The Effect of Packed Column Addition to the Performance of Biogas Stove
Syamsuri1,*, Nyoman Puspa Asri2, Yustia Wulandari3, Suheni1,
Muhammad Ali1
1Mechanical Engineering Department, Institut Teknologi Adhi Tama Surabaya, Surabaya, Indonesia
2Chemical Engineering Department, Universitas WR Supratman Surabaya, Surabaya, Indonesia
3Chemical Engineering Department, Institut Teknologi Adhi Tama Surabaya, Surabaya, Indonesia
Abstract
Keywords
Absorption, Biogas, CH4, Packed Column, K2CO3
Received: August 9, 2016
Accepted: August 19, 2016
Published online: September 3, 2016
@ 2016 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY license. http://creativecommons.org/licenses/by/4.0/
1. Introduction
Human dependence upon fossil fuels caused the reserves of these energy sources are gradually reduced, in addition to the impact on the environment, such as air pollution. In situations like this, the search, development, and deployment of new alternative energy technologies that are cheap, environmentally friendly and renewable (renewable) energy such as solar, wind energy, water energy and other alternative energy sources become important.Especially when there is an increase in fuel prices will have an impact on the poor who are most affected by this price increase.Alternative energy sources have been found as a substitute for fuel, one of which is Biogas. Research on biogas was generally conducted by previous researchers, among others: [1], [2], [3], [4], [5], [6], [7], [8], [9], and [10].
Several methods of purifying biogas i.e. physical absorption, chemical absorption, the absorption of the surface of a solid, cryogenic methods, methods of chemical conversion and separation by membrane. Each method has advantages and disadvantages in terms of economic processes, raw materials, technology and operation. It takes an easy, simple and easy on the raw material that can be for a biogas purification process.
Efforts to purify biogas from CO2 gases have been carried out using absorbent K2CO3 in a packed column.
Next [11] conducted a research by comparing the rate of absorption and balance, vapor- liquid CO2 in solution K2CO3 hot, with DEA and DEA solution with stericaly hindered amines.
Another research is research by CO2 removal where absorbennya K2CO3 in a packed column and the stripper such as a tray columns which is carried out by [12].
Next is the study by [13] studying the process of stripping CO2 from a solution of K2CO3 / KHCO3 with a piperazine catalyst using a speed -based approach.
Then [14] conducted a simulation of the CO2 recovery using the absorption column and sripping along with CO2 purification unit for a gas-turbine exhaust gases.
Later [15] upgrading the quality of biogas by removing CO2 and H2S using a packed column reactor. The liquid solution used is sodium hydroxide (NaOH), calcium hydroxide (Ca(OH)2) and mono-ethanolamine (MEA). Liquid solvent circulated through the column, direct contact with biogas flows through the opposite (counter flow). This technique proved to promise to upgrade the quality of biogas.
This study specifically learns about the effect of adding a packed column and without a packed column with absorbent K2CO3 to the performance of biogas stove where this research has not been done by previous researchers.
1.1. Definition of Biogas
Biogas is a gas produced from the decomposition of organic matter, carried out in anaerobic (closed on free air) to produce a gas which is mostly methane (which has flammable properties) and carbon dioxide. The gas formed is called swamp gas or biogas. A good temperature, for fermentation is 30°-55°C. At this temperature, microorganisms can work optimally, remodel organic ingredients. In general, biogas is a mixture of 50-70% methane [CH4], 30-40% of carbon dioxide [CO2], 5-10% hydrogen gas [H2] and the rest of other gases.
Table 1. Biogas composition and amount in a bio-gas unit.
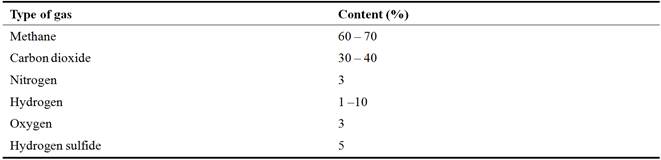
Source: [1].
Judging from the huge potential of biogas then do the assessment and making installation as well want to know the extent to which alternative energy biogas by using absorbent can be developed by way of the test this fuel on a biogas stove.
