Investigation on Antimicrobial Activity of Newly Synthesized Mixed Ligand Ni(II) Ion Complexes
Gagi Tauhidur Rahman1, A. B. M. Nazmul Islam2, M. M. Haque3, Md. Kudrat-E-Zahan3, *
1Department of Material Science and Engineering, University of Rajshahi, Rajshahi, Bangladesh
2Department of Chemistry, Khulna University, Khulna, Bangladesh
3Department of Chemistry, University of Rajshahi, Rajshah, Bangladesh
Abstract
Four new mixed ligand complexes of Ni(II) have been prepared. The complexes were characterized on the basis of elemental analyses, conductivity measurements, magnetic measurements, infrared spectral and some physico-chemical analysis. The complexes showed strong to moderate activity against both the gram positive and gram negative bacteria indicating the higher zone of inhibition.
Keywords
Transition Metal Ni(II) Complexes, Glutamic Acid, Mixed Ligand, Antimicrobial Studies
Received: August 13, 2015
Accepted: August 26, 2015
Published online: September 2, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Amino acids are the basic building blocks of all proteins. Proteins are the most abundant biological macromolecules, occurring in all organ and cells. The monomers of amino acids that are covalently linked to each other in sequence by peptide bonds. This chain-linked structure is called a polypeptide or simply a peptide. Proteins exhibit enormous diversity of biological function and are the most important final products of the informal pathways. These are the molecular instruments through which genetic information is expressed. The biological importance of the amino acids includes specific functions involving the metabolism of individual amino acids.
The increasing interest in metal-amino acid [1-3] and peroxo-metal system [4-5] mainly attributable to their potential as models for understanding biological important molecules [6-9] promoted us to establish rational synthetic routes of metals complexes containing an organic acid and amine bases as coligand. Amines are common ligands in all of the present complexes the heterocyclic amines, although they contain tertiary nitrogen, coordinate readily with metal ion. Most of the heterocyclic amines are used as corrosion inhibitor [10-12] and their complexes with platinum and copper have been tested as antitumor [13] and antibacterial agents [14]. Studies on the metal complexes of heterocyclic amine bases have been carried out by many other workers and thus confirmed the complexation ability of amine with metal [17-20].
In continuation of the previous work [21-23] we herein report the synthesis, structural and antimicrobial activity of four new mixed ligand complexes of Nickel(II) containing glutamic acid as primary and heterocyclic amines as secondary ligands.
2. Experimental Procedure
2.1. Reagents
All the reagents and chemicals used were reagent grade and were used as supplied.
2.2. Physical Measurements
The weighing operation was performed on a METTLER PM-200 electronic balance. Conductivity measurements were carried out in dimethyl sulfoxide (DMSO) using a WPACMS 35 conductivity meter and dip-cell with a platinized electrodes. The melting or decomposition temperatures of all the prepared metal complexes were observed in an electro thermal melting point apparatus model No.AZ6512. The SHERWOOD SCIENTIFIC Magnetic Susceptibility Balance was used for the magnetic Susceptibility measurement. Infrared spectra as KBr disc were recorded in a SIMADZU FTIR-8400 (Japan) infrared spectrophotometer, from 4000-400 cm-1. The absorbances of the complexes were recorded on SHIMUDZU Spectrophotometer. Analyses of the complexes for carbon, hydrogen and nitrogen were carried out by Perking 2400 organic elemental analyzer- II at Okayama University, Japan.
3. Preparation of the Complexes [Co(Glu)(L)]
Ni2+ + Glu + L ¾® [Ni(Glu)(L)]
Where, Glu= Glutamic acid, and L = Heterocyclic amine bases such as Pyridine, Quinoline, Isoquinoline, and 2-Picoline.
An ethanolic solution (just dissolved) of Nickel(II)chloride (2 m mol) and ethanolic potassium hydroxide solution (just dissolved) of glutamic acid (4 m mol) were mixed and gently heating with stirring for half an hour. No precipitate was observed but the color has been changed and then secondary ligand in calculated ratio was added and stirred in warm condition until the complex precipitated. The precipitate was filtered, washed several times with alcohol and then dried in vacuum desiccator over phosphorus pentoxide (P2O5).
