Risk Factors Associated with Gastroenteritis in Children 2-5 Years of Age Attending Rehman Medical Institute Peshawar
Ayesha Mushtaq1, Saleem Khan1, Falak Zeb1, *, Qurratul Ain1, Ayesha Syed1, Fozia Khattak2, Hidayatullah1
1Department of Human Nutrition, the University of Agriculture, Peshawar, Pakistan
2Gynecology Department, Government Maternity Hospital Hashtnaghri, Peshawar, Pakistan
Abstract
A case control study was conducted to determine the risk factors associated with gastroenteritis in children 2-5 years of age at Rehman Medical Institute Hayatabad, Peshawar during Oct, 2014 to Feb, 2015. A total of 200 children mothers (100 cases and 100 controls) were interviewed regarding socio-demographic variables, anthropometric measurements, mother’s knowledge and approach during disease, hygiene and sanitation status. Data was analyzed by SPSS and standard chi-square test was applied on categorical data to establish an association between the variables at 5% level of significance; t-test was used for quantitative data analysis of mean comparison. The results showed that 44% of children with gastroenteritis had low socioeconomic status, 25% children had family income <12000 rupees. Nutritional status of children showed that 10% controls and 27% cases were severely malnourished. Wasting was recorded 3% in controls and 10% in cases. The percentage of underweight was 27% and 35% in controls and cases respectively. Stunting was found lower in controls (26%) and higher in cases (46%). Significant difference was found in height for age, mid upper arm circumference and weight for height z-score. Odds ratio (OR) for the association of gastroenteritis with joint family structure was 3.11 (95% CI 1.736-5.594), weight for height z-score was 3.593 (95% CI: 958-0.863), weight for age z-score was 1.456 (95% CI: 0.796-2.661), age of complementary feeding <6 months was 2.405 (95% CI: 1.233-4.689), child eating picked stuff 3.977 (95% CI: 2.180-7.256), child admitted in hospital 1.860 (95% CI: 1.008-3.432), child who does not washed hand before meal was 0.288 (95% CI: 0.164-0.677) and child who does not washed hand after attending washing were 0.288 (95% CI: 0.161-0.517). It was concluded that low socioeconomic status, parental education, malnutrition, poor hygiene and inappropriate complementary feeding are associated with gastroenteritis.
Keywords
Gastroenteritis, Anthropometry, Stunting, Poor Hygiene
Received: June 20, 2016
Accepted: July 1, 2016
Published online: August 16, 2016
@ 2016 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY license. http://creativecommons.org/licenses/by/4.0/
1. Introduction
Gastroenteritis is the inflammation of gastrointestinal tract exemplified by a combination of abdominal pain, cramping, nausea, vomiting, diarrhea and dehydration. It may be acute or chronic as acute gastroenteritis usually lasts less than 14 days, while chronic gastroenteritis lasts among 14 and 30 days [1]. It can have an effect on individuals of any age and is a major health hazard [2]. Chronic gastroenteritis is the most widespread cause of childhood mortality [3]. Diarrhea stays one of the most common illnesses in children around the world [4]. Gastroenteritis is primarily caused by viruses, but protozoa, bacteria and helminthes may also be the cause in developing countries [5]. In developed countries almost 87% of the acute gastroenteritis is caused by virus and of which rotavirus is the most common. In infants 40% of the cases of diarrhea are attributed to rotavirus globally. Rotavirus was responsible for almost 145,000 deaths each year in Asia, with the maximum numbers taking place in India, Pakistan, and Indonesia. An estimated 1 in 40 infants experience a severe episode of rotavirus gastroenteritis annually in Pakistan [6]. Breastfeeding is thought to be protective factor against gastrointestinal infections [7]. Zinc supplementation along with ORS is found to be highly effective in the management of acute gastroenteritis [8,9].
In acute diarrhea excessive fluid and electrolyte losses leads to dehydration and acidosis which is the main cause of death. By providing fluid on time and replacing electrolytes losses greater part of deaths are preventable. Other direct outcomes of gastroenteritis in children in developing countries are malnutrition reduced growth and impaired cognitive development [10]. Acute gastroenteritis is the main cause of morbidity in developed countries [11]. Malnutrition raises the occurrence and severity of diarrhea and other infections [12]. The basic reason for malnutrition is diarrhea changing from minor to severe and children suffer from loss of appetite, food restriction and malabsorption syndrome [13].
Most important risk factors associated with gastroenteritis are diarrhea, and its occurrence remains a remarkable burden on children in low and middle income countries [14] due to various elements [15] like child malnutrition [16] low socioeconomic status and low education of mothers [17,18] lack of safe drinking water, inadequate sanitation and poor hygiene [19,20] crowding [21] and low maternal age [22]. Moreover, diarrheal incidence turn down as a child grows older is greatest in the first two years of life, environmental factors and hospitalization [23] and unemployment [24]. The behaviors that promote gastroenteritis are lack of hand washing after defecating and before food handling [25].
