies, the severity and prevalence of this disease are not yet under control. Therefore, alternative and complementary medicines including herbal supplements, phytochemicals and extracts are being utilized in the management of AD. 154-160The current hypothesis about the mechanisms by which neurons come into necrotic or apoptotic processes has led to believe that the therapeutic use of natural antioxidants may be beneficial in aging and neurodegenerative disorders.161-163
In the present study, the effect of improving memory deficit of EA was evaluated using chronically administered scopolamine induced Alzheimer’s type dementia in rats.
It is well known that scopolamine as a cholinergic receptor antagonist has been shown to impair learning and memory processing.95,97,100,103 Scopolamine produces deficits in acquisition, immediate retention and working memory.164-169
The current study has revealed that long term administered scopolamine significantly increased the levels of lipid peroxidation products such as MDA, and decreased the levels of antioxidants viz., GSH, SOD and catalase. The increase in oxidative stress was found to be associated with increase in AChE activity and spatial cognitive deficit. Present findings are in tune with previous reports. 99,102, 111,118
Scopolamine induced Alzheimer’s type dementia model has been widely used to provide a pharmacological model of memory dysfunction for screening potential cognition enhancing agents.99,110-112,170The cognitive-enhancing activity of EA on chronically administered scopolamine induced memory impairments in rats was investigated by using behavioral and biochemical parameters.
During elevated plus maze, decrease in retention latency indicated improvement of memory and vice versa. 142,171-173In EPM, it was shown that long term injected scopolamine also drastically increase in TL, demonstrating that the central cholinergic neuronal system plays an important role in learning acquisition. EA dose-dependently decreased TL prolongation induced by scopolamine. These results suggested that the neuroprotective effect of EA on scopolamine-induced memory impairment may be related to mediation of the cholinergic nervous system.
In order to confirm the effects of EA, MWM was used to test spatial learning in rats, where scopolamine treated rats were taking more time to reach at the hidden platform which shows memory impairments in this spatial task. EA treated rats impressively reduced the escape latency prolonged by scopolamine. Moreover, EA exhibited appreciable improvement of cognitive performance as indicated by significant decrease in ELT. It is important to notice that MWM test investigating spatial learning and memory has been used in detecting changes of the central cholinergic system.174-178 If the animals spent more time in target quadrant where the platform had previously been placed during the training session, this would indicate that the animals learned from prior experience with the MWM test, showing the spatial memory improvement. Scopolamine treated rats decreased TSTQ, on the other side EA treated rats expressively increased the TSTQ. Both the test doses viz., 25 mg/kg, p.o. and 50mg/kg, p.o. significantly attenuated these behavioral changes in rats with chronically administered scopolamine induced memory and cognitive impairment.
Along with EPM and MWM, Locomotor activity also was investigated using actophotometer to determine any modulation in locomotor activity by treatment drugs which may affect locomotion in EPM and MWM. However no significant difference in locomotor activity was observed in any of the animal groups. These results suggest that there was not any sedative effect or interference in EPM and MWM locomotion. Therefore, transfer latency in EPM, escape latency and TSTQ in MWM were purely result of improved memory. Thesefore, EA can repair the long-term memory in chronically injected scopolamine-induced memory impairments.
To investigate the effect of EA on cholinergic function, that governs vital aspects of memory and other cognitive functions, brain acetylcholinesterase activity was measured in the present study. The hippocampus, amygdala and cortical regions of the brain are mainly involved in cholinergic transmission to monitor learning and memory processing and seem to be more prone to oxidative damage. 9,179-181 Moreover, oxidative damage to the rat synapse in these regions of brain has been reported to contribute to cognitive deficits. 182,183The AD is characterized by alterations at the level of various neurotransmitters. The most severely affected is the cholinergic system, which is responsible for the storage and retrieval of items in memory and its degradation correlates well with the severity of cognitive and memory impairment.10,184
In this study, scopolamine was found to significantly elevate AChE activity, an enzyme responsible for degradation of ACh, which is in tune with earlier reports. 102,118 This increase in AChE activity was significantly restored dose dependently by EA. These observations suggest the modulation of cholinergic neurotransmission and/or prevention of cholinergic neuronal loss.
Recently, many studies have reported that memory impairments is associated to oxidative damage in the scopolamine-induced dementia in rats.110-112 Moreover, many clinical studies have reported that oxidative stress is closely involved in the pathogenesis of AD.13,185-188
Lipid peroxidation is an important indicator of neurodegenration of brain. Unlike other body membranes, neuronal membranes contain a very high percentage of long chain polyunsaturated fatty acids because they are used to construct complex structures needed for high rates of signal transfer. ROS are generated continuously in nervous tissues during normal metabolism and neuronal activity. The brain is subjected to free radical induced lipid peroxidation because it uses one-third of the inspired oxygen.189,190Lipids and proteins, the major structural and functional components of the cell membrane, are the target of oxidative modification by free radicals in neurodegenerative disorders.191 Extensive evidence exists on lipid peroxidation and protein oxidation leading to loss of membrane integrity, an important factor in acceleration of aging and age-related neurodegenerative disorders. Oxidative stress has been implicated in the pathogenesis of AD in humans. 192-194
In the present study, scopolamine-injection in rats significantly induced peroxidation of lipids and proteins, and reduced antioxidant defense indicating increased oxidative stress. MDA is an end product of lipid peroxidation and is a measure of free radical generation and scopolamine injected rats showed extensive lipid peroxidation as evidenced by increase in MDA levels. In order to evaluate the effect of EA on lipid peroxidation in brain, MDA level was assessed. MDA level was remarkably increased by scopolamine and EA dose-dependently reduced MDA level, indicating the reduced peroxidation of lipids.
Lipid peroxidation may enhance due to depletion of GSH content in the brain, which is often considered as the first line of defense of the cell by this endogenous antioxidant against oxidative stress.191,195-197 Evidence has been presented that the neuronal defense against H2O2, which is the most toxic molecule to the brain, is mediated primarily by the glutathione system.198-200 GSH is a tri-peptide, an endogenous antioxidant found in all animal cells in variable amounts and is a very accurate indicator of oxidative stress.197Consistent with previous studies, in present study, scopolamine treatment significantly decreased the GSH levels. Further, co-administration of EA markedly improved GSH levels.
The most important antioxidant enzymes are SOD and catalase. SOD plays a key role in detoxifying superoxide anions, which otherwise damages the cell membranes and macromolecules. Scopolamine administration showed a significant reduction in enzymatic activity of SOD and catalase. On the other side, Catalase has the capability to detoxify H2O2 radicals. Release of H2O2 promotes the formation of numerous other oxidant species that greatly contributes for oxidative stress leading to the pathogenesis of AD.189,201 Scopolamine treatment was found to be decreased SOD and catalase activities. Treatment of rats with EA significantly preserved the activities of SOD and catalase.