4. Results and Discussion
4.1. Elemental Analysis and Conductivity Measurement
The analytical data and their physical properties of the complexes are tabulated in Tables 1 and 2 respectively. The elemental data of the complexes showed good agreement with the calculated values of proposed structures. All the complexes are non-hygroscopic and stable at room temperature. The complexes are insoluble in common organic solvents but are soluble in Dimethyl sulfoxide (DMSO), Dimethylformamide (DMF) and Chloroform (CHCl3). The molar conductances of 10-3M solution of the complexes in DMSO were measured at 30°C. The molar conductance values (Table 2), lie in the range 0.12-4.82 W-1cm2mol-1 these values are lower than expected for 1:1 electrolyte [22-23]. These observations suggest that all the complexes are non-electrolyte in DMSO at room temperature and indicated that the anions are covalently bonded in all the cases.
Table 1. Analytical data of Ni(II) complexes.
| Complexes | Yield % | M % | C % | H % | N % |
| [Ni(Glu)2(Q)2] | 69 | 11.51 (12.5) | 55.75 (56.89) | 3.05 (4.74) | 11.05 (12.06) |
| [Ni(Glu)2(IQ)2] | 72 | 11.03 (12.5) | 53.78 (56.89) | 3.45 (4.74) | 12.03 (12.06) |
| [Ni(Glu)2(Py)2] | 68 | 14.62 (15.93) | 45.13 (46.15) | 4.05 (4.94) | 13.66 (15.38) |
| [Ni(Glu)2(2-Pico)2] | 76 | 13.05 (14.79) | 47.05 (48.97) | 5.05 (5.61) | 14.33 (14.28) |
Figure in the parenthesis indicates the calculated values.
Where, Glu= Glutamic acid, Py= Pyridine, IQ= Quinoline, IQ= Isoquinoline, 2-Pico= 2-Picoline.
Table 2. Physical properties of Ni(II) complexes.
| Complexes | Colour | Melting point (± 0.51°C) | Molar conductance W-1 cm2 mol-1 | Magnetic moment meff (B.M.) |
| [Ni(Glu)2(Q)2] | Green | 124 | 2.84 | 3.75 |
| [Ni(Glu)2(IQ)2] | Green | 183 | 4.82 | 2.58 |
| [Ni(Glu)2(Py)2] | Light green | 157 | 0.12 | 2.80 |
| [Ni(Glu)2(2-Pico)2] | Pest | 164 | 3.49 | 2.9 |
Where, Glu= Glutamic acid, Py= Pyridine, IQ= Quinoline, IQ= Isoquinoline, 2-Pico= 2-Picoline.
Table 3. Electronic spectral data of Ni(II) complexes.
| Complexes | λmax (nm) | ||
| Band I | Band II | Band III | |
| [Ni(Glu)2(Q)2] | 212 | 276 | 525 |
| [Ni(Glu)2(IQ)2] | 195 | 280 | 478 |
| [Ni(Glu)2(Py)2] | 220 | 312 | - |
| [Ni(Glu)2(2-Pico)2] | 210 | 325 | - |
Where, Glu= Glutamic acid, Py= Pyridine, IQ= Quinoline, IQ= Isoquinoline, 2-Pico= 2-Picoline.
Table 4. IR spectral data of Ni(II) complexes.
| Complexes | n(N-H) cm-1 | n(C=O) cm-1 | n(M-N) cm-1 | n(C-O) cm-1 | n(COO-) symmetry cm-1 | Aromatic ring cm-1 |
| [Ni(Glu)2(Q)2] | 3140 | 1650 | 310 | 1370 | 1395 | 840 |
| [Ni(Glu)2(IQ)2] | 3096 | 1565 | 370 | 1440 | 1335 | 825 |
| [Ni(Glu)2(Py)2] | 3290 | 1600 | 305 | 1340 | 1368 | 890 |
| [Ni(Glu)2(2-Pico)2] | 3256 | 1627 | 398 | 1147 | 1365 | 835 |
Where, Glu= Glutamic acid, Py= Pyridine, IQ= Quinoline, IQ= Isoquinoline, 2-Pico= 2-Picoline.