Rotavirus gastroenteritis is passed on through the fecal oral means [26]. Other modes of transmission may include indigestion of food and water infected by fecal matter, person to person contact, or direct contact with feces [27]. It can also be transmitted through respiratory droplets and hands [28]. Some studies reported that contaminated food and water are responsible for 70% of all cases of diarrhea [29,30]. Sometimes the basis of gastroenteritis are inappropriately cooked food or reheating of meat, dairy, seafood, bakery products and non-infectious causes may include poisoning with heavy metals like arsenic and cadmium [12]. Unhygienic and unsafe environment put children at risk of death [31]. In children with acute gastroenteritis breastfeeding should be sustained at all times, even during the initial rehydration phase. On the other hand widespread contradictions about lactose-free or lactose-reduced formulas are found but do not have to be removed and usually are needless. Banana, rice, apple, toast, cereals and potatoes diet should be offered [32]. Probiotics builds an improved equilibrium in intestinal micro flora and promotes best possible health and are very useful in reducing and preventing the length and severity of illnesses related to diarrhea [33].
Since Pakistan harbors a high burden of gastroenteritis infection and very limited studies related to gastroenteritis have been conducted on children so the present study was hypothesized to assess the risk factors associated with gastroenteritis in children 2-5 years of age in North West of Pakistan as children are more prone to gastroenteritis.
2. Material and Methods
2.1. Design and Location
This case-control study was conducted in Rehman Medical Institute, Hayatabad Peshawar- Pakistan.
2.2. Sample Selection and Criteria
A total of 200 children were randomly selected (100 cases and 100 controls) for determination of risk factors associated with gastroenteritis in children 2 to 5 years of age. Inclusion criteria for cases involved children with gastroenteritis and controls were healthy children. An exclusion criterion for cases was children who were admitted in ICU or seriously ill patients other than gastroenteritis and controls were children with diseases.
2.3. Study Protocol
The approval for conducting the study was taken from the Chief Executive of the Rehman Medical Institute Peshawar. Pediatric ward of hospital was used as a platform for enrolled subjects. Children admitted in Pediatric ward were interviewed for different risk factors associated with gastroenteritis through standard questionnaire. The subjects meeting the inclusion criteria of cases were enrolled for the study and informed consent was obtained prior to their enrollment.
2.4. Data Collection
A questionnaire was used to collect data regarding socio-demographic variables, anthropometric measurements, and assessment of different risk factors associated with gastroenteritis in children.
2.5. Demographic and Socioeconomic Status
Demographic and socioeconomic characteristics of the subjects such as family type, education, monthly income, occupation were assessed by interviewing the subjects and recording their responses in the questionnaire.
2.6. Anthropometric Measurement
Weight, Height and Mid Upper Arm Circumference was measured on the day of admission. The calibrated equipments were used for anthropometric measurements. Weight was measured on pediatric beam scale. Children were weighed standing on a scale. All heavy clothes and hair ornaments were removed while weighing. Height was measured using height board (stadiometer), height was measured standing upright, children greater than 2 years or 87 cm. Age of patient was asked from the mother. Mid Upper Arm Circumference (MUAC) was measured by MUAC tape.
2.7. Clinical and Health Assessment
Parents of the subjects were asked about different symptoms associated with gastroenteritis and their response was recorded in questionnaire. Mothers of subjects were interviewed about their child’s feeding history such as breastfeeding, weaning food, type of milk, mother’s approach when child gets diarrhea and care imparting behavior of mother. They were also asked about their sanitation and hygiene status such as source of drinking water, hand hygiene and garbage disposal.
2.8. Statistical Analysis
Data was entered into the Statistical package for social sciences (SPSS) for error checking and statistical analysis. Descriptive statistics including mean, median, frequency, and standard deviation were determined and checked for distribution of the data to apply appropriate statistics. Standard chi-square test was applied on categorical data to establish an association between the variables at 5% level of significance; t-test was used for quantitative data analysis for mean comparison. Odds ratio was applied to estimate the relationship between risk factors and gastroenteritis.