1.2. Stage of Mixing / Absorption
Because the solvents used are K2CO3 then absorption will occur chemically because of chemical reactions directly between CO2 with a solution of K2CO3. The process of absorption or the separation of CO2 by K2CO3 can be seen in the following reaction:
CO2(aq) + K2CO3(aq) + H2O ![]() 2KHCO3(aq)
2KHCO3(aq)
Absorption above is a chemical reaction that occurs, due to the occurrence of a chemical reaction directly between CO2 with a solution of K2CO3. But in the liquid phase, in addition there is a thin layer of liquid also contained the reaction zone. The chemical reaction that occurs is irreversible, where the CO2 in the gas phase will be absor bed by the liquid phase K2CO3 solution.
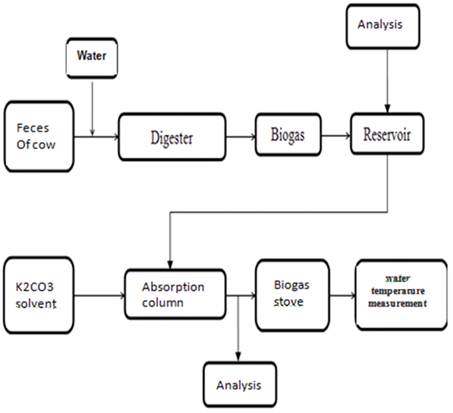
Figure 1. Research scheme.
1.3. Working Principle of Packed Column
a. Absorption column is a column in which, there are different substances phase flow in opposite directions which can cause chemical components transferred from one liquid phase to another phase occurring almost every chemical reactor. This process can be either gas absorption, distillation, dissolution occurs in all chemical reactions.
b. The gas mixture which is the output of the reactor is fed down absorber tower. In the absorber, there will be contact between the two phases namely the gas phase and liquid phase resulting in diffusional mass transfer in a feed gas from the bottom of the tower.
2. Method
2.1. Location and Time Research
Making the installation of the reactor for the production of biogas with a capacity of a reservoir 1 m3 / day and retrieval of data conducted in June - December 2015 on the island of Madura more precisely in the City Bangkalan, the village of Tanah Merah, Desah Dlambah Dajah.
2.2. Stages of Research
This research was conducted through experiments with the stage activities as follows: Preparation of reactor installations for biogas production, preparation of biogas, setup tool absorption/packed column, preparation of research procedures, preparations of the gauges are calibrated, measurement and data retrieval. After that analysis of the data research, writing and reporting of research results.
2.3. Water Boilling Test
In subsequent experiments then simply replace the burner of the stove with the burner of the second (hole 3 mm) and third (hole 4 mm) and the steps are the same as is done in the first trial.

Figure 2. Experiments with Water BoillingTest.
2.4. The Performance of the Stove Power of Stove
To calculate the amount of power from biogas stoves used the following equation [16]:
![]() (1)
(1)
where: P is the power of the stove (kW), mf is the fuel consumption during the period of time t (kg), E is the Lower Heating Value (LHV) b.bakar, kJ / kg. bb and t is the time of the test (s).
Efficiency
Efficiency is the ratio of useful heat divided by the heat supplied by the fuel during the test. The equation used is as follows [16],
![]() (2)
(2)
where: η is the total efficiency of the stove, mw is the mass of water which is heated, kg, mpa is the mass of the pan used (kg), Cp is the specific heat of water, kJ / kg, Cpa is specific heat pan, kJ/kg, T2 is the temperature of boiling water °C, T1 is the initial temperature °C water, ms is the mass of water vapor (kg), mf is the mass of fuel used (kg), hfg is the latent heat of vaporization of water, and E is Lower Heating Value (LHV).
3. Result and Discussions
3.1. Analysis of Power Stove
Data obtained from experiments using a burner with fire pit diameter 2 mm were as follows:
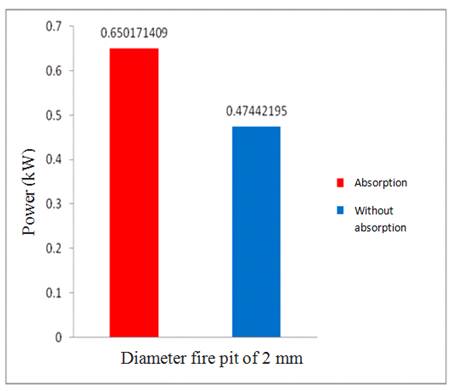
Figure 3. Comparison of the power of the stove for a device that uses the absorption and without absorption with the burner diameter is 2 mm.
