Low-Cost Green Modifiers for Selective Flotation of Au-Sulfide Minerals from Refractory Ores
Matveeva Tamara*, Ivanova Tatiana, Gromova Nadezda
Institute of Complex Exploitation of Mineral Resources, Russian Academy of Sciences (IPKON RAS), Moscow, Russia
Abstract
The paper presents the results of study of adsorption and flotation properties of the reagents of plant origin - tannin and natural extracts - of the oak bark and leaves and stems of Heracleum plants, with a view to their application as low-cost environmentally friendly modifiers of Au-sulfide minerals in flotation of refractory ores containing precious metals. The technique of preparation of oak bark extract with a fixed content of extractable tannins has been developed. The mechanism of tannin adsorption on the surface of pyrrhotite and arsenopyrite was identified as to form stable ferrous (III) complexes at concentrations below micelle formation. The use of plant modifiers as selective depressants for pyrite, pyrrhotite and arsenopyrite could help to increase the quality of Au and Cu concentrates and reduce the ecological impact on the environment.
Keywords
Plant Extracts, Modifier, Adsorption, Flotation
Received: April 5, 2015 / Accepted: April 27, 2015 / Published online: May 19, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Actual application of new reagents and plant extracts as modifiers in flotation of precious metals mineral is the possibility of replacing of highly toxic chemical compounds by environmentally safe substances of plant origin.
In world practice, agents of plant origin (starch, dextrin, quebracho, etc.) are used in flotation of sulfide ores and low-sulfide for depression of oxidized gangue minerals. Tannin is applied as a depressant of calcite and dolomite in the flotation of scheelite and fluorite [10]. Organic polymers such as guar gum and carboxymethyl cellulose, that incorporate also phenol ingredients with large number of OH groups, are widely used in the flotation of Pt-containing ores for depression for talc-containing gangue minerals [8], [11].
However the wide dissemination of these reagents is prevented because of insufficient knowledge of their composition and physico-chemical properties. The main disadvantage of tannin used as depressant in sulfide flotation, is considered to be their non-selectivity [3]. Nevertheless results achieved by Trusov demonstrated the possibility of separating certain pairs of sulfide minerals particularly, chalcopyrite and galena in soda flotation medium [12]. The results of the studies that have been carried out recently in IPKON RAS using as a cheap tannin-bearing oak bark extract reagent (reagent OBE) showed that by changing the pH and the concentration of the reagent OBE, the introduction order of depressant and collector, you could improve the quality of Cu-Ni-Pt concentrate [6] and reached high results in separation of sulfides, especially pyrite and arsenopyrite, to obtain auriferous pyrite concentrate containing less than 2 % of As [2].
The main task of the experiments is to examine adsorption and flotation properties of the reagents obtained from plant raw materials, with the aim to determine their potential application as low-cost green modifiers for selective flotation of gold-host sulfides from refractory ores and improve the quality of flotation concentrates.
2. Methodology
The oak bark extract (OBE) and Heracleum stems and leaves extract (HSE) were tested as the extracts of wildly spread and most available eco-friendly tannin-containing plants. Besides the hydrophilic properties these agents are able to form complex compounds with the metallic ions which may be used for modifying sulfide minerals in selective flotation.
OBE extract was prepared by 3 step-by-step extraction of oak shavings (ground to -3 +1 mm) into water under heating 70-80°C for 120 minutes at the ratio S:L = 1:15 [7]. Flask for extraction was equipped with a reflux condenser, a thermometer and a magnetic stirrer. The first extract was filtered hot. Two subsequent extractions were continued for 60 minutes at 70-80°C. After filtration extracts were combined and diluted by distilled water up to 250 ml volume. The method helped to obtain an extract containing approximately 13 % of extractives, including about 47 % of tannins. The UV spectrum of the extract has a characteristic absorption maximum at 275 nm.
To determine the concentration of tannin in OBE extract a photometric method of analysis was used. The methodology is based on using Folin’s reagent and sodium carbonate for determination of tannin concentration in solution [4]. The absorbance of the sample solution was measured by photocolorimeter at 660 nm in a cuvette L-30 mm.
