Antioxidant Activity, Total Phenol Content, and Bioactive Components of Lindur Leave (Bruguierra gymnorrhiza)
Nurjanah Nurjanah, Agoes M. Jacoeb, Taufik Hidayat*, Siti Hazar,
Roni Nugraha
Department of Aquatic Product Technology, Faculty of Fisheries and Marine Science Bogor, Agricultural University Agatis Street Darmaga Bogor, West Java, Indonesia
Abstract
Lindur are mangrove plants that has been widely used as drug, yet informations on potential drug compounds and antioxidants contained in lindur were still limited. This study were determined to determine the chemical composition, bioactive components, antioxidant activity, and total phenol contained in lindur. The result showed that lindur fresh leaves had a moisture content, ash, fat, protein, and crude fiber of 54.91%, 4.49%, 1.87%, 4.45%, and 8.67%; respectively. Lindur dried leaves had water, ash, fat, protein, and crude fiber content of 9.79%, 8.19%, 3.90%, 9.78%; and 18.05%, respectively. The methanol extract had the highest yield (15,79%) compared to the ethyl acetate extract (2.85%), and n-hexane (1.53%). Phytochemical analysis showed the bioactive components of n-hexane extract (phenol steroids and hydroquinone); ethyl acetate extract (steroids, hydroquinone phenols, flavonoids, saponins) and methanol extract (steroids, hydroquinone phenols, flavonoids, tannins, saponins). Methanol extract showed the highest antioxidant activity with IC50 values of 81.11 ppm, while the n-hexane extract showed the lowest activity with IC50 values of 815.67 ppm. Total phenolic extracts of n-hexane, ethyl acetate, and methanol, were 12.41, 97.57, and 30.07 mg GAE/ g extract, respectively.
Keywords
Antioxidant, Bioactive, Bruguiera gymnorrhiza, Leave, Phenol
Received: June 6, 2016
Accepted: June 21, 2016
Published online: July 16, 2016
@ 2016 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY license. http://creativecommons.org/licenses/by/4.0/
1. Introduction
Modern society today tend to have a fast-paced lifestyle and instant, included in the diets. Langseth (1995) states that the improper diet will lead to long-term accumulation of free radicals in the body. Free radicals can also be caused by the metabolism of the body and outside the body, such as cigarette smoke, environmental pollution, radiation, drugs, pesticides, as well as ultraviolet radiation. A free radical is a compound or molecule having one or more unpaired electrons in its outer orbital. The presence of unpaired electrons cause the highly reactive compound looking for a partner by attacking and binding electrons in the molecules in the surrounding areas (Winarsi 2007). Macromolecules are attacked can have conditions that cause oxidative damage to proteins, DNA, premature aging, cancer, heart attack (Middleton et al. 2000), cataracts, and skin diseases, and other degenerative diseases (Reyneston 2007). Damage caused by free radicals in the body can be treated with antioxidants.
Antioxidants are chemical compounds that can donate electrons to free radicals they contain to inhibit or prevent oxidation of the substrate is easily oxidized (Middleton et al. 2000). The human body naturally produces endogenous antioxidant that is able to overcome the effects of free radicals, but when the supply of free radicals increased supply of antioxidants needed from outside (exogenous). Exogenous antioxidants can be obtained in the form of synthetic (man made) or natural. Synthetic antioxidants for example benzoic acid, BHA (Butylated Hydroxy anisole), BHT (Butylated Hydroxy Toluene) and TBHQ (Tertiary Butylated Hydroxy Quinones) can cause side effects on health because it is carcinogenic. Results wichi research (1988) shows the BHA and BHT can cause tumors and liver damage in long-term use. Concerns about adverse effects that result in natural antioxidant potential to be developed. Natural antioxidants can be obtained from fruits or herbs. The herbs contain secondary metabolites such as flavonoids and phenolics that can catch free radicals (Duenas et al. 2009). One of the plants with the potential to be developed as a source of natural antioxidants derived from mangrove ecosystems.