By Figure 3 it can be concluded that, overall, the presence of absorption will increase the power of the stove and overall as well with the absorption of substances impurities, which are still present in biogas then biogas becomes purer, so that the calorific value becomes better. This corresponds to the research conducted by [15].
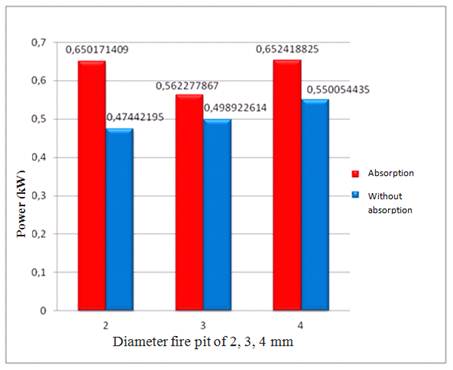
Figure 4. Comparison of power from the stove to the tool using absorption and without absorption for a wide range of diameters burner.
3.2. Analysis for the Efficient Stoves
In the same way, then the calculation of the efficiency obtained in all subsequent experiments by using Microsoft Excel. More results can be seen from the table below:
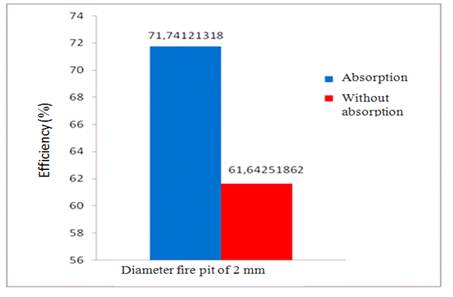
Figure 5. The influence of absorption and without absorption to the efficiency of the burner 2 mm diameter.
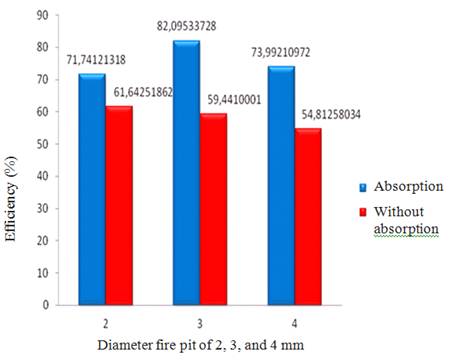
Figure 6. Comparison of efficiency of a biogas stove using the tool absorbs and without absorption for all burner with a diameter of 2, 3, and 4 mm.
Through Figure 6 indicates that, overall efficiency of biogas stove using the absorption is greater than the efficiency of biogas stoves without using absorption. This is caused using absorption, the heat produced is greater than without absorption.
3.3. Analysis of the Mass of Steam Against Time
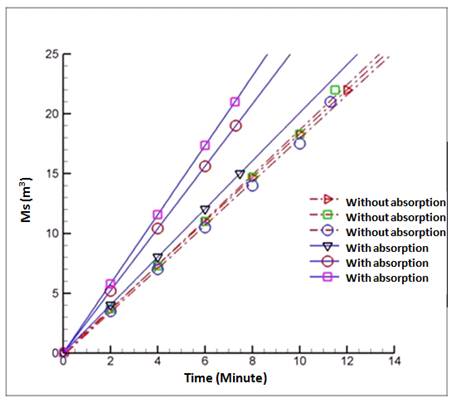
Figure 7. The relationship between the mass of steam (Ms) against time.
From the inside of Figure 7 seen that, with the rise time the mass of evaporated water will further increase. There are interesting things here, namely in the picture the increased difference in mass of steam and shown in a fire pit diameter of 4 mm. The increase in the mass of water that evaporates more quickly than others. This is because with a diameter fire pit on a stove burner which is great then the heat is supplied also getting bigger. The temperature will increase and consequently, the mass of vapor produced is also faster increases. This is in accordance with the equation of latent heat [17] namely:
q = Ms. Hfg
In the equation when the value of q greater than the value to the vapor mass (Ms) was also great.
4. Conclusions
1.In the experiment Water Boilling Test, using 4 mm diameter burner obtained power of 0.65 kW and for no absorption 0.55 kW. The power is greater than the diameter of burner 2mm and 3mm.