It has been well documented that persistent administration of scopolamine in response to degradation of ACh and increase the level of AChE enzyme, further responsible for the production of oxidative stress and pro-inflammatory mediators viz., cytokines and further activation of these cells.99,110-112 A strong and long lasting administration of scopolamine has been demonstrated to cause cholinergic dysfunction while inhibition of this scopolamine mediated abnormalities has shown to reverse cholinergic dysfunction as well as inhibit the release of oxidative and inflammatory markers. 99,103,112 The results of the present study suggest that chronic administration of EA perse did not have any significant effect on cognitive performance in normal animals. But, EA treatment groups at the dose of 25 & 50 mg/kg, p.o. showed marked improvement in cognitive tasks when compared to scopolamine treated rats suggesting the significant role of ACh in long lasting administrated scopolamine mediated cognitive dysfunction. Reports also support that ACh is involved in memory acquisition and retention.10,155,202,203Moreover, scopolamine injection drastically impaired memory retention, resembling Alzheimer's dementia.103,112The same has been reported to be attenuated by pretreatment with herbal supplements and extracts, and phytochemicals.156-158,160
The presented data in this study also suggests that EA possesses potent antioxidant activity by scavenging ROS and exerting a neuro-protective effect against oxidative damage induced by long term administration of scopolamine(Fig. 12). Predominant role of AChE inhibition, antioxidant activity reveal an important contributory factor to the beneficial effects of EA against dementia. Higher dose of Ellagic acid i.e. 50 mg/kg, p.o. was found more neuroprotective in all behavioral and biochemical evaluations. At lastly, the neuroprotective effects of EA might result from the regulation of AChE and the anti-oxidative defense system. These results suggest that EA can be used as a constructive herbal drug to impede cholinergic dysfunctions and oxidative stress in AD.
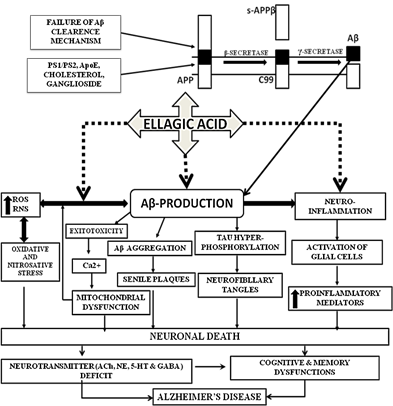
Fig. 12. Neuroprotective action of Ellagic Acid via modulating various signaling pathways involved in the progression of Alzheimer’s Disease.
5. Conclusion
It was concluded that long term injected scopolamine could persuade Alzheimer’s type dementia via increase AChE levels and oxidative stress like bio-markers. Scopolamine mediated Alzheimer’s type dementia is mainly associated with cognitive and memory impairments in behavioral models like elevated plus maze and morris water maze. Ellagic acid diminished the acetylcholinesterase level and improves the anti-oxidant defense system. Further, Ellagic acid downturned the cognitive impairments induced by scopolamine. Like Donepezil, Ellagic acid reversed the scopolamine induced Alzheimer’s type dementia in rats. Therefore, Ellagic Acid can be used as an effectual herbal treatment to prevent cholinergic dysfunctions and oxidative stress associated with Alzheimer’s type dementia.
On the basis of this study, the major bio-markers of Alzheimer’s disease like amyloid beta, inflammatory cytokines and histopathological changes can be further evaluated according to current protocol schedule to confirm and justify the strong evidence of Ellagic acid in long term injected scopolamine mediated dementia.
Acknowledgements
We express our gratitude to Chairman Dr. Rajender Sra and Secretary Dr. Om Parkash, Rajendra Institute of Technology and Sciences, Sirsa, Haryana, India, for their inspiration and constant support.
References
- Chong ZZ, Li F, Maiese K. Stress in the brain: novel cellular mechanisms of injury linked to Alzheimer’s disease. Brain Res Rev. 2005; 49:1-21.
- Walsh DM, Klyubin I, Shankar GM, Townsend M, Fadeeva JV, Betts V, et al. The role of cell-derived oligomers of Abeta in Alzheimer's disease and avenues for therapeutic intervention. Biochem Soc Trans. 2005; 33(Pt 5):1087-90.
- Jellinger KA. Alzheimer 100-highlights in the history of Alzheimer research. J Neural Transm. 2006; 113:1603-23.
- Muthaiyah B, Essa MM, Chauhan V, Chauhan, A. Protective Effects of Walnut Extract Against Amyloid Beta Peptide-Induced Cell Death and Oxidative Stress in PC12 Cells. Neurochem Res. 2011; 36: 2096-103.
- Anand R, Gill KD, Mahdi AA. Therapeutics of Alzheimer's disease: Past, present and future. Neuropharmacology. 2014; 76 Pt:A 27-50.
- Alzheimer’s Association. Alzheimer’s disease Facts and Figures, Alzheimer’s & Dementia. 2014; 10:16.
- Anderson DC. Alzheimer’s disease Biomarkers: More Than Molecular Diagnostics. Drug Develop Res. 2013; 74:92–111.
- Blennow K, Zetterberg H, Fagan AM. Fluid biomarkers in Alzheimer disease. Cold Spring Harb Perspect Med. 2012; 2: a006221.
- Mehan S, Meena H, Sharma D, Sankhla R. JNK: a stress-activated protein kinase therapeutic strategies and involvement in Alzheimer's and various neurodegenerative abnormalities. J Mol Neurosci. 2011; 43: 376-90.
- Kihara T, Shimohama S. Alzheimer's disease and acetylcholine receptors. Acta Neurobiol Exp. 2004; 64:99-105.
- Verdile G, Fuller S, Atwood CS, Laws SM, Gandy SE, Martin RN. The role of beta amyloid in alzheimers disease: still a cause of everything or the only one who got caught? Pharmaclo Res. 2004; 50:397-409.
- Anandatheerthavarada HK, Biswas G, Robin MA, Avadhani NG. Mitochondrial targeting and a novel transmembrane arrest of Alzheimer’s amyloid precursor protein impairs mitochondrial function in neuronal cells. J Cell Biol. 2003; 161: 41-53.
- Marcus DL, Thomas C, Rodriguez C, Simberkoff K, Tsai JS, Strafaci JA, et al. Increased peroxidation and reduced antioxidant enzyme activity in Alzheimer’s disease. Exp Neurol. 1998; 150:40-4.
- Nunomura A, Perry G, Aliev G, Hirai K, Takeda A, Balraj EK, et al. Oxidative damage is the earliest event in Alzheimer’s disease. J Neuropathol Exp Neurol. 2001; 60: 759-67.
- Sarkar PK. Degeneration and death of neurons in adult neurodegenerative diseases. Curr Sci. 2005; 89:746-73.
- Heneka MT, O’Banion MK. Inflammatory processes in Alzheimer’s disease. J Neuroimmunol. 2007; 184: 69-91
- Galasko D, Montine TJ. Biomarkers of oxidative damage and inflammation in Alzheimer's disease. Biomark Med. 2010; 4: 27-36.
- Engelhart MJ, Geerlings MI, Ruitenberg A, van Swieten JC, Hofman A, Witteman JC, et al. Dietary intake of antioxidants and risk of Alzheimer disease. JAMA. 2002; 287:3223-9.
- Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Aggarwal N, et al. Dietary Intake of Antioxidant Nutrients and the Risk of Incident Alzheimer Disease in a Biracial C-ommunity Study. JAMA. 2002; 287:3230-7.