Mangrove is a plant that thrives in coastal areas that have the potential of bioactive compounds is very high. Indonesian territorial waters with very broad (2/3 of the total area) and tropical climate is the ideal place for the growth of mangroves. Indonesia is a country that has the largest mangrove forest in the world, with an area of mangrove forests around 3.5 million hectares (Noor et al. 2006). There are 202 species of mangrove species that have been identified in Indonesia. Mangrove plants have been screened their activities, as antiviral, antibacterial, antibisul, and anti-inflammatory (Agoramoorthy et al., 2008; Premanathan et al. 1999). One alternative source of natural antioxidants is a leaf lindur (Bruguiera gymnorrhiza).
Lindur plant in Indonesia spread in Java, Sumatra, Kalimantan, Maluku and Bali (Duke and James 2006). Fruit lindur Tual widely used by people in the district of Sulawesi as a source of carbohydrate substitute for rice when the famine as well as drugs to treat eye diseases and herpes (Haq et al., 2011). Bark is used to treat diarrhea and malaria in the Solomon Islands (Allen and Duke in 2006), while its leaves are used to treat burns (Haq et al., 2011). Lindur leaves suspected to contain bioactive components that are very useful for the body and potentially used as a natural source of antioxidants because it has been used as a traditional medicine in the community, but still not enough information to explain these things scientifically. The purpose of this study was to determine the bioactive compounds, antioxidant activity and total phenolic extract coarse leaf lindur.
2. Material and Method
2.1. Materials and Tools
The main material used in this study is the leaves lindur (B. gymnorrhiza) derived from the old Mangrove Conservation Area in Pantai Indah Kapuk, North Jakarta (Figure 1). Other materials were used that material for proximate analysis, material for phytochemical test, test ingredients for antioxidant activity by DPPH method, and materials to test the levels of total phenols. Material for analysis of antioxidant activity that is crystalline 1,1-diphenyl-2-pycrlhydrazil (DPPH), ethanol, and vitamin C. Material for analysis of total phenol that is 95% ethanol, distilled water, reagent Folin Ciocelteau 50%, 5% Na2CO3, and a solution standard (gallic acid).

Figure 1. Lindur leaf (Brugueiera gymnorrhiza).
The tools used in the study include tools for preparation, container, digital scales, aluminum foil, blender, Whatman filter paper 42, electric stoves, furnaces, incineration, pumpkin Kjeldahl, tube soxhlet, spectrophotometer (Spectro UV-Vis 2500), desiccator, vortex, pipettes, and other tools eg glass test tubes, glass beakers, centrifuges, glass bottles, glass funnel, flask, and Erlenmeyer flasks.
2.2. Research Methods
This research was conducted in several stages of sampling; sample preparation; Proximate analysis that refer to SNI 01-2891-1992 (BSN 1992); multilevel extraction (Darusman et al. 1995); Phytochemical analysis (Harborne 1987); analysis of antioxidant activity (Aranda et al. 2009), and analysis of total phenols (modification Ukieyanna 2012).
3. Results and Discussion
3.1. Chemical Composition
Table 1. Chemical compostion Lindur leaf.
| Component (b/b) | Bruguiera gymnorrhiza | |
| fresh | wet | |
| Moisture | 54.91 | 979 |
| Ash | 4.49 | 8.19 |
| Fat | 1.87 | 3.90 |
| Protein | 4.45 | 9.78 |
| Dietary fiber | 8.76 | 18.05 |
Lindur leaf nutrient content was determined by proximate analysis. Proximate analysis is an analysis conducted to predict the chemical composition of a substance, including water content, fat, protein, ash, and crude fiber. The sample used for proximate analysis is lindur fresh and dried leaves. The chemical composition of fresh and dried leaves lindur presented in Table 1.
3.2. Moisture Content
Lindur leaf water content decreased by 45.12% after the drying process. The decline in water levels caused by the drying process evaporate most of the water on the material. According Kusnandar (2010) that changes in water levels can be caused by water easily evaporates when subjected to the heating process. Heat transfer and movement of water and air flow causes evaporation and drying process on groceries.