2.Efficiency results obtained from experiments using either absorption or no absorption. For the hole diameter of the burner 4 mm by absorption is 73.99% and 54.81% without absorption. This efficiency larger than the hole diameter of the burner 2mm and 3mm.
3. The fastest time of boiling water is obtained by using a burner which has a diameter of 4 mm over a period of 11 minutes 30 seconds without absoption and 7 minutes 26 seconds with absorption.
Acknowledgments
This research was funded through Competitive Research Grant No. 051 / SP2H / PKM / K7 / KM / 2015 from the government.
References
- Meynell D.S.1976. Methane: Planning a digester.Great Britain: Prism Press.
- Sihombing D.T.H.1980. Prospects use of biogas for rural energy in Indonesia, LPL, No. 11 Tahun XIV,LEMIGAS, Jakarta, Indonesia.
- Indartono Y. S.2005. Biogas reactor for small and medium scale (part-one). http://www.beritaiptek.com/statik.php [11 January, 2007].
- Widodo T. W., Asari Ahmad.,Nurhasanah A. and Rahmarestia E.2006. Engineering and testing biogas reactor for farmer group scale, Balai Besar Pengembangan Mekanisasi Pertanian, Jurnal Enjinering Pertanian. pp. 41-52.
- Sofian A.2008. Improving the quality of biogas as a fuel combustion engine by way of a reduction in the levels of CO2 in the biogas using slurry of Ca (OH) 2,Jurusan Teknik Mesin-Fakultas Teknik, Universitas Muhamadiyah Surakarta.
- Jianbin Guo, Joachim Clemens, Xiaoping Li, Pan Xu and Renjie Dong.2012. Performance evaluation of a Chinese medium-sized agricultural biogas plant at ambient temperature,Engineering in Life Sciences, Vol.12,Issue 3, pp. 336–342.
- Cassiano Rossetto, Samuel Nelson Melegari de Souza, Reginaldo Ferreira Santo, Juliano de Souzaand Otávia Lidia Klaus.2013. Performance of an otto cycle engine using biogas as fuel, AfricanJournal of Agricultural Research, Vol. 8(45), pp. 5607-5610.
- Chamarthi S., N.Srinivasa Reddy, and G Ravi Chandra.2013. Experimental investigation onperformance improvement of biogas plant, International Journal of Engineering Research andApplications, Vol. 3, Issue 3, pp.1218-1223.
- Rabiu, A., Yaakub, H., Liang, J. B., and Samsudin, A. A.2014. Enhancing biogas production rate ofcattle manure using rumen fluid of ruminants, IOSR Journal of Agriculture and Veterinary Science, Vol.7, Issue 3, pp. 25-28.
- Patrick Mukumba, Golden Makaka, and Chipo Shonhiwa.2015. An assessment of the performance of a biogas digester when insulated with sawdust, International Journal of Energy and Power Engineering, Vol. 4, Issue 2, pp. 24-31.
- Satoni,G, dan Savage,D.W.1983. Sterically hindered amines for CO2 removal fromgases. Ind.Eng.Chem.Fundam, 22, 239.
- Alatiqi, I.M., Dadkhah, A.A., Akbar,A.M., dan Hamouda,H.F. 1994. Comparison between dynamics and control performance of mesophilic and thermophilic anaerobic sludge digesters. Chem. Eng. J., 55, B55-B66 (1994).
- Oyenekan, Rochelle, dan Abo. 2007. Alternative Stripper Configurations for CO2 Capture by Aqueous Amines. AIChE Journal, vol. 53, No. 12, pp. 3144-3154.
- Mofarahi M., Khojasteh Y., Khaledi H., dan Farahnak A. 2008. Design of CO2 absorption plant for recovery of CO2 from flue gases of gas turbine. Energy 33, 1311-1319.
- Tippayawong, N., Thanompongchart, P. 2010. Biogas quality upgrade by simultaneous removal of CO2 and H2S in a packed column reactor. Energy, 35, 4531-4535.
- Bhattacharya S.C., Kumar S., Leon M.A. and Khang A.M. (2003). Design and performance of natural draft gasifier stove for use in institutional and industrial. International Conference. Yogyakarta, Indonesia.
- Incropera & David.2011. Fundamentals of heat and mass transfer. 7th Ed. John Wiley and Sons Inc. New York, USA.