- Dai Q, Borenstein AR, Wu Y, Jackson JC, Larson EB. Fruit and vegetable juices and Alzheimer's disease: the Kame Project. Am J Med. 2006; 119:751-9.
- Mancuso C, Bates TE, Butterfield DA, Calafato S, Cornelius C, De Lorenzo A, et al. Natural antioxidants in Alzheimer's disease. Expert Opin Investig Drugs. 2007; 16:1921-31.
- Staehelin HB. Neuronal protection by bioactive nutrients. Int J Vitam Nutr Res. 2008; 78: 282-5.
- Harvey BS, Musgrave IF, Ohlsson KS, Fransson A, Smid SD. The green tea polyphenol (-)-epigallocatechin-3-gallate inhibits amyloid-b evoked fibril formation and neuronal cell death in vitro. Food Chemistry. 2011; 129:1729-36
- Obulesu M, Rao DM. Effect of plant extracts on Alzheimer's disease: An insight into therapeutic avenues. J Neurosci Rural Pract. 2011; 2: 56-61
- Bastianetto S, Ramassamy C, Doré S, Christen Y, Poirier J, Quirion R. The Ginkgo biloba extract (EGb761) protects hippocampal neurons against cell death induced by beta-amyloid. Eur J Neurosci. 2000; 12:1882-90.
- Choi YT, Jung CH, Lee SR, Bae JH, Baek WK, Suh MH, et al. The green tea polyphenol (-)-epigallocatechin gallate attenuates beta-amyloid-induced neurotoxicity in cultured hippocampal neurons. Life Sci. 2001; 70:603-14.
- Li MH, Jang JH, Sun B, Surh YJ. Protective effects of oligomers of grape seed polyphenols against beta-amyloid-induced oxidative cell death. Ann N Y Acad Sci. 2004; 1030:317-29.
- Mishra S, Palanivelu K. The effect of curcumin (turmeric) on Alzheimer's disease: An overview. Ann Indian Acad Neurol. 2008; 11:13-9.
- Craggs L, Kalaria RN. Revisiting dietary antioxidants, neurodegeneration and dementia. Neuroreport. 2011; 22:1-3.
- Choi DY, Lee YJ, Lee SY, Lee YM, Lee HH, Choi IS, et al. Attenuation of scopolamine-induced cognitive dysfunction by obovatol. Arch Pharm Res. 2012; 35:1279-86.
- Lim GP, Chu T, Yang F, Beech W, Frautschy SA, Cole GM. The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J Neurosci. 2001; 21:8370-7.
- Thomas P, Wang YJ, Zhong JH, Kosaraju S, O'Callaghan NJ, Zhou XF, Fenech M. Grape seed polyphenols and curcumin reduce genomic instability events in a transgenic mouse model for Alzheimer's disease. Mutat Res. 2009; 661:25-34.
- Fernández-Fernández L, Comes G, Bolea I, Valente T, Ruiz J, Murtra P, et al. LMN diet, rich in polyphenols and polyunsaturated fatty acids, improves mouse cognitive decline associated withaging and Alzheimer's disease. Behav Brain Res. 2012; 228:261-71.
- Gomez-Pinilla F, Nguyen TT. Natural mood foods:the actions of polyphenols against psychiatric and cognitive disorders. Nutr Neurosci. 2012; 15:127-33
- Anhe FF, Desjardins Y, Pilon G, Dudonne S, Genovese M, Lajolo FM, et al. Polyphenols and type 2 diabetes: A prospective review. PharmaNutrition. 2013; 1:105–114.
- Hakkinen S, Heinonen M, Karenlampi Mykkanen H, Ruuskanen J, Torronen R. Screening of selected favonoids and phenolic acids in 19 berries. Food Res Int. 1999; 32: 345-53.
- Gil MI, Tomas-Barberan FA, Hess-Pierce B, Holcroft DM, Kader AA. Antioxidant Activity of Pomegranate Juice and Its Relationship with Phenolic Composition and Processing. J Agric Food Chem. 2000; 48:4581-89.
- Hartman RE, Shah A, Fagan AM, Schwetye KE, Parsadanian M, Schulman RN, et al. Pomegranate juice decreases amyloid load and improves behavior in a mouse model of Alzheimer's disease. Neurobiol Dis. 2006; 24:506-15.
- Nantitanon W, Yotsawimonwat S, Okonogi S. Factors influencing antioxidant activities and total phenolic content of guava leaf extract. LWT - Food Sci Technol. 2010; 43:1095-1103.
- Landete JM. Ellagitannins, ellagic acid and their derived metabolites: A review about source metabolism, functions and health. Food Res Int. 2011; 44:1150–60.
- Singh K, Khanna AK, Chander R. Hepatoprotective activity of ellagic acid against carbon tetrachloride induced hepatotoxicity in rats. Indian J Exp Biol. 1999; 37: 1025-6.
- Ateşşahín A, Ceríbaşi AO, Yuce A, Bulmus O, Cikim G. Role of Ellagic Acid against Cisplatin-Induced Nephrotoxicity and Oxidative Stress in Rats. Basic Clin Pharmacol Toxicol. 2007; 100:121-6.
- Yüce A, Ateşşahin A, Ceribaşi AO, Aksakal M. Ellagic Acid Prevents Cisplatin-Induced Oxidative Stress in Liver and Heart Tissue of Rats. Basic Clin Pharmacol Toxicol. 2007; 101:345-9.
- Chao PC, Hsu CC, Yin MC. Anti-inflammatory and anti-coagulatory activities of caffeic acid and ellagic acid in cardiac tissue of diabetic mice. Nutr Metab (Lond). 2009; 6:33.
- Özkaya A, Celik S, Yüce A, Şahin Z, Yilmaz O. The Effects of Ellagic Acid on Some Biochemical Parameters in the Liver of Rats Against Oxidative Stress Induced by Aluminum. Kafkas Univ Vet Fak DerG. 2010; 16:263-268.
- Türk G, Sönmez M, Çeribaş AO, Yüce A, Ateşşahin A. Attenuation of cyclosporine A-induced testicular and spermatozoal damages associated with oxidative stress by ellagic acid. Int Immunopharmacol. 2010; 10:177–182
- Papoutsi Z, Kassi E, Chinou I, Halabalaki M, Skaltsounis LA, Moutsatsou P. Walnut extract (Juglans regia L.) and its component ellagic acid exhibit anti-inflammatory activity in human aorta endothelial cells and osteoblastic activity in the cell line KS483. Z. Br J Nutr. 2008; 99:715-22.
- Bae JY, Choi JS, Kang SW, Lee YJ, Park J, Kang YH. Dietary compound ellagic acid alleviates skin wrinkle and inflammation induced by UV-B irradiation. Exp Dermatol. 2010; 19: e182-90.
- Umesalma S, Sudhandiran G. Differential Inhibitory Effects of the Polyphenol Ellagic Acid on Inflammatory Mediators NF-jB, iNOS, COX-2, TNF-a, and IL-6 in 1,2-Dimethylhydrazine-Induced Rat Colon Carcinogenesis. Basic Clin Pharmacol Toxicol. 2010; 107:650-5.