In order to identify the mechanism of depressive action of OBE on iron sulfides in flotation its effect on the sorption of the collector (xanthate) on the surface of pyrrhotite was studied. 1 g sample of crushed mineral -0.16 +0.063 mm particle size was contacted with xanthate solution (20 mg/l) for 3 minutes then OBE was added and stirred for another 3 minutes. The liquid phase was decanted after centrifugal and the residual concentration of xanthate was analyzed by spectrophotometer SHIMADZU-1700 and tannin concentration was determined by photometric method with Folin’s reagent on UNICO-2100 photocolorimeter. OBE adsorption was calculated (in terms of tannin) after interaction with pyrrhotite.
The chemical composition of pyrite, arsenopyrite, chalcopyrite and pyrrhotite samples is demonstrated in Table 1.
Table 1. The chemical composition of the mineral samples.
| Minerals | Content % | ||||||
| Fe | S | As | Cu | Oxides | Others | Au, ppm | |
| Pyrite | 39.3 | 43.7 | 2.5 | 0.8 | 12 | 1. 7 | 7 |
| Arsenopyrite | 29.7 | 19.3 | 40.5 | 0.04 | 10.4 | <0.1 | 15 |
| Pyrrhotite | 59 | 38.1 | - | 0.04 | - | 2.86 | - |
| Chalcopyrite | 28.1 | 28.3 | - | 26.4 | 15.4 | 1.8 | 1 |
3. Results and Discussion
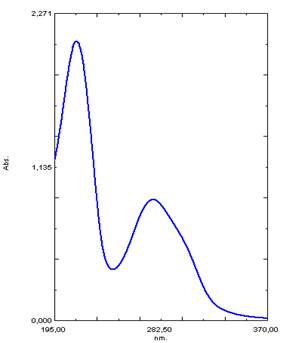
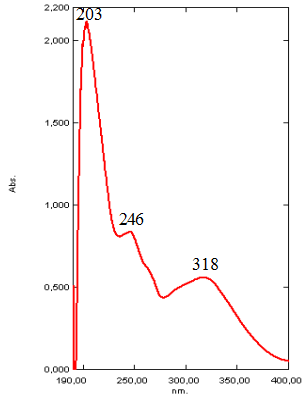
Analyzing the UV spectrum of OBE extract the characteristic absorption maxima at 275 and 200 nm (Figure 1a) were determined, which are very close to peaks in pure tannin UV spectrum at 275 and 212 nm. Similar peaks at 275 nm showed tannin incorporated into OBE extract, and shift of the peaks at 203-212 nm may be explained as the result of the other herbal compounds extracted together with tanning substances under the experimental conditions. Figure 2a shows characteristic absorption maxima at 318, 246 and 203 nm in aqua-alcoholic HSE extract. The absorption peak at 203 nm in HSE extract indicates the presence of substances identical to tannin. The same peak presents in the spectrum of OBE. The absorption peak at 318 nm is closed to the absorption peak of coumarin [9]. Shift of the peaks at 203 and 318 nm in the UV spectrum of the extract relative to the peaks of absorption of pure tannin solutions and coumarin, and a peak at 246 nm may be explained by extraction of other components into HSE extract (amino acids, essential oils, etc.) that composed the plant material.
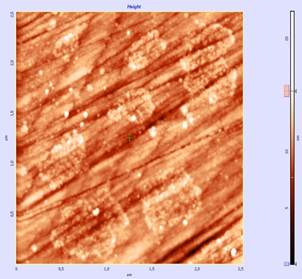
Using scanning atomic force microscope NT-MDT we defined tannin adsorption on the surface of polished section of pyrite for changes under the action of the reagent relief (topography) of the mineral surface and adsorbed on the surface of the mineral phase reactant (phase). As a result of contact of mineral with tannin solution on the mineral surface there was found the appearance of the newly formed phase of reactant, which differs greatly from sulfide phase by its morphology and discrete non-uniform distribution. Images obtained with a scanning atomic force microscope NT-MDT, helps to study the topography (relief) of the surface with a resolution of tens of angstroms up to atomic dimension. Vision changes in relief (topography) of the mineral surface and adsorbed reactant phase (small white points) on the surface of the mineral (phase) under the action of the reagent can be clearly seen on the images on Figure 1b and 2b.
a
b
Figure 1. UV spectrum of OBE extract (a) and the image of phase contrast of pyrite surface (5x5 micron portion) after its contact with tannin solution (Scanning atomic force microscope NT-MDT) (b).