3.3. Ash Content
Lindur dried leaves showed an increase by 3.7% ash content compared to fresh lindur leaves. Wibowo et al. (2009) states that the level of ash content can be caused by differences in habitat or the environment is different. Each organism also has a different ability to regulate and absorb minerals so that it will also affect the ash content value of each ingredient.
3.4. Fat Content
Lindur dried leaves unchanged the value of the fat content of 2,03% compared with leaves of fresh lindur. Low fat content can be caused by water content is high enough so that the fat content proportionally decreased. According Yunizal et al. (1998), the water content is generally inversely related to fat content. That relationship led to increasingly low levels of fat if the water content contained in the materials have high numbers.
3.5. Protein Content
Lindur leaf protein levels change proportionally from 4.45% to 8.19% after drying. Lindur fresh leaves contain protein in relatively small amounts, but tend to be higher than the protein content of other fresh mangrove leaves. Vegetable food materials generally have lower protein than animal food (Muchtadi and Fitriyono 2010).
3.6. Crude Fiber Content
Crude fiber content in the leaves dry lindur higher by 9.29% compared lindur fresh leaves. Muchtadi (1993) stated that the fiber content in foods can be altered by processing conducted on the original material. The fiber content in the plants will generally experience increased due to the heat treatment. Overall, the leaves have dried lindur ash, fat, protein and crude fiber is higher than in the leaves of fresh lindur. This is due to the drying process on the dried leaf samples lindur According Muchtadi and Fitriyono (2010), with the drying process reduces the moisture content and result in materials containing compounds such as proteins, carbohydrates, and minerals in higher concentrations.
3.7. The yield Extract
Lindur leaf extract is done in stages with the type of solvent that has a different polarity. Lindur leaf extraction performed by maceration for 48 hours until the resulting filtrate translucent color. The extract obtained in the form of a thick paste with different colors depending on the type of solvent used. N-hexane extract greenish yellow color, while the ethyl acetate extract and methanol respectively green and red-brown. The color change that occurs in the n-hexane extract in accordance with the results of Ismail et al. (2012), which showed a clear color change to yellow, it indicates nonpolar component extracted by solvents which allegedly is a carotenoid pigment.
The green color of the ethyl acetate extract showed the pigment chlorophyll. Chlorophyll is generally bind to the protein and fat can be extracted with a solvent which has a low polarity level (Harborne 1987). Red-brown discoloration on the methanol extract is also consistent with the results of Ismail et al. (2012) believed to be the anthraquinone pigment. Estraksi with different types of solvent extract also produce different yields. The yield of leaf extract lindur with each solvent are presented in Figure 2.
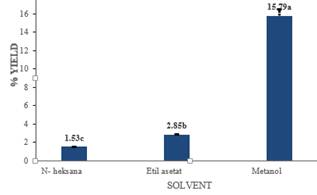
Figure 2. Proportion crude extract leaf lindur.
The yield on the smallest of n-hexane extract of 1.53%, while the yield of the total of the methanol extract of 15.79%. This is in line with the results of research Salama et al. (2008) which showed that the extract yield results different maceration using a greater percentage yield different.
3.8. Bioactive Components Extract
Bioactive components in extracts of leaves lindur determined through analysis of phytochemicals. Phytochemical analysis was conducted on the alkaloids, steroids, flavonoids, phenols hydroquinone, tannins and saponins. Phytochemical analysis results are presented in Table 2.
Table 2. Component bioaktif crude extract leaf lindur.
| Component boactive | Result | ||
| Extract n-heksane | Extract etil asetat | Extract methanol | |
| Alkaloid | |||
| - Wagner | - | - | - |
| - Dragendorff | - | - | - |
| - Meyer | - | - | - |
| Steroid | + | + | + |
| Flavonoid | - | + | + |
| Phenol hidroquinon | + | + | + |
| Saponin | - | + | + |
| Tanin | - | - | + |
Phytochemical analysis results showed positive results in the third extract their steroid. Steroids found on the third leaf extract lindur allegedly functions as a stamina enhancer. The assumption is reinforced on the antioxidant activity of the extract n-hexane weak (> 200 ppm). Third lindur leaf extract also showed the presence of phenol hydroquinone. Phenol compounds tend to be readily soluble in water and non-polar compounds as generally binds to sugars as glycosides and is usually found in the cell vacuole (Harborne 1987).