- Rosillo MA, Sánchez-Hidalgo M, Cárdeno A, Aparicio-Soto M, Sánchez-Fidalgo S, Villegas I, et al. Dietary supplementation of an ellagic acid-enriched pomegranate extract attenuates chronic colonic inflammation in rats. Pharmacol Res. 2012; 66:235-42.
- Cornélio Favarin D, Martins Teixeira M, Lemos de Andrade E, de Freitas Alves C, Lazo Chica JE, Artério Sorgi C et al. Anti-Inflammatory Effects of Ellagic Acid on Acute Lung Injury Induced by Acid in Mice. Mediators Inflamm. 2013; 2013:164202.
- Malik A, Afaq S, Shahid M, Akhtar K, Assiri A. Influence of ellagic acid on prostate cancer cell proliferation: A caspase dependent Pathway. Asian Pac J Trop Med. 2011; 4:550-5.
- Srigopalram S, Ilavenil S, Jayraaj IA. Apoptosis associated inhibition of DEN-induced hepatocellular carcinogenesis by ellagic acid in experimental rats. Biomedicine & Preventive Nutrition. 2012; 2:1-8.
- Umesalma S, Sudhandiran G. Ellagic acid prevents rat colon carcinogenesis induced by 1, 2 dimethyl hydrazine through inhibition of AKT-phosphoinositide-3 kinase pathway. Eur J Pharmacol. 2011; 660:249-58.
- Qiu Z, Zhou B, Jin L, Yu H, Liu L, Liu Y et al. In vitro antioxidant and antiproliferative effects of ellagic acid and its colonic metabolite, urolithins, on human bladder cancer T24 cells. Food Chem Toxicol. 2013; 59:428-37.
- Zhao M, Tang SN, Marsh JL, Shankar S, Srivastava RK. Ellagic acid inhibits human pancreatic cancer growth in Balb c nude mice. Cancer Lett. 2013; 337:210-7.
- Malini P, Kanchana G, Rajadurai M. Antibiabetic efficacy of ellagic acid in streptozotoc induced diabetes mellitus in albino wistar rats. Asian J Pharm Clin Res. 2011; 4:124-8.
- You Q, Chen F, Wang X, Jiang Y, Lin S. Anti-diabetic activities of phenolic compounds in muscadine against alpha-glucosidase and pancreatic lipase. LWT - Food Sci Technol. 2012; 46:164-8.
- Akileshwari C, Raghu G, Muthenna P, Mueller NH, Suryanaryana P, Petrash JM et al. Bioflavonoid ellagic acid inhibits aldose reductase: Implications for prevention of diabetic complications. J Funct Foods. 2014; 6:374-83
- Kannan MM,Quine SD. Ellagic acid inhibits cardiac arrhythmias, hypertrophy and hyperlipidaemia during myocardial infarction in rats.Metabolism.2013; 62:52-61.
- Rani UP, Kesavan R, Ganugula R, Avaneesh T, Kumar UP, Reddy GB et al. Ellagic acid inhibits PDGF-BB-induced vascular smooth muscle cell proliferation and prevents atheroma formation in streptozotocin-induced diabetic rats. J Nutr Biochem. 2013; 24:1830-9.
- Hassoun EA, Vodhanel J, Abushaban A. The modulatory effects of ellagic acid and vitamin E succinate on TCDD-induced oxidative stress in different brain regions of rats after subchronic exposure. J Biochem Mol Toxicol. 2004; 18:196-203.
- Pavlica S,Gebhardt R. Protective effects of ellagic and chlorogenic acids against oxidative stress in PC12 cells.Free Radic Res.2005; 39:1377-90.
- Shukitt-Hale B, Lau FC, Carey AN, Galli RL, Spangler EL, Ingram DK, et al. Blueberry polyphenols attenuate kainic acidinduced decrements in cognition and alter inflammatory gene expression in rat hippocampus. Nutr Neurosci. 2008; 11:172-82.
- Tan HP, Wong DZ, Ling SK, Chuah CH, Kadir HA. Neuroprotective activity of galloylated cyanogenic glucosides and hydrolysable tannins isolated from leaves of Phyllagathis rotundifolia. Fitoterapia. 2012; 83:223-9.
- Uzar E, Alp H, Cevik MU, Fırat U, Evliyaoglu O, Tufek A et al. Ellagic acid attenuates oxidative stress on brain and sciatic nerve and improves histopathology of brain in streptozotocin-induced diabetic rats. Neurol Sci. 2012; 33:567-74.
- Gaire BP, Jamarkattel-Pandit N, Lee D, Song J, Kim JY, Park J et al. Terminalia chebulaextract protects OGD-R induced PC12 cell death and inhibits LPS induced microglia activation.Molecules.2013; 18:3529-42.
- Rojanathammanee L, Puig KL, Combs CK. Pomegranate polyphenols and extract inhibit nuclear factor of activated t-cell activity and microglial activation in vitro and in a transgenic mouse model of Alzheimer disease. J Nutr. 2013; 143:597-605.
- Feng Y, Yang SG, Du XT, Zhang X, Sun XX, Zhao M et al. Ellagic acid promotes Aβ42 fibrillization and inhibits Aβ42-induced neurotoxicity. Biochem Biophys Res Commun. 2009; 390:1250-4.
- Wilson GN, Mickley GA, Matera KM. The efficacy of ellagic acid in attenuating neurophysiological and cognitive-behavioral symptoms associated with infusion of amyloid-beta (Aβ) peptide fragments in adult rats. The Baldwin-Wallace College Journal of Research and Creative Studies, Spring 2010; 3:15-30.
- Sheean P, Rout MK, Head RJ, Bennett LE. Modulation of in vitro activity of zymogenic and mature recombinant human β-secretase by dietary plants. FEBS J. 2012; 279:1291-1305.
- Messier C,Gagnon M. Glucose regulation and cognitive functions: relation to Alzheimer's disease and diabetes.Behav Brain Res.1996; 75:1-11.
- Beal MF. Energetics in the pathogenesis of neurodegenerative diseases.Trends Neurosci.2000; 23:298-304.
- Dhingra D, Parle M, Kulkarni SK. Effect of combination of insulin with dextrose, d(-) fructose and diet on learning and memory in mice. Indian J Pharmacol. 2003; 35:151-156.
- Mehan S, Arora R, Sehgal V, Sharma D, Sharma G. Dementia – A Complete Literature Review on Various Mechanisms Involves in Pathogenesis and an Intracerebroventricular Streptozotocin Induced Alzheimer’s Disease. Inflammatory Diseases – Immunopathology, Clinical and Pharmacological Bases. 2012; 4-19.
- Poulose N, Prasad CNV, Haridas PAN, Anilkumar G.Ellagic acid stimulates glucose transport in adipocytes and muscles through AMPK mediated pathway.J Diabetes Metab. 2011; 2:7
- Makino-Wakagi Y, Yoshimura Y, Uzawa Y, Zaima N, Moriyama T, Kawamura Y. Ellagic acid in pomegranate suppresses resistin secretion by a novel regulatory mechanism involving the degradation of intracellular resistin protein in adipocytes. Biochem Biophys Res Commun. 2012; 417:880-5
- Dhingra D, Chhillar R. Antidepressant-like activity of ellagic acid in unstressed and acute immobilization-induced stressed mice. Pharmacol Rep. 2012; 64:796-807.