The newly formed phase is localized on the mechanical irregularities of the mineral surface (cracks, edge of grains, ets.), boundaries and accretion of mineral grains. Most of the tumors are characterized by elevated oxygen content, relatively unchanged mineral that indicated the oxide nature of the newly formed compounds. The thickness of the surface layer of tumors depended on the solution concentration of the reagent and duration of its contact with the solution.
Additionally, polished thin section of Berezovsky pyrite after its contact with HSE extract was analyzed by laser scanning microscope KEYENCE VK- 9700. On the images of pyrite surface after its contact with HSE one can see discrete films of newly formed phase that cover the mineral (Figure 3). Contactless measuring of surface irregularities performed by the scanning laser microscope helped to estimate the dimensions of newly formed phase. For example, the height was estimated as 0.3 – 1.2 μm and the diameter – 1-20 μm.
a

b
Figure 2. UV spectrum of aqua-alcoholic HSE extract (a) and the image of phase contrast of pyrite surface (5x5 micron portion) after its contact with HSE extract (Scanning atomic force microscope NT-MDT) (b).
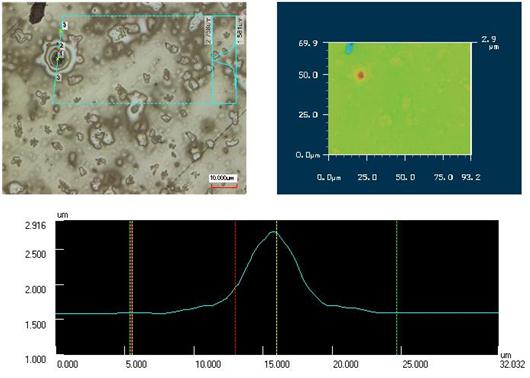
Figure 3. Berezovsky pyrite surface images after its contact with plant HSE extract. (Laser microscope KEYENCE VK-9700. Label 10 microns).
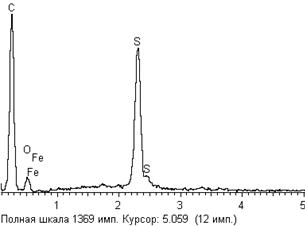
Moreover, scanning electron microscope microphotoes demonstrate the distribution of the extract on the surface of pyrite (Figure 4a). The photo shows that HSE extract adsorbed on the mechanical irregularities of the mineral surface (cracks, accretions and boundaries of mineral grains. In the X-rays spectrum O and C peaks were identified additionally to Fe and S of pyrite (Figure 4b), characterizing the elevated oxygen and carbon content, relatively unchanged mineral surface that indicated the oxide-organic nature of the newly formed compounds. The thickness of the surface layer of the new phase depended on the solution concentration of the reagent and duration of its contact with the solution. It should be mentioned that adsorbed extract components were still reserved on pyrite surface after its cleaning in distilled water. This fact means that extract form very stable covering on the mineral surface.
Since the main component of OBE extract is tannin, in order to analyze the mechanism of depressing action of OBE, tannin complex-forming ability in relation to Fe (II) and Fe (III) salts aqua solutions was additionally studied [7]. Tannin has characteristic absorption bands in the UV spectrum at 275 nm and 212 nm. Colour compounds of tannin complexes with Fe (II) and Fe (III) were obtained and corresponding UV spectra were analyzed. Fe (III) formed green colour complex that has light absorption maximum at 648 nm. Fe (II)-tannin formed purple colour complex in one hour after the start, its light absorption maximum can be seen at 564 nm, and then a blue-black precipitate was formed. In the spectra of the starting materials such peaks were absent. Experimental identification of tannin complex compounds in the Fe (II) and Fe (III) solutions may indirectly confirm the probability of tannin adsorption on the surface of iron sulfide minerals forming insoluble complex compounds. Tannin covering helps to provide hydrophilic properties to mineral surface and cause the depression of Fe-sulfide minerals.

a
b
Figure 4. Microphoto of Berezovsky pyrite polished surface after its contact with HSE extract (a) and X-rays spectrum of newly formed phase of HSE extract on the surface(b). (Scanning electron microscope LEO. Label 100 microns).
Laboratory tests identified the effect of tannin, OBE and HSE extract dosages on flotation of monomineralic fractions of pyrite, arsenopyrite, pyrrhotite and chalcopyrite. The results demonstrated depressing effect on arsenopyrite and pyrrhotite. Floatability of arsenopyrite decreased by 30 %, pyrrhotite - by 75 %. Reduced flotation of chalcopyrite under similar conditions did not exceed 15 %. Active flotation of chalcopyrite may be explained by low concentration of Cu (II) and Fe (II) that may form complexes with tannin incorporated in OBE on the mineral surface, and low concentrations of the reagent (0-20mg / L), moreover having reductive properties.