Flavonoid compounds found only in the methanol extract and ethyl acetate extract. This is because it is influenced by a polar solvent in extracting flavonoids. According to Middleton et al. (2000), flavonoids are the active compounds are included in this type of intermediates of antioxidants, which act as hydrophilic and lipophilic antioxidants. Phytochemical analysis results showed the methanol extract tannin. According to Harborne (1987), tannins can react with proteins to form copolymers strong and mostly insoluble, except terkondenasi tannins that can be extracted with methanol. Phytochemical analysis results also indicate the presence of saponins in ethyl acetate and methanol extracts. Saponins are surface active compounds and are like soap. Saponins can be detected by their ability to form foam and menghemolisis blood cells (Harborne 1987). Results of research Homhual et al. (2006) showed that the bioactive components contained in B. gymnorrhiza consisting of phenol compounds, flavonoids, steroids, sulfur content, and components terpenoids.
3.9. Antioxidant Activity Crude Extract
The antioxidant activity of leaf extract lindur determined by DPPH is expressed in percentage inhibisinya against free radical DPPH (Ukieyanna 2012). Measurement of antioxidant activity of the extract n-hexane, ethyl acetate extract and methanol extract. The average IC50 value of each extract is presented in Figure 3.
Methanol and ethyl acetate extract has antioxidant activity in the range of test concentrations (75-175 ppm). The methanol extract had strong antioxidant activity with IC50 value of 81.11 ppm, while the ethyl acetate has a moderate antioxidant activity with IC50 value of 114.18%. The high value of IC50 in the extract of n-hexane and ethyl acetate showed weak antioxidant activity of the extract compared with the methanol extract. This is because the solvent n-hexane and ethyl acetate has a polarity lower than methanol extract. Something similar is evidenced by the results of research Ukieyanna (2012) which showed that the antioxidant activity of ethanol extract lower may be caused by the presence of nonpolar compounds extracted is not a powerful antioxidant compounds, such as essential oils, fats, and oils. Another factor causing the high antioxidant activity of the methanol extract that is because many bioactive compounds extracted, for example tannins including polyphenolic compounds. The third extract is still relatively crude extract so there is still a compound suspected bullies, including and other compounds that block the capture of free radicals. The purity of a sample when the extraction process affects the antioxidant activity of the sample.
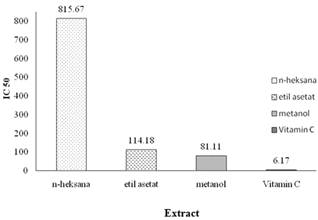
Figure 3. Antioxidant activty crude extract lindur leaf.
The overall results show that the difference in the solvent in the extraction process affect the antioxidant activity of the extract. Values obtained antioxidant activity is still very low compared to the results of research Haq et al. (2011) with IC50 values of 0.038 ppm ethanol extract and extract of methanol and chloroform respectively 0,027 ppm and 0.28 ppm. The low IC50 values obtained in this study may be due to the extraction time is too long.
3.10. Total Phenol Crude Extract
Table 3. Total phenolic crude extract leaf lindur.
| Ekstrak | Total phenolic (mg GAE/g extract) | ||
| N-heksane | 30.07 | 134.16 | 178.73% |
| Etil asetat | 97.57 | - | - |
| Metanol | 12.41 | - | - |
Determination of total phenolic content can be done using the Folin-Ciocalteau principle is based on the oxidation-reduction reaction. The content of total phenols expressed in GAE (gallic acid equivalent). Total phenol content of each extract are presented in Table 3.