3. Results and Discussion
3.1. Socio Demographic Characteristics of the Children
Age of the subjects was 2-5 years with mean age of controls 3.56±1.19 and case was 3.11±1.07 years. No significant difference (p < 0.005) was found in patient’s father occupation, mother occupation, and family size between the two groups while significant difference was found in patient’s age, gender, patient’s education, patient’s mother education, family type, family income and socioeconomic status. The results indicate that low education level of parents; poor socioeconomic status and low family income may be the risk factors leads to gastroenteritis. Zolaly et al. (2011) reported that the mean age of the children was 4.4±2.72 years for cases and significant difference (p=0.000) was found between three age groups. He also reported that 1.6% fathers were illiterate, 12.1% had educational level below intermediate and 86.3% had greater than intermediate with a significant difference of p= 0.000 while 4.8% mothers of were illiterate, 24.2% were below intermediate and 71% had educational level greater than intermediate with significant difference of p= 0.000 between three different levels of education [34]. Rheingans et al. (2009) reported that in Bangladesh 33% and in Pakistan 61% children with gastroenteritis were not going to school [35]. Bhandari et al. (1989) reported that 24% father whose child is suffering from gastroenteritis had no education [36]. Mansour et al. (2013) reported that 36% of the cases mothers were illiterate as reported in the present study [37]. Kurugol et al. (2003) reported that low income was found in 24% controls and 27.8% cases [38]. Nguyen et al. (2006) reported that socio demographic status was unsatisfactory in 25.9% and satisfactory in 20.6% cases while in controls 23.3% had unsatisfactory and 21.3% satisfactory level with a significant difference of p=0.006 respectively [39]. Ahmed et al. (1995) conducted a study in Karachi and reported that in family income <2000 rupees per month was 36% chronic gastroenteritis patients and 24% acute gastroenteritis patients [40]. Augistina et al. (2013) conducted a study in East Jakarta and reported that 52% gastroenteritis patients had nuclear and 48% had joint family structures and socioeconomic status reported to be very low in 54% cases and medium-low in 46% of the case group [41]. Khattak et al. (2007) reported that family income was less than Pakistani 5000/rupees per month in 60% children with gastroenteritis while 40% had income greater than 5000 rupees [42].
Table 1. Socio demographic characteristics of the Cohort (n=200).
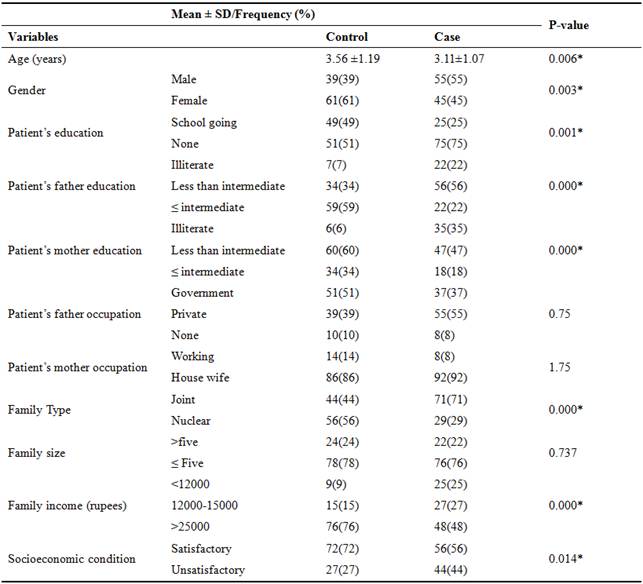
SD=Standard deviation, * significant at p<0.05, p=probability, %=percentage.
3.2. Anthropometric Measurements of the Subjects
Weight, height, MUAC, weight for height z-score, weight for age z-score, height for age z-score of controls was reported 15.39±3.36, 96.60±11.29, 15.32±2.13, 15.32±2.13, 0.09±1.15 and -0.68±1.49 while cases were 13.08±3.02, 89.44±10.51, 13.78±2.04, 0.22±1.2, -0.77±1.52 and -1.68![]() 1.39 respectively. As these anthropometrics indicates the nutritional status of the individuals, so no significant difference was found in weight for height z-score whereas there was significant difference (p>0.005) in the rest of the parameters with in both groups. Mostly in gastroenteritis dehydration occurs and it leads to muscles wasting which elevate the anthropometrics of the children. This significant difference in anthropometric measurements indicates that by losing nutrients, body fluids and low absorption of nutrients may affect nutritional status of children in gastroenteritis.
1.39 respectively. As these anthropometrics indicates the nutritional status of the individuals, so no significant difference was found in weight for height z-score whereas there was significant difference (p>0.005) in the rest of the parameters with in both groups. Mostly in gastroenteritis dehydration occurs and it leads to muscles wasting which elevate the anthropometrics of the children. This significant difference in anthropometric measurements indicates that by losing nutrients, body fluids and low absorption of nutrients may affect nutritional status of children in gastroenteritis.
Table 2. Anthropometric Measurements of the Cohort (n=200).