Flotation tests on selective flotation of Au-As-concentrate of Olympiada deposit showed that the application of OBE extract in designed reagent mode helped to decrease As content in Au concentrate less than 2% (Figure 5).
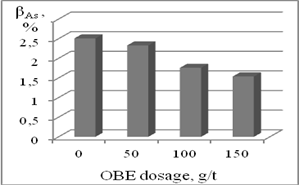
Figure 5. Influence of OBE dosage on As content in Au concentrate.
4. Conclusion
New data on adsorption of tannin and herbal extracts OBE and HSE on pyrite, arsenopyrite and pyrrhotite in the selective flotation of sulfide minerals were obtained. It was identified that tannin and OBE may form stable insoluble complex compounds with Fe (III) on the surface of pyrrhotite and arsenopyrite that helped to hydrophilize mineral surface and decrase its floatability under flotation conditions. At low concentrations tannin and OBE are selective depressants of pyrrhotite and arsenopyrite in a weakly acidic, neutral or weakly alkaline medium. The mechanism of action of depressing tannin-bearing reagents is based on their adsorption on the surface of pyrrhotite and arsenopyrite to form strong complexes with Fe (III) at concentrations below the concentration of micelle formation, and the pre-adsorption of xanthate does not prevent the consolidation of OBE on the surface of the mineral. The application of tannin and the OBE as depressors of pyrrhotite, pyrite and arsenopyrite helped to increase the extraction of valuable components and quality of concentrates by improving the selection of gold-bearing sulfide minerals. Flotation tests on selective separation of Au-As-concentrate of Olympiada deposit showed that the application of OBE extract in designed reagent mode helped to decrease As content in Au concentrate less than 2%.
Acknowledgements
This study is supported by the Russian Foundation for basic research (project RFFI no.13-05-00675).
References
- I.G.Antropova, A.A.Fenin, A.A Revina,Radiation-chemical coumarin transformations in organic solvents,Chemistry of high energy.2 (2007), 90-94.
- V.A.Chanturia, T.N.Matveeva, T.A.Ivanova, N.K.Gromova, L.B. Lantsova, New complexing agents to select auriferous pyrite and arsenopyrite, Journal of Mining Science, 47, 1 (2011), 102–108.
- G.A.Khan, L.I.Gabrielova, N.S.Vlasova, Floatation reagents and their usage, Nedra, Moscow, 1986.
- I.M. Korenman, Fotometrical analysis. Methods of definition of organic compounds, Khimiya, Moscow, 1970.
- V.L. Kretovich, Biochemistry of plants, Second edition, Vysshaya shkola, Moscow, 1986.
- T.N.Matveeva, Scientific grounds for high-performance agent modes in platiniferous sulfide mineral flotation from rebellious ores, Journal of Mining Science, 47, 6, (2011), 824—828.
- T.N.Matveeva, T.A.Ivanova, N.K. Gromova, Sorption and flotation properties of reagents of plant origin – OBE and tannin – in selective flotation of sulfide minerals containing noble metals, Tzevtnye metally, 12 (2012), 16–2
- C.Robertson, D.Bradshaw, P.Harris, Decoupling the effects of depression and dispersion in the batch flotation of a platinum bearing ore, Proc. XXII IMPC (2003), 29 Sept - 3 Oct 2003, Cape Town, South Africa, 920–928.
- E.N. Semenistaja, High performance liquid chromatography in the study of physical and chemical properties of coumarins and their complexes with transition metals, Abstract of dissertation for the degree of Candidate of Chemical Sciences.Moscow,2007.
- L.Ya.Shubov, S.I.Ivankov, N.K. Shcheglova, Floatation reagents in the concentration processes of mineral raw materials, Book 1, Nedra, Moscow, (1990).
- P.Somasundaran, J.Wang, Z.Pan, et al., Interactions of gum depressants with talk: study of adsorption by spectroscopic and allied techniques, Proc. XXII IMPC (2003), 29 Sept - 3 Oct 2003, Cape Town, South Africa, 912–919.
- P.D.Trusov, Organic colloids and their usage in flotation, Proc. of the Leningrad Mining Institute, XII, 3. (1939).