- Girish C,Raj V,Arya J,Balakrishnan S. Involvement of the GABAergic system in the anxiolytic-like effect of the flavonoid ellagic acid in mice. Eur J Pharmacol.2013; 710:49-58.
- Girish C, Raj V, Arya J, Balakrishnan S. Evidence for the involvement of the monoaminergic system, but not the opioid system in the antidepressant-like activity of ellagic acid in mice. Eur J Pharmacol. 2012; 682:118-25.
- Friedman JI, Adler DN, Davis KL. The role of norepinephrine in the pathophysiology of cognitive disorders: potential applications to the treatment of cognitive dysfunction in schizophrenia and Alzheimer's disease. Biol Psychiatry. 1999; 46:1243-52.
- Brambilla P,Perez J,Barale F,Schettini G,Soares JC. GABAergic dysfunction in mood disorders.Mol Psychiatry.2003; 8:721-37.
- Tatton W, Chen D, Chalmers-Redman R, Wheeler L, Nixon R, Tatton N. Hypothesis for a common basis for neuroprotection in glaucoma and Alzheimer's disease: anti-apoptosis byalpha-2-adrenergic receptor activation. Surv Ophthalmol. 2003; 48:S25-37.
- Wenk GL,McGann K,Hauss-Wegrzyniak B,Rosi S. The toxicity of tumor necrosis factor-alpha upon cholinergic neurons within the nucleus basalis and the role of norepinephrine in the regulation of inflammation: implications for Alzheimer's disease.Neuroscience.2003; 121:719-29.
- Choudary PV, Molnar M, Evans SJ, Tomita H, Li JZ, Vawter MP et al. Altered cortical glutamatergic and GABAergic signal transmission with glial involvement in depression. Proc Natl Acad Sci. 2005; 102:15653-8.
- Ciranna L. Serotonin as a modulator of glutamate- and GABA-mediated neurotransmission: implications in physiological functions and in pathology. Curr Neuropharmacol. 2006; 4:101-14.
- Madsen K,Neumann WJ,Holst K,Marner L,Haahr MT,Lehel Set al. Cerebral serotonin 4 receptors and amyloid-β in early Alzheimer's disease.J Alzheimers Dis.2011; 26:457-66.
- Xu Y,Yan J,Zhou P,Li J,Gao H,Xia Yet al. Neurotransmitter receptors and cognitive dysfunction in Alzheimer's disease and Parkinson's disease.Prog Neurobiol.2012; 97:1-13.
- Chalermpalanupap T, Kinkead B, Hu WT, Kummer MP, Hammerschmidt T, Heneka MT et al. Targeting norepinephrine in mild cognitive impairment and Alzheimer’s disease. Alzheimers Res Ther. 2013; 5:21.
- Yu JT,Wang ND,Ma T,Jiang H,Guan J,Tan L. Roles of β-adrenergic receptors in Alzheimer's disease: implications for novel therapeutics.Brain Res Bull.2011; 84:111-7.
- Coutellier L. Ardestani PM, Shamloo M. β1-adrenergic receptor activation enhances memory in Alzheimer's disease model.Ann Clin Transl Neurol.2014;1:348-60.
- Bierhaus A,Schiekofer S,Schwaninger M,Andrassy M,Humpert PM,Chen Jet al. Diabetes-associated sustained activation of the transcription factor nuclear factor-kappa B.Diabetes.2001; 50:2792-808.
- Cheng X,Wu J,Geng M,Xiong J. The role of synaptic activity in the regulation of amyloid beta levels in Alzheimer's disease.Neurobiol Aging.2014; 35:1217-32.
- Spencer DG, La H. Effects of Anticholinergic Drugs on Learning and Memory. Drug Develop. Res. 1983; 3:489-502
- Chen KC, Baxter MG, Rodefer JS. Central blockade of muscarinic cholinergic receptors disrupts affective and attentional set-shifting. Eur J Neurosci. 2004; 20:1081-8.
- Wang D, Yu R, Lu YQ. Protective effect of Pregnenolone sulfate against scopolamine induced memory impairment in an experimental animal model. Med hypotheses res. 2005; 2:295-302.
- Terry AV Jr. Muscarinic Receptor Antagonists in Rats. In: Levin ED, Buccafusco JJ, editors. Animal Models of Cognitive Impairment. Boca Raton (FL): CRC Press. 2006.
- Lee YK, Yuk DY, Kim TI, Kim YH, Kim KT, Kim KH, et al. Protective effect of the ethanol extract of Magnolia officinalis and 4-O-methylhonokiol on scopolamine-induced memory impairment and the inhibition of acetylcholinesterase activity. J Nat Med. 2009; 63:274-82.
- Kwon SH, Lee HK, Kim JA, Hong SI, Kim HC, Jo TH, et al. Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via anti-acetylcholinesterase andanti-oxidative activities in mice. Eur J Pharmacol. 2010; 649:210-7.
- Liem-Moolenaar M, de Boer P, Timmers M, Schoemaker RC, van Hasselt JG, Schmidt S et al. Pharmacokinetic-pharmacodynamic relationships of central nervous system effects of scopolamine in healthy subjects. Br J Clin Pharmacol. 2011; 71:886-98.
- Sahraei E, Soodi M, Jafarzadeh E, Karimivaghef Z. Investigation of the scopolamine effect on acetylcholinesterase activity. Res Pharmaceutic Sci. 2012; 7.
- Arafa NMS, Abdel-Rahman M, Mahmoud RAHA. Prophylactic Effect of Hypericum Perforatum L. extract in scopolamine rat model of cognitive dysfunction. TOPROCJ. 2013; 4:23-30.
- Kwon SH, Ma SX, Joo HJ, Lee SY, Jang CG. Inhibitory effects of Eucommia ulmoides Oliv. bark on scopolamine induced learning and memory deficits in mice. Biomol Ther (Seoul). 2013; 21:462-9.
- Tsukada H, Yamazaki S, Noda A, Inoue T, Matsuoka N, Kakiuchi T, et al. FK960 [N-(4-acetyl-1-piperazinyl)-p-fluorobenzamide monohydrate], a novel potential antidementia drug, restores the regional cerebral blood flow response abolished by scopolamine but not by HA-966: a positron emission tomography study with unanesthetized rhesus monkeys. Brain Res. 1999; 832:118-23.
- Tsukada H, Kakiuchi T, Ando I, Ouchi Y. Functional activation of cerebral blood flow abolished by scopolamine is reversed by cognitive enhancersassociated with cholinesterase inhibition:a positron emission tomography study in unanesthetized monkeys. J Pharmacol Exp Ther. 1997; 281:1408-14.
- Pachauri SD, Tota S, Khandelwal K, Verma PR, Nath C, Hanif K, et al. Protective effect of fruits of Morinda citrifolia L. on scopolamine induced memory impairment in mice: A behavioral, biochemical and cerebral blood flow study. J Ethnopharmacol. 2012; 139:34-41.