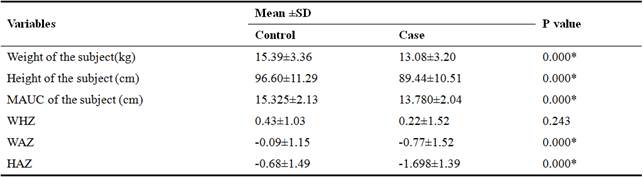
MUAC= mid upper arm circumference, WHZ=Weight for height z-score, WAZ=Weight for age z-score, HAZ=Height for age z-score, p=probability, SD=standard deviation, %=percentage, *significant at p<0.05, p=probability.
3.3. Nutritional Status of the Subjects
The study suggest that on the basis of MUAC severely malnourished children were 10% and 27%, moderately malnourished 11% and 20% and healthy were 79% and 53% in control and cases group respectively. According to WHZ, wasting in children was recorded 3% in control and 10% in case group. The percentage of underweight children in control and case group was 27% and 35% respectively. Stunting was found 26% in controls and 46% in cases whereas 74% of the children in control group were normal and 54% in case group and significant difference was found in Height for age z-score, MUAC z-score and Weight for height z-score while non-significant results were observed in Weight for age z-score. Mwangome et al. (2011) revealed that severe malnutrition was found in 13% of cases, moderate in 21% and healthy were 66% on the basis of MUAC in gastroenteritis patients and observed significant difference (p= 0.001) between three categories of MUAC which is partially similar to our findings [43]. Augistina et al. (2013) reported that 32% children showed stunting, 19% underweight and 12% wasting and significant difference of (p<0.005) was found between three variables [41]. Nitiemia et al. (2011) considered that 31% children with gastroenteritis suffered from stunting [44]. Opintan et al. (2010) conducted a study on Pediatric diarrhea in Southern Ghana and reported that weight for age z-score <-2 in 16.7% cases and 10% controls showed underweight, weight for height z-score <-2 in 22% cases and 6.3% controls showed wasting and height for age z-score <-2 in 37.2% cases and 14.3% controls showed stunting in gastroenteritis children [45].
Table 3. Nutritional status of the subjects (n=200).
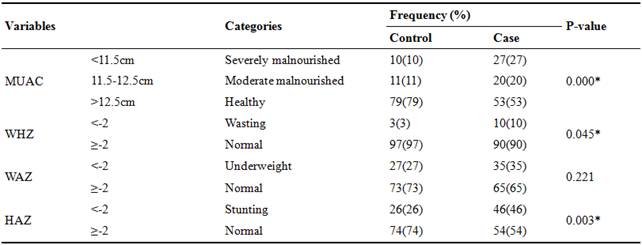
MUAC= mid upper arm circumference, WHZ=weight for height z-score, WAZ=weight for age z-score, HAZ=height for age z-score, p=probability, SD=standard deviation, %=percentage, * significant at p<0.05, p=probability.
3.4. Influence of Mother’s Education on Child Health
The study reported significant difference in feeding pattern, child’s daily meal intake, child taking lunch from home, mother’s approach when child gets diarrhea, boiled water consumed, child’s habit of eating picked stuff, boiling of feeder while non-significant difference was reported in age of complementary feeding, ORS offered to child, consumption of water by the child during diarrhea, hospitalization for overnight during diarrhea and increase in weight and height of child. From the above result, it has been revealed that mother education regarding adequate feeding practices, adequate child meal intake and child eating habits may improve gastroenteritis. Khattak et al. (2007) conducted a study in Peshawar and reported that 39% of the children with gastroenteritis started complementary feeding less than six months of age and 49.5% of the cases started complementary feeding greater than six months of age. He also reported that boiled water was consumed by 46.6% gastroenteritis children [42]. Van Derslice et al. (1994) reported in Philippines that 19% of the cases started complementary feeding less than six months of age [46]. Quigely et al. (2007) reported that 24% of the cases stop receiving breast milk at the age less than 4-6 months [47]. Robertson et al. (2002) reported that 23.5% of the mothers gave medicines to stop diarrhea. He also reported that significant difference (p<0.05) was found between four different levels of water consumed by the child during diarrhea [48]. Augistina et al. (2013) conducted a study in East Jakarta in diarrheal patients and reported that 8% of the mothers boil feeder before feeding their child [41]. Bonig et al. (2010) conducted a study in Germany and reported that 21.4% of the gastroenteritis children were breastfeed till 6 months of age and results were almost similar to the present. No home treatment was given by 39.3%, homemade fluids were used by only 32.5% and 27.5% of the respondents were given medicine, and 56.2% of the children with gastroenteritis were taken to doctor [49]. Hafeez et al. (2014) reported that children buying food from vendor (rahri) were at high risk to the disease [4]. Nguyen et al. (2006) reported that 22.3% of the cases were breastfed less than six months of age and 36.4% in controls and significant difference of (p=<0.01) was found between both groups. He also reported that children with age 7-12 month who received supplemental food were higher risk of diarrhea with a significant difference of 32% and 22% in cases and control respectively [39].