The highest total phenolic compounds contained in ethyl acetate extract is equal to 97.57%. It shows the amount of phenolic compounds in the extracts of leaves that are semi-polar lindur. The amount of total phenolic compounds in the ethyl acetate extract does not correspond to its antioxidant activity, as well as the methanol extract. The methanol extract had strong antioxidant activity (50-100 ppm), but the total compounds phenoliknya less than the ethyl acetate extract. This is due to the methanol extract has antioxidant activity in addition to phenolic compounds (Ismail et al. 2012). Other possible causes for the antioxidant activity of the ethyl acetate extract is lower than the methanol extract impurities due to their compounds chlorophyll. This is consistent with the results of research Ukieyanna (2012), which indicates a low antioxidant activity in the ethyl acetate extract of the leaves errand.
Results of research Haq et al. (2011) demonstrated the value of total phenol lindur leaf ethanol extract of 189.4 mg / g, while the methanol extract of 178.73 mg GAE / g of extract (Haq et al., 2011). Total phenolic content of the methanol extract is higher than the total phenolic methanol extract of research results. In general, in each extract has a total phenolic different. According to Harborne (1987), differences in the level of polar solvent determine the chemical structure of phenolic compounds extracted. Testing of total phenolic highly dependent on their chemical structure. Phenolic compounds having hydroxyl functional groups that is a lot or in the free state (aglycone) will produce a high total phenolic content. A polar solvent capable of attracting phenolic compounds in considerable amounts. Polar phenolic compounds have a high antioxidant activity. The high total phenolic in ethyl acetate extract compared with methanol for ethyl acetate extracts are semi-polar to extract the phenolic compounds are polar and non-polar. According Andayani et al. (2008), the solvent ethyl acetate extract phenolic compounds, either polar or nonpolar. Results of research Haq et al. (2011) also showed that the ethanol extract had the highest total phenolic extract than methanol and chloroform.
Kubota et al. (2000) suggest that differences in levels of total phenolics in plants caused by nutrients, habitats and different varieties. Folin-Ciocalteu reagent reacted with phenolic compounds will cause the color changes from yellow to blue green.
4. Conclusion
Lindur leaf extracts of fresh and dried percentage of different chemical composition. The results of the highest yield in the methanol extract, followed by the yield of the ethyl acetate extract, and n-hexane. Bioactive components in the extract n-hexane, ethyl acetate, and methanol are flavonoids, phenols hydroquinone, and steroids. The methanol extract had strong antioxidant activity, while the antioxidant activity of n-hexane extract is very weak (> 200 ppm). The total value of the highest phenolic on ekstrat ethyl acetate, followed by methanol extract and extract n-hexane.
References
- Allen JA, Duke NC. 2006. Bruguiera gymnorrhiza (large-leaf mangrove). www.traditionaltree.com [9 Juli 2013].
- Agoramoorthy G, Chen F, Venkatesalu V, Kuo DH, Shea, PC. 2008. Evaluation of antioxidant polyphenols from selected mangrove plants of India. Asian Journal of Chemistry 20(2):1311-1322.
- Andayani R, Y Lisawati, Maimunah. 2008. Penentuan aktivitas antioksidan, kadar phenol total, dan likopen pada buah tomat (Solanum lycupersicum L). Jurnal Sains dan Teknologi Farmasi 13(1): 1-9.
- Aranda SR, Perez-Lopez LA, Arroyo JL, Alanis-Garza BA, de Torres NW. 2009. Antimicrobial and antioxidant activities of plants from Northeast of Mexico. Journal of Evidence-Based Complementary and Alternative Medicine 41(5):233-236.
- Darusman LK, Sajuthi D, Sutriah K, Pamungkas D. 1995. Ekstraksi komponen bioaktif sebagai bahan obat dari karang-karangan, bunga karang dan ganggang di perairan Pulau Pari Kepulauan Seribu [laporan penelitian]. Bogor: Fakultas Matematika dan Ilmu Pengetahuan Alam, Institut Pertanian Bogor.
- Duenas M, Manzano SO, Paramas AG, Buelga SC. 2009, Antioxidant evaluation of O-methylated metabolites of catechins, epicatechin, and quersetin. Journal of Pharmaceutical and Biomedical Analysis.
- Duke NC, James AA. 2006. Bruguiera gymnorrhiza (large-leafed mangrove). Species Profiles for Pacific Island Agroforestry Apr; Ver 2.I. www.traditionaltree.org [10 Oktober 2013].