- Tota S, Nath C, Najmi AK, Shukla R, Hanif K. Inhibition of central angiotensin converting enzyme ameliorates scopolamine induced memory impairment in mice: role of cholinergic neurotransmission, cerebral blood flow and brain energy metabolism. Behav Brain Res. 2012; 232:66-76.
- Hebbel RP, Shalev O, Foker W, Rank BH. Inhibition of erythrocyte Ca2+-ATPase by activated oxygen through thiol- and lipid-dependent mechanisms. Biochim Biophys Acta. 1986; 862: 8-16.
- El-Sherbiny DA, Khalifa AE, Attia AS, Eldenshary Eel-D. Hypericum perforatum extract demonstrates antioxidant properties against elevated rat brain oxidative status induced by amnestic dose of scopolamine. Pharmacol Biochem Behav. 2003; 76: 525-33.
- Fan Y, Hu J, Li J, Yang Z, Xin X, Wang J, Ding J, Geng M. Effect of acidic oligosaccharide sugar chain on scopolamine-induced memory impairment in rats and itsrelatedmechanisms. Neurosci Lett. 2005; 374: 222-6.
- Jeong EJ, Lee KY, Kim SH, Sung SH, Kim YC. Cognitive-enhancing and antioxidant activities of iridoid glycosides from Scrophularia buergeriana in scopolamine-treated mice. Eur J Pharmacol. 2008; 588:78-84.
- Hancianu M,Cioanca O,Mihasan M,Hritcu L. Neuroprotective effects of inhaled lavender oil on scopolamine-induced dementia via anti-oxidative activities in rats.Phytomedicine.2013; 20:446-52.
- Jain NK, Patil CS, Kulkarni SK, Singh A. Modulatory role of cyclooxygenase inhibitors in aging- and scopolamine or lipopolysaccharide-induced cognitivedysfunction in mice. Behav Brain Res. 2002; 133:369-76.
- Kim S, Kim DH, Choi JJ, Gu J, Lee CH, Park SJ, et al. Forsythiaside, a Constituent of the Fruits of Forsythia suspense,Ameliorates Scopolamine-Induced Memory Impairment in mice. Biomolecules & Therapeutics. 2009; 17:249-255
- Lee B, Shim I, Lee H, Hahm DH. Rehmannia glutinosa ameliorates scopolamine-induced learning and memory impairment in rats. J Microbiol Biotechnol. 2011; 21:874-83.
- Lee B, Sur B, Shim I, Lee H, Hahm DH. Phellodendron amurense and Its Major Alkaloid Compound, Berberine Ameliorates Scopolamine-Induced Neuronal Impairment and Memory Dysfunction in Rats. Korean J Physiol Pharmacol. 2012; 16:79-89.
- Jang YJ, Kim J, Shim J, Kim CY, Jang JH, Lee KW et al. Decaffeinated coffee prevents scopolamine-induced memory impairment in rats. Behav Brain Res. 2013; 245:113-9.
- Ahmad A, Ramasamy K, Jaafar SM, Majeed AB, Mani V. Total isoflavones from soybean and tempeh reversed scopolamine-induced amnesia, improved cholinergic activities and reduced neuroinflammation in brain. Food Chem Toxicol. 2014; 65:120-8.
- Abd-El-Fattah MA, Abdelakader NF, Zaki HF. Pyrrolidine dithiocarbamate protects against scopolamine-induced cognitive impairment in rats. Eur J Pharmacol. 2014; 723: 330-8.
- Haroutunian V, Greig N, Pei XF, Utsuki T, Gluck R, Acevedo LD et al. Pharmacological modulation of Alzheimer’s b-amyloid precursor protein levels in the CSF of rats with forebrain cholinergic system lesions. Brain Res Mol Brain Res. 1997; 46:161-8
- LiskowskyW, Schliebs R. Muscarinic acetylcholine receptor inhibition in transgenic Alzheimer-like Tg2576 mice by scopolamine favours the amyloidogenic route of processing of amyloid precursor protein. Int. J. Devl Neuroscience. 2006; 24:149-56
- Bihaqi SW, Singh AP, Tiwari M. Supplementation of Convolvulus pluricaulis attenuates scopolamine-induced increased tau and Amyloid precursor protein (AβPP) expression in rat brain.Indian J Pharmacol. 2012; 44: 593-8
- Preston GC,Brazell C,Ward C,Broks P,Traub M,Stahl SM.The scopolamine model of dementia: determination of central cholinomimetic effects of physostigmine on cognition and biochemical markers in man.J Psychopharmacol.1988;2:67-79.
- Wesnes K, Anand R, Lorscheid T. Potential of moclobemide to improve cerebral insufficiency identified using a scopolamine model of aging and dementia. Acta Psychiatr Scand Suppl. 1990; 360:71-2.
- Molchan SE,Mellow AM,Lawlor BA,Weingartner HJ,Cohen RM,Cohen MRet al.TRH attenuates scopolamine-induced memory impairment in humans.Psychopharmacology (Berl).1990; 100:84-9.
- Lines CR, Ambrose JH, Heald A, Traub M. A double-blind, placebo-controlled study of the effects of eptastigmine on scopolamine-induced cognitive deficits in healthy male subjects.Human Psychopharmacology: Clinical and Experimental. 1993; 8: 271-8.
- Gattu M, Boss KL, Terry AV Jr, Buccafusco JJ. Reversal of scopolamine-induced deficits in navigational memory performance by the seed oil of Celastrus paniculatus. Pharmacol Biochem Behav. 1997; 57:793-9.
- Buccafusco JJ. The Revival of Scopolamine Reversal for the Assessment of Cognition-Enhancing Drugs. Methods of Behavior Analysis in Neuroscience. 2nd edition. Boca Raton (FL): CRC Press, 2009.
- Rogers J, Lue LF. Microglial chemotaxis, activation, and phagocytosis of amyloid beta peptide as linked phenomena in Alzheimer’s disease. Neurology. 2001; 39:333-40.
- Sugimoto H, Yamanishi Y, Iimura Y, Kawakami Y. Donepezil hydrochloride (E2020) and other acetylcholinesterase inhibitors. Curr Med Chem. 2000; 7:303-39.
- Bartolini M, Bertucci C, Cavrini V, Andrisano V. beta-Amyloid aggregation induced by human acetylcholinesterase: inhibition studies. Biochem Pharmacol. 2003; 65:407-16.
- Kimura M, Akasofu S, Ogura H, Sawada K. Protective effect of Donepezil against Abeta (1-40) neurotoxicity in rat septal neurons. Brain Res. 2005a; 1047:72-84.
- Kimura M, Komatsu H, Ogura H, Sawada K. Comparison of donepezil and memantine for protective effect against amyloid-beta(1-42) toxicity in rat septal neurons. Neurosci Lett. 2005b; 391:17-21.
- Reale M, Iarlori C, Gambi F, Feliciani C, Isabella L, Gambi D. The acetylcholinesterase inhibitor, Donepezil, regulates a Th2 bias in Alzheimer's disease patients. Neuropharmacology. 2006; 50:606-13.