Table 5. Zone of inhibition of antifungal activity of test complexes.
| Test fungus | Diameter of zone of inhibition (mm) of test complexes | Nystatin ( 45µg/ disc) | |||||
| [Ni(Glu)2(Q)2] | [Ni(Glu)2(IQ)2] | [Ni(Glu)2(Py)2] | [Ni(Glu)2(2-Pico)2] | ||||
| Tinea pedis | 13 | 11 | 9 | 10 | 20 | ||
| Asperigillus niger | 13 | 10 | 10 | 10 | 26 | ||
| Conlitotrium sp. | 15 | 14 | 11 | 11 | 22 | ||
Where, Glu= Glutamic acid, Py= Pyridine, IQ= Quinoline, IQ= Isoquinoline, 2-Pico= 2-Picoline.
Table 6. Antibacterial activity of the test complexes.
| Test Bacteria | Diameter of Zone of inhibition (mm) of test complexes | Kanamycin (Ts/25µg/disc) | |||
| [Ni(Glu)2(Q)2] | [Ni(Glu)2(IQ)2] | [Ni(Glu)2(Py)2] | [Ni(Glu)2(2-Pico)2] | ||
| Bacillus subtilis | 10 | 12 | 9 | 9 | 16 |
| Streptococcus- Β-haemolytica | 11 | 11 | 9 | 8 | 25 |
| Escharicia coli | 11 | 9 | 8 | 11 | 17 |
| Sarcina lutea | 10 | 11 | 7 | 10 | 19 |
| Klebsella | 11 | 12 | 8 | 9 | 17 |
| Shigella flexeri | 9 | 11 | 9 | 11 | 14 |
| Shigella voydii | 11 | 11 | 8 | 10 | 24 |
| Shigella dysenteriac | 11 | 10 | 8 | 10 | 14 |
| Pseudomonas aeruginosa | 12 | 11 | 10 | 8 | 13 |
Where, Glu= Glutamic acid, Py= Pyridine, IQ= Quinoline, IQ= Isoquinoline, 2-Pico= 2-Picoline.
4.2. IR Studies
Prominent features of the IR spectra of the complexes are tabulated in the Table 4. The complexes display bands in the regions 1604-1682 and 1324-1456 cm-1 due to n(C=O) and n(C-O) respectively, significantly lower than that of free ligand n(C=O) = 1700 cm-1 and n(C-O) = 1600 cm-1 respectively indicating the coordination of ligand through its carboxylate anion. The symmetric stretching of COO- frequencies is observed at 1410-1545 cm-1. The disappearance of the n(O-H) mode observed in the free amino acid molecule clearly indicate the loss of proton for O-H group upon coordination, revealing that acids are dinegative bidentate ligand coordinating through the carboxylate anion. The complexes showed n(N-H) bands from 3200-2850 cm-1 which is significantly lower than the free ligand (amine base bands from 3300-3200 cm-1) clearly suggests the coordination of amino group through nitrogen atoms of amine bases. The in-plane and out-of-plane ring deformation modes of heterocyclic amines observed at 680 and 620 cm-1 respectively undergo a positive shift in mixed ligand complexes confirming their coordination through nitrogen. The presence of metal nitrogen bonding in the complexes is evident from the appearance of n(M-N) modes at 440-478 cm-1 in the spectra of the complexes.