- Haq M, Wirakarnain S, ABMS Hossain, Rosrna Mat Taha, KM Monneruzzaman. 2011. Total phenolic contents, antioxidant and antimicrobial activities of Bruguiera gymnorrhiza 6(17): 4112-4118.
- Harborne JB. 1987. Metode Fitokimia. Edisi ke-2. Padmawinata K, Soediro I, penerjemah. Bandung: Institut Teknologi Bandung. Terjemahan dari: Phytochemical Methods.
- Homhual S, Bunyapraphatsara N, Kondratyuk T, Herunsalee A, Chaukul W, Puzzuto JM, Fong HHS, Zhang HJ. 2006. Bioactive dammarane triterpenes from the mangrove plant Bruguiera gymnorrhiza. Journal of Natural Product 69 (3):421-424.
- Ismail J, Max RJM, Feti F. 2012. Penentuan total phenolik dan uji aktivitas antioksidan pada biji dan kulit buah pinang yaki (Areca vestiaria Giseke). Jurnal Ilmiah Sains 12(2): 84-88.
- Kusnandar F. 2010. Kimia Pangan. Jakarta (ID): PT Dian Rakyat.
- Langseth L. 1995. Oxidants, Antioxidants and Disease Prevention. Belgium: ILSI Europe.
- Middleton EC, Kandaswami, TC Theoharides. 2000. The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharmacological Reviews 52:673-751.
- Muchtadi D. 1993. Aspek Biokimia dan Gizi dalam Keamanan Pangan. Bogor (ID): Pusat Antar Universitas Pangan dan Gizi, IPB Pr.
- Muchtadi TR dan Fitriyono A. 2010. Teknologi Proses Pengolahan Pangan. Bandung (ID): PT Alfabeta.
- Noor YR, Khazali M, Suryadiputra INN. 2006. Panduan Pengenalan Mangrove di Indonesia. Wetlands International-Indonesia Programme. Bogor: Ditjen PHKA.
- Premanathan M, Arakaki R, Izumi H, Kathiresan K, Nakano M, Yamamoto N, Nakashima H. 1999. Antiviral properties of a mangrove plant, Rhizophora apiculata Blume, against human immunodeficiency virus. Antiviral Res. 44(2):113-22.
- Reynertson KA. 2007. Phytochemical Analysis of Bioactive Constituens from Edible Myrtaceae Fruit. [Dissertation]. The City University of New York.
- Salamah E, Ayuningrat E, Purwaningsih S. 2008. Penapisan awal komponen bioaktif dari kijing taiwan (Anadonta woodiana Lea.) sebagai senyawa antioksidan. Buletin Teknologi Hasil Perikanan 11(2):119-132.
- Ukieyanna E. 2012. Aktivitas antioksidan, kadar phenolik, dan flavonoid tumbuhan suruhan (Peperomia pellucida L. Kunth) [skripsi]. Bogor: Fakultas Matematika dan Ilmu Pengetahuan Alam, Institut Pertanian Bogor.
- Wibowo C, Kusmana C, Suryani A, Hartati Y, Oktadiyani P. 2009. Pemanfaatan pohon mangrove api-api (Avicennia spp.) sebagai bahan pangan dan obat. Prosiding Seminar Hasil-Hasil Penelitian IPB 2009 Buku 1: bidang pangan dan energi. Bogor: LPPM-IPB.
- Wichi HP. 1988. Enhanced tumour development by butylated hydroxytoluene (BHT) from the properties of effect on fure stomach and esophageal aquamoua epithelium. Food Chemical Toxicology 26:723-727.
- Winarsi H. 2007. Antioksidan Alami dan Radikal Bebas. Yogyakarta (ID): Kanisius.
- Yunizal, Murtini JT, Dolaria N, Purdiwoto B, Abdulrokhim, Carkipan. 1998. Prosedur Analisis Kimiawi Ikan dan Produk Olahan Hasil-Hasil Perikanan. Jakarta (ID): Pusat Penelitian dan Pengembangan Perikanan.