Table 4. Influence of Mother’s Education on Child Health.
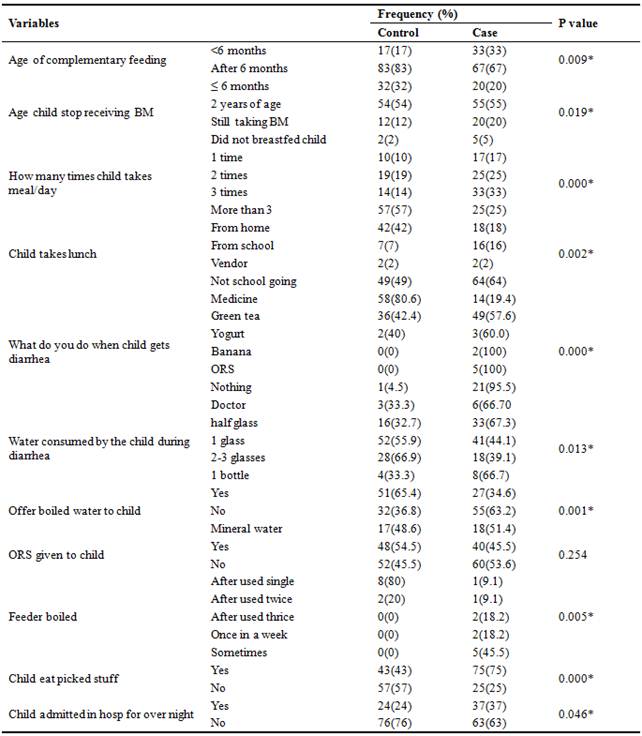
ORS=oral rehydrating salts, BM=breast milk, %=percentage, p=probability, *significant at 0.05.
3.5. Risk Estimation of Socio-economic and Nutritional Status of the Children
Children having WHZ and WAZ ≤ -2 was odd ratio 3.593 and 1.456 respectively. Odd ratio for age of complementary feeding <6 months was 2.405, for children eat picked stuff was 3.977, for children admitted in hospital was 1.860 and for increment in child weight and height was 0.782. Those who had open drain were 0.441 times more exposed to the disease (95% CI: 0.237-0.821). Child who does not wash hand before meal was 0.288 times exposed to disease (95% CI: 0.164-0.677). Child who does not wash hand after attending washing were 0.288 times more exposed to the disease (95% CI:0.161-0.517). Those who do not use soap for washing hands were 0.392 times exposed to the disease (95% CI: 0.221-0.692). More odds ratio shows more probability of disease from the above results it indicated that less WHZ, inadequate complementary feeding, pica and staying in hospitals for long time are the risk factors of gastroenteritis. My study findings are inconsistent with the previous studies as Augistina et al. (2013) conducted a study in East Jakarta and reported that OR for the association of socio economic status and gastroenteritis was 1.58 (95% CI: 0.70-3.56), OR in the association between anthropometric status and gastroenteritis showed that for wasting was 2.3, underweight was 1.67 and stunted was 0.68. OR for children living in joint families were 2.27, for bottle hygiene and gastroenteritis was 0.56 (95% CI: 0.11-2.78). The OR in the association between hand hygiene and gastroenteritis, child’s washing hand before eating meal and after attending washroom was 1.02 (95% CI: 0.41-1.95) and 1.00 (95% CI: 0.36-2.77) respectively whereas for using soap after attending washroom was 0.89 (95% CI: 0.41-1.95) [41]. Das et al. (2013) conducted a study in urban Bangladesh and reported that OR for hospital stay in last 24 hours was 1.66 (95% CI-1.05 - 2.62) [50]. Molbak et al, (1997) reported that OR for no of meals per day was 0.80 (95% CI; 0.71-0.91) and OR children living in nuclear family structure was 0.949 (95 % CI 0.923–0.975) [51]. Nguyen et al. (2006) reported that OR for supplemental food started at age 7-12 months were 2.69 (95% CI: 1.42-5.09) [39].
Table 5. Risk Estimation of Socio-Economic and Nutritional Status of the Children.