- Molino I, Colucci L, Fasanaro AM, Traini E, Amenta F. Efficacy of memantine, donepezil, or their association in moderate-severe Alzheimer's disease: a review of clinical trials. Scientific World J. 2013; 2013:925702.
- Yatabe Y, Hashimoto M, Kaneda K, Honda K, Ogawa Y, Yuuki S, et al. Efficacy of increasing donepezil in mild to moderate Alzheimer's disease patients who show a diminished response to 5 mg donepezil: a preliminary study. Psychogeriatrics. 2013; 13:88–93.
- Schwarz RD, Callahan MJ, Davis RE, Jaen JC, Tecle H. Development of M1 Subtype Selective Muscarinic Agonists for Alzheimer’s Disease: Translation of In Vitro Selectivity Into In Vivo Efficacy. Drug Develop Res. 1997; 40:133-43.
- Riedel G, Kang SH, Choi DY, Platt B. Scopolamine-induced deficits in social memory in mice: reversal by donepezil. Behav Brain Res. 2009; 204:217-25.
- Lindner MD, Hogan JB, Hodges DB Jr, Orie AF, Chen P, Corsa JA et al. Donepezil primarily attenuates scopolamine-induced deficits in psychomotor function, with moderate effects on simple conditioning and attention, and small effects on working memory and spatial mapping. Psychopharmacology. 2006; 188:629-40.
- Agrawal R, Tyagi E, Shukla R, Nath C. Effect of insulin and melatonin on acetylcholinesterase activity in the brain of amnesic mice. Behav Brain Res. 2008; 189: 381-86
- Snyder PJ, Bednar MM, Cromer JR, Maruff P. Reversal of scopolamine-induced deficits with a single dose of donepezil, an acetylcholinesterase inhibitor. Alzheimers Dement. 2005; 1:126-35.
- Sumanth M, Sowmya H, Nagaraj SV, Narasimharaju K efficacy of donepezil and galantamine in retrograde amnesia. AJPCR. 2010; 3: 23-25
- Alkalay A, Rabinovici GD, Zimmerman G, Agarwal N, Kaufer D, Miller BL, et al. Plasma acetylcholinesterase activity correlates with intracerebral β-amyloid load. Curr Alzheimer Res. 2013; 10:48-56.
- Sharma M, Gupta YK. Intracerebroventricular injection of streptozotocin in rats produces bothoxidative stress in the brain and cognitive impairment. Life Sci. 2001; 68:1021-9.
- Morris R. Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods. 1984; 11:47-60.
- Kumar A, Dogra S, Prakash A. Neuroprotective Effects of Centella asiatica against Intracerebroventricular Colchicine-Induced Cognitive Impairment and Oxidative Stress. Int J Alzheimers Dis. 2009; 2009: 972178.
- Ellman GL, Courtney KD, Anders V, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity.Biochem Pharmacol. 1961; 7:88-94.
- Wills ED. Mechanism of lipid peroxide formation in animal tissue. Biochem J. 1966; 99:667-76.
- Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys.1959; 82:70-4.
- Misra HP,Fridovich I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase.J Biol Chem.1972; 247:3170-5.
- Aebi H, Wyss, Scherz B, Skvaril F. Heterogeneity of Erythrocyte Catalase II. Isolation and Characterization of Normal and Variant Erythrocyte Catalase and Their Subunits. Eur J Biochem. 1974; 48:137-45.
- Annicchiarico R, Federici A, Pettenati C, Caltagirone C. Rivastigmine in Alzheimer's disease: Cognitive function and quality of life. Ther Clin Risk Manag. 2007; 3:1113-23.
- Fisher A. Cholinergic treatments with emphasis on m1 muscarinic agonists as potential disease-modifying agents for Alzheimer’s disease.Neurotherapeutics. 2008; 5:433-42.
- Raskind MA, Peskind ER, Wessel T, Yuan W. Galantamine in AD: A 6-month randomized, placebo-controlled trial with a 6-month extension. The Galantamine USA-1 Study Group. Neurology. 2000; 54:2261-8.
- Rockwood K, Mintzer J, Truyen L, Wessel T, Wilkinson D. Effects of a flexible galantamine dose in Alzheimer’s disease: a randomized, controlled trial. J Neurol Neurosurg Psychiatry. 2001; 71:589-95.
- Mahadevan S, Park Y. Multifaceted therapeutic benefits of Ginkgo biloba L.: chemistry, efficacy, safety, and uses. J Food Sci. 2008; 73:R14-9.
- Goswami S, Saoji A, Kumar N, Thawani V, Tiwari M, Thawani M. Effect of Bacopa monnieri on Cognitive functions in Alzheimer’s disease patients. Int J Collab Res Internal Med Public Health. 2011; 3:285-92.
- Hajiaghaee R, Akhondzadeh S. Herbal Medicine in the Treatment of Alzheimer’s disease. J Med Plants. 2012; 11:2-7.
- Downey LA, Kean J, Nemeh F, Lau A, Poll A, Gregory R et al. An acute, double-blind, placebo-controlled crossover study of 320 mg and 640 mg doses of a special extract of Bacopa monnieri (CDRI 08) on sustained cognitive performance. Phytother Res. 2013; 27:1407-13.
- Canevelli M, Adali N, Kelaiditi E, Cantet C, Ousset PJ, Cesari M et al. Effects of Gingko biloba supplementation in Alzheimer's disease patients receiving cholinesterase inhibitors: Data from the ICTUS study. Phytomedicine. 2014;21:888-92.
- Di Matteo V, Esposito E. Biochemical and therapeutic effects of antioxidants in the treatment of Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis. Curr Drug Targets CNS Neurol Disord. 2003; 2:95-107.
- McGhie TK, Walton MC, Barnett LE, Vather R, Martin H, Au J, Alspach PA, Booth CL, Kruger MC. Boysenberry and blackcurrant drinks increased the plasma antioxidant capacity in an elderly population but had little effect on other markers of oxidative stress. J Sci Food Agri. 2007; 87: 2519-27.
- Zhou C, Huang Y, Przedborski S. Oxidative stress in Parkinson's disease: a mechanism of pathogenic and therapeutic significance. Ann N Y Acad Sci. 2008; 1147:93-104.
- Beninger RJ, Jhamandas K, Boegman RJ, el-Defrawy SR. Effects of scopolamine and unilateral lesions of the basal forebrain on T-maze spatial discrimination and alternation in rats. Pharmacol Biochem Behav. 1986; 24:1353-60.
- Smith G. Animal models of Alzheimer's disease: experimental cholinergic denervation.Brain Res.1988; 472:103-18.
- Ennaceur A, Meliani K. Effects of physostigmine and scopolamine on rats performances in object-recognition and radial-maze tests. Psychopharmacology (Berl). 1992; 109:321-30.
- Wolff M, Benhassine N, Costet P, Hen R, Segu L, Buhot MC. Delay-dependent working memory impairment in young-adult and aged 5-HT1BKO mice as assessed in a radial-arm water maze. Learn Mem. 2003; 10:401-9.
- Carballo-Márquez A, Vale-Martínez A, Guillazo-Blanch G, Torras-Garcia M, Boix-Trelis N, Martí-Nicolovius M. Differential effects of muscarinic receptor blockade in prelimbic cortex on acquisition and memory formation of anodor-reward task. Learn Mem. 2007; 14:616-24.
- Halder S, Mehta AK, Kar R, Mustafa M, Mediratta PK, Sharma KK. Clove oil reverses learning and memory deficits in scopolamine-treated mice. Planta Med. 2011; 77:830-4.
- Ebert U, Kirch W. Scopolamine model of dementia: electroencephalogram findings and cognitive performance. Eur J Clin Invest. 1998; 28:944-9.
- Itoh J, Nabeshima T, Kameyama T. Utility of an elevated plus-maze for the evaluation of memory in mice: effects of nootropics, scopolamine and electroconvulsive shock. Psychopharmacology (Berl). 1990; 101:27-33.
- Miyazaki S, Imaizumi M, Onodera K. Ameliorating effects of histidine on scopolamine-induced learning deficits using an elevated plus-maze test in mice. Life Sci. 1995; 56:1563-70.
- Kruk-Słomka M, Budzyńska B, Biała G. Involvement of cholinergic receptors in the different stages of memory measured in the modified elevated plus maze test in mice. Pharmacol Rep. 2012; 64:1066-80.
- D'Hooge R, De Deyn PP. Applications of the Morris water maze in the study of learning and memory. Brain Res Rev. 2001; 36:60-90.
- Wisman LA, Sahin G, Maingay M, Leanza G, Kirik D. Functional convergence of dopaminergic and cholinergic input is critical for hippocampus-dependent workingmemory. J Neurosci. 2008; 28: 7797-807.
- Hosseini-Sharifabad A, Mohammadi-Eraghi S, Tabrizian K, Soodi M, Khorshidahmad T, Naghdi N, et al. Effects of training in the Morris water maze on the spatial learning acquisition and VAChT expression in male rats. Daru. 2011; 19:166-72.
- Ma Y, Wang S, Tian Y, Chen L, Li G, Mao J. Disruption of persistent nociceptive behavior in rats with learning impairment. PLoS One. 2013; 8: e74533.
- Wan D, Xue L, Zhu H, Luo Y. Catalpol Induces Neuroprotection and Prevents Memory Dysfunction through the Cholinergic System and BDNF. Evid Based Complement Alternat Med. 2013; 2013:134852.
- Francis PT, Palmer AM, Snape M, Wilcock GK. The cholinergic hypothesis of Alzheimer’s disease: a review of progress. J Neurol Neurosurg Psychiatry. 1999; 66:137-47.
- Deshmukh R, Sharma V, Mehan S, Sharma N, Bedi KL. Amelioration of intracerebroventricular streptozotocin induced cognitive dysfunction and oxidative stress by vinpocetine - a PDE1 inhibitor. Eur J Pharmacol. 2009; 620:49-56.
- Sharma R, Thakur V, Singh SN, Guleria R. Tumor Necrosis Factor and Alzheimer’s disease: A Cause and Consequence Relationship. Bulletin of Clinical Psychopharmacology. 2012; 22:86-97
- Praticò D, Delanty N. Oxidative injury in diseases of the central nervous system: focus on Alzheimer's disease. Am J Med. 2000; 109:577-85.
- Zana M, Janka Z, Kálmán J. Oxidative stress: a bridge between Down's syndrome and Alzheimer's disease. Neurobiol Aging. 2007; 28:648-76.
- Yan Z, Feng J. Alzheimer’s Disease: Interactions between Cholinergic Functions and beta-amyloid. Curr Alzheimer Res. 2004; 1:241-8.
- Smith MA, Rottkamp CA, Nunomura A, Raina AK, Perry G. Oxidative stress in Alzheimer's disease. Biochim Biophys Acta. 2000; 1502:139-44.
- McGrath LT, McGleenon BM, Brennan S, McColl D, McILroy S, Passmore AP. Increased oxidative stress in Alzheimer's disease as assessed with 4-hydroxynonenal but not malondialdehyde. QJM. 2001; 94:485-90.
- Perry G, Cash AD, Smith MA. Alzheimer Disease and Oxidative Stress. J Biomed Biotechnol. 2002; 2:120-123.
- Gella A, Durany N. Oxidative stress in Alzheimer disease. Cell Adh Migr. 2009; 3:88-93.
- Balu M, Sangeetha P, Haripriya D, Panneerselvam C. Rejuvenation of antioxidant system in central nervous system of aged rats by grape seed extract. Neurosci Lett. 2005; 383:295-300.
- Attrey DP,Singh AK,Naved T,Roy B. Effect of seabuckthorn extract on scopolamine induced cognitive impairment.Indian J Exp Biol.2012; 50:690-5.
- Lobo V, Patil A, Phatak A, Chandra N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn Rev. 2010; 4: 118–126.
- Rahman K. Studies on free radicals, antioxidants, and co-factors. Clin Interv Aging. 2007; 2:219-36.
- Mangialasche F, Polidori MC, Monastero R, Ercolani S, Camarda C, Cecchetti R, Mecocci P. Biomarkers of oxidative and nitrosative damage in Alzheimer's disease and mild cognitive impairment. Ageing Res Rev. 2009; 8:285-305.
- Uttara B,Singh AV,Zamboni P,Mahajan RT. Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeuticoptions.Curr Neuropharmacol.2009; 7:65-74.
- Younes M, Siegers CP. Mechanistic aspects of enhanced lipid peroxidation following glutathione depletion in vivo. Chem Biol Interact. 1981; 34:257-66.
- Casini AF, Pompella A, Comporti M. Liver glutathione depletion induced by bromobenzene, iodobenzene, and diethylmaleate poisoning and its relation to lipid peroxidation and necrosis. Am J Pathol. 1985; 118:225-37.
- Schuessel K,Leutner S,Cairns NJ,Müller WE,Eckert A. Impact of gender on upregulation of antioxidant defence mechanisms in Alzheimer's disease brain.J Neural Transm.2004; 111:1167-82.
- Desagher S, Glowinski J, Premont J. Astrocytes protect neurons from hydrogen peroxide toxicity. J Neurosci. 1996; 16:2553-62.
- Mulier B, Rahman I, Watchorn T, Donaldson K, MacNee W, Jeffery PK. Hydrogen peroxide-induced epithelial injury: the protective role of intracellular nonprotein thiols (NPSH). Eur Respir J. 1998; 11:384-91.
- Dringen R, Gutterer JM, Hirrlinger J. Glutathione metabolism in brain metabolic interaction between astrocytes and neurons in the defense against reactive oxygen species. Eur J Biochem. 2000; 267:4912-6.
- Mann H,McCoy MT,Subramaniam J,Van Remmen H,Cadet JL. Overexpression of superoxide dismutase and catalase in immortalized neural cells: toxic effects of hydrogenperoxide.Brain Res.1997; 770:163-8.
- Blokland A. Acetylcholine: a neurotransmitter for learning and memory?Brain Res Brain Res Rev.1995; 21:285-300.
- Oda Y. Choline acetyltransferase: the structure, distribution and pathologic changes in the central nervous system. Pathol Int. 1999; 49:921-37.