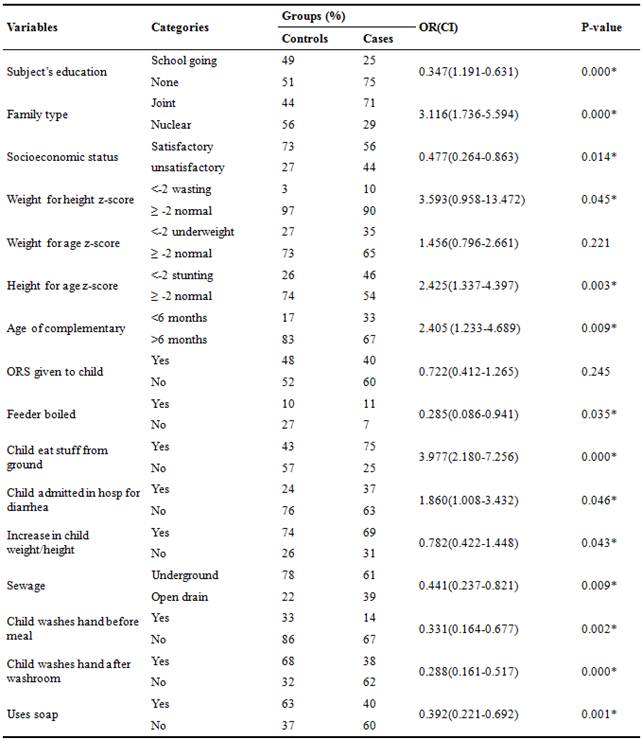
%=percentage, *significant at p=0.05, p=probability, OR=odds ratio, CI=confidence interval, ORS=oral rehydrating salts
4. Conclusion and Recommendations
Low socio economic status, poor hygiene and sanitation practices, malnutrition and low parental education leads to gastroenteritis (GE). Mother’s improper health care practices and knowledge regarding GE can increase the diarrheal complications. Good hygiene and better sanitation conditions can reduce the spread of disease. Mother’s care imparting behavior to the child during GE and knowledge about disease and management can reduce the severity of the disease.
References
- Nancy, S.G. (2013). Acute gastroenteritis. Primary care: clinics in office practice, 40 (3), 727–741.
- Jones, S.A. (2003). Clinical pathway for pediatric gastroenteritis. Gastroenterology Nursing, 26, 7-18.
- Zmir, D., Weiler, Z., Kogan, E., Ben-Valid, E., Hay, E., Reitblat, E. and Polychuck, I. (2006). Single-dose quinolone treatment in acute gastroenteritis. J. Cli. Gastroenterl, 40, 186-190.
- Hafeez, U., Pervaiz, M.K. and Nouman, S. 2014. Determination of risk factors of gastroenteritis through statistical technique in Punjab, Pakistan. Global. J. Agri and Food Sciences Research, 1 (1), 38-44.
- Webb, A. and Starr, M. (2005). Acute gastroenteritis in children. National Center for Biotechnology Information.U.S. National Library of Medicine,34 (4), 227-231.
- Kawai, K., O'Brien, M.A., Goveia, M.G., Mast, T.C. and Khoury, A.C.E. (2012). Burden of rotavirus gastroenteritis and distribution of rotavirus strains in Asia. National institute of health. U.S. National Library of Medicine, 30 (7), 1244-1254.
- Golding, J., Emmet, P.M. and Rogers, I.S. (1997). Gastroenteritis, diarrhea and breast feeding. Hum. Dev, 49, 83–103. www.ncbi.nlm.nih.gov/pubmed/9363419.
- Bhutta, Z.A., Bird, S.M., Black, R.E., Brown, K.H., Gardner, J.M., Hidayat, A. (2000). Therapeutic effects of oral zinc in acute and persistent diarrhea in children in developing countries: pooled analysis of randomized controlled trials. Am. J. Clinic. Nutr, 72 (6), 1516–1522.
- Al-Sonboli, N.G.R. and Shenkin, A. (2003). Zinc supplementation in Brazilian children with acute diarrhoea. Ann. Trop. Paed, 23, 3–8.
- Farthing, M.J.G. (2000). Symposium on the treatment of diarrhoeal disease Diarrhoea: a significant worldwide problem. Intl. J. Antimicrobial Agents, 14, 65–69.
- Karsten, C., Baumgarte, S., Friedrich, A.W., VonEiff, C., Becker, K., Wosniok, W., Ammon, A., Bockemühl, J., Karch, H. and Huppertz, H.I. (2009). Incidence and risk factors for community-acquired acute gastroenteritis in north-west Germany in 2004. Eur. J. Clin. Microbiol. Infect. Dis,28 (8), 935-43.
- Revelas, A. (2012). Acute gastroenteritis among children in the developing world. Southern African Journal of Epidemiology and Infection, 27 (4), 156-162.
- Bhan, M.K. (2000). Symposium on the treatment of diarrhoeal disease current and future management of childhood diarrhea. Intl. J. of Antimicrobial Agents,14, 71–73.
- Fischer, W.C., Perin, J., Aryee, M., Boschi-Pinto, C. and Black, R. (2012). Diarrhea incidence in low- and middle-income countries in 1990 and 2010: a systematic review. BMC Public Health, 12 (1), 220.
- Boschi-Pinto, C., Lanata, C.F. and Black, R.E. (2009). The global burden of childhood Diarrhoea’. In maternal and child health: global challenges, programs, and policies. Maternal and child health. 225-243. springer.com/chapter/10.1007/b106524_13.
- Guerrant, R.L., Schorling, J.B., McAuliffe, J.F. and de Souza, M.A. (1992). Diarrhea as a cause and an effect of malnutrition diarrhea prevents catch-up growth and malnutrition increases diarrhea frequency and duration. Am. J.Trop. Med. Hyg,47 (1), 28–35.
- Hatt, L.E. and Waters, H.R. (2006). Determinants of child morbidity in Latin America: a pooled analysis of interactions between parental education and economic status. Soc. Sci.Med,62 (2), 375–386.
- Boadi, K. and Kuitunen, M. (2005). Childhood diarrheal morbidity in the accrametropolitan area, Ghana: socio-economic, environmental and behavioral risk determinants. J. World Health Popul, 2–13.
- Makoni, F.S., Ndamba, J., Mbati, P.A. and Manase, G. (2004). Impact of waste disposal on health of a poor urban community in Zimbambwe. East. Afr. Med. J, 81 (8), 422–426.
- WHO. (2013). Child growth standards and the identification of severe acute malnutrition in infants and children. Accessed fromhttp://www.who.int/nutrition/publications/severemalnutrition/9789241598163_eng.
- Rahman, M.M., Rahaman, M., Wojtyniak, B. and Aziz, K.M. (1985). Impact of environmental sanitation and crowding on infant mortality in rural Bangladesh. Lancet, 2 (8445), 28–31.
- Lanata, C.F. and Black, R.E. (2008). Diarrheal diseases. In Nutrition and health in developing countries. Nutr. J. Humana Press, 139–178.
- Dennhey., H.P., Mararet, C.M., Rodolfo, B.E., Jenifer, J.L., Robert, E.S., Zhang, N., Rhodes, R., Gentsch, P., Bernstein, W.R., David, I., Brese, C., Staat, S.J. and Allen, M. (2006). A case-control study to determine risk factors for hospitalization for rotavirus gastroenteritis in U.S. children. Pead. Infect. Dis. J, 25 (12), 1123-1131.
- Ethelberg, S., Bente, O., Schiellerup, J., Helms, P., Jensen, M., Bottiger, C., Olsen, B., Katharina, E.P., Flemming, S., Gerner-Smidt, P. and Kare, M. (2006). Risk factors for gastroenteritis among children in an industrialized country. Paed. Infect. Dis. J, 17 (1), 24-30.
- LeBaron, C.W., Furutan, N.P., Lew, J.F., Allen, J.R., Gouvea, V. and Moe, C. (1990). Viral agents of gastroenteritis. Public health importance and outbreak management. Morbidity Mortality Weekly Report. Recommendations and Reports. 39 (RR-5), 1–24.
- Curtis, V., Cairncross, S. and Yonli, R. (2000). Domestic hygiene and diarrhoea pinpointing the problem. Tropical Medicine and International Health, 5 (1), 22–32.
- Black, R.E., Dykes, A.C., Anderson, K.E., Wells, J.G., Sinclair, S.P. and Gary, G.W. (1981). Handwashing to prevent diarrhea in day-care centers. Am. J. Epidemiol, 113 (4), 445–451.
- Dennehy., H.P. (2000). Transmission of rotavirus and other enteric pathogens in the home. Pead. Infect. Dis. J, 10, 103-105.
- Esrey, S.A. and Feachmen, R.G. (1989). Interventions for control of diarrheal diseases among young children: promotion of food hygiene [WHO/CDD/ 89.30]. Geneva: World Health Organization.
- Motarjemi, Y., Kaferstein, F., Moy, G. and Quevado, F. (1993). Contaminated weaning food: a major risk factor for diarrhea and associated malnutrition. Bulletin of the World Health Organization, 71 (1), 79–92.
- Guilbert, J.J. (2003). The world health report 2002 reducing risks, promoting healthy life. Educ. Health. Abingdon, 16 (2), 230.
- Wittenberg, D.F. (2012). Management guidelines for acute infective diarrhoea/gastroenteritis in infants. South African Medical Journal, 102 (2), 104-107.
- McGee, S. (2009). Diarrhoea in Children. Professional Nursing Today, 13 (1), 34-36.
- Zolaly, M.A. and Hanafi, M.I. (2011). Factors affecting antibiotics prescription in general pediatric clinics. J. T. U. Med. Sc, 6 (1), 33-41.
- Rheingans, R., Kukla, M., Faruque, A.S.G., Sur, D., Zaidi, K.M.A. and Nasrin, D. (2009). Determinants of Household Costs Associated With Childhood Diarrhea in 3 South Asian Settings. Eur.J.Clin. Microbiol. Infect. Dis,28, 935–943.
- Bhandari, N., Bhan, M.K., Sazawal, S., Clemens, J.D., Bhatnagar, S. and Khoshoo, V. (1989). Association of antecedent malnutrition with persistent diarrhoea: a case-control study. BMJ, 989 (298), 1284-1287.
- Mansour, A.M., Mohmmad, H.E., Shabraw, M.E., Shabaan, S.Y., Zekri, M.A., Nassar, M., Salem, M. E., Mostafa, M., Riddle, M.S., Kiena, J.D., Messih. I.A.A. and Young, S.Y.N. (2013). Modifiable diarrhea in Egyptian children. Epidemiol. Infect, 141, 2547-2559.
- Kurugol, Z., Geylani, S., Karaca, Y., Umay, F., Erensoy, S., Vardar, F., Bak, M., Yaprak, I. and Ozkynayl, F. (2003). Rotavirus gastroenteritis among children under five years of age in Ýzmir, Turkey. Turk. J. Peads, 45, 290-294.
- Nguyen, V.T., Van, P.L., Huy, C.L., Gia, K.N. and Weintraub, A. (2006). Etiology and epidemiology of diarrhea in children in Hanoi, Vietnam. Intl. J. Infect. Dis, 10, 298-330.
- Ahmad, A., Hafiz, S. and Zafar, A. (1997). Isolation of Aeromonas spp. From human stools. J. Pak. Med. Assoc, 47, 305-308.
- Agustina, R., Sari, T.P., Satroamidjojo, S. and Bovee-Oudenhoven, I.M.J. (2013). Association of food-hygiene practices and diarrhea prevalence among Indonesian young children from low socioeconomic urban areas. BMC. 13, 977.
- Khattak, A.M., Gul, S., Muntaha, S.T. and Jamaluddin. (2007). Weaning evaluation of nutritional knowledge of mothers about their children. Gomal. J. Med. Sc, 5 (1), 17-21.
- Mwangome, M.K., Fegan, G., Prentice, A.M. and Berkley, J. A. (2011). Are diagnostic criteria for acute malnutrition affected by hydration status in hospitalized children.? A repeated measures study. Nutr. J, 10:92. http://www.nutritionj.com.
- Nitema, L.W., Nordgern, J., Quermi, D., Dianou, D., Alfred, S., Svensson, T.L. and Simpore, J. (2011). Burden of rotavirus and other enter pathogens among children with diarrhea in Burkina Faso. Intl. J. Infect. Dis, e646-e652.
- Opintan, J.A., Newan, M.J., Ayeh-Kumi, P.F., Affrim, R., Gepi-Attee, R., Sevilleja, J.E.A.D., Roche, J.K., Roche, J.K., Nataro, J.P., Warren, C.A. and Guerrant, R.L. (2010). Pediatric Diarrhea in Southern Ghana: Etiology and Association with Intestinal Inflammation and Malnutrition. Am. J. Trop. Med. Hyg, 83 (4), 936–943.
- VanDersclice, J., Popkin, B. and Briscoe, J. (1994).Drinking-water quality, sanitation, and breastfeeding: their interactive effects on infant health. Bull World Health Organ, 72(4): 589-601.
- Quigley, M.A., Kelly, Y.J. and Sacker, A. (2007). Breastfeeding and hospitalization for diarrheal and respiratory infection in the United Kingdom millennium cohort study. J. Pead, 119 (4), e837-842.
- Roberston, B., Sinclair, M.I., Forbes, A.B., Veitch, M., Kikr, M., Cunlifee, D., Wills, J. and Fairley, C.K. (2002). Animal contact and water consumption glasses Case-control studies of sporadic cryptosporidiosis in Melbourne and Adelaide, Australia. Epidemiol. Infect. J, 128, 419-431.
- Plenge-Bonig, A., Soto-Ramirez, N., Karmaus, W., Petersen, G., Davis, S. and Forster, J. (2010). Breastfeeding protects against acute gastroenteritis due to rotavirus in infants. Eur J Pediatr, 169 (12), 1471-1476.
- Das, S.K., Chisti, M.J., Malek, M.A., Ahmed, S., Farzana, F.D., Ferdous, F., Shahunja, K.M., Salam, M.A., Ahmed, T. and Faruque, A.S.G. (2013). Socio-demographic, host and clinical characteristics of people with typhoidal and non-typhiodal Salmonella gastroenteritis in urban Bangladesh. J. Biomedical Science and Engineering, 6, 834-842.
- Molbak, K., Jensen, H., Ingholt, L. and Aabyl, P. (1997). Risk Factors for Diarrheal Disease Incidence in Early Childhood: A Community Cohort Study from Guinea-Bissau. Am. J. Epidemiol, 146 (3), 273-283.