4.3. Magnetic Moment and Electronic Spectra
The observed values of effective magnetic moment (meff) of the complexes at room temperature are given in Table 2. The Ni(II) complexes give magnetic moment in the range 2.80 to 3.94 B.M which indicates a small but definite orbital contribution to the magnetic moment and in agreement with octahedral geometry [24]. The electronic spectrum of Ni(II) complexes showed peaks characteristic of octahedral geometry. The reflectance spectra of Ni(II) complex consist of transition bands from 195 to 525 nm assignable to the transitions 3A2g ® 3T2g and 3A2g(F) ® 3T1g(F)respectively. The spectral components are shown in Table 3.
5. Zone of Inhibition of Antifungal Activity of Test Complexes
The antifungal activity of the test complexes against different fungi were investigated by using the doses of 80 µg/ disc, where standard antibiotic disc of Nystatin (45µg/ disc) was used for comparison purpose. The diameter was evaluated 13 mm, 11 mm, 9 mm, 10 mm against tinea pedis, 13 mm, 10 mm, 10 mm, 10 mm against asperigillus niger, 15 mm, 14 mm, 11 mm, 11 mm against Conlitotrium sp., respectively for test complexes [Ni(Glu)(Q)3], [Ni(Glu)(IQ)3], [Ni(Glu)(Py)3], and [Ni(Glu)(2-Pico)3] whereas diameter of zone of inhibition of Nystatin was found to be 20 mm, 26 mm, 22 mm respectively against the organism. The antifungal activity (zone of inhibition) of the test complexes against respective fungai is presented in Table-5.
6. The Result of Antibacterial Activity of Test Complexes
The antibacterial activity of the test complexes were determined by using the dose of 80 µg/disc. The results of antibacterial activity measured in terms of zone of inhibition is shown in Table-6. The complexes showed minimum sensitivity against the following number of both gram positive and gram negative bacteria and the results were compared with antibiotic disc of kanamycin.
7. Conclusion
It is concluded that the analytical data were in good agreement with the proposed empirical formulae of the complexes. Magnetic susceptibility measurements showed the complexes to be paramagnetic. The IR spectral data indicated the ligand coordinate to the metal through O and N atoms. The electronic spectral data are in conformity with the transitions of octahedral Ni(II) complexes. On the basis of the above investigations and other physical interpretation the probable molecular structure of the complex [Ni(Glu)(Py)3] could be illustrated as shown below (Figure-1):
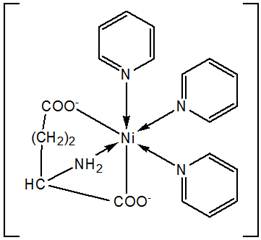
Figure 1. Proposed structure of complex [Ni(Glu)(Py)3].
References
- Moszner, M., (2004) "Water replacement on the decaaqua-di-rhodium(II) cation; synthesis of superoxo and peroxo rhodium(III) complexes with N-donor ligands", Inorg. Chim. Acta., 357(12), 3613-3620.
- Teramae, S., Osako, T., Nagatomo, S., Kitagawa, T., Fukuzumi, S., and Itoh, S., (2004). "Dinuclear copper-dioxygen intermediates supported by polymine ligands", J. Inorg. Biochem., 98(5), 746-757.
- Ahmed, M., Schwendt, P., Marek, J., and Sivak, M., (2004). "Synthesis, solution and crystal structures of dinuclear vanadium(V) oxo monoperoxo complexes with mandelic acid: (NR4)2[V2O2(O2)2(mand)2]xH2O [R=H, Me, Et; MAND = mandelato(2-) = C8H6O32-]", Polyhedron., 23(4), 655-663.
- Aquino, A.R., Isolani, P.C., Zukerman-Schpector, J., Zinner, L.B., and Vicentini, G., (2001). "Uranyl nitrate complexes with lactams", J. Alloys and Com., 323, 18-21.
- Bryliakov, K. P., Karpyshev, N. N., Fominsky, S. A., Tolstikov, A. G., and Talsi, E. P., (2001). "51V and 13C NMR spectroscopic study of the peroxovanadium intermediates in vanadium catalyzed enantioselective oxidation of sulfides", J. Mol. Catal. A: Chem., 171(1-2), 73-80.
- Fricker, S. P., (1999). "A screening strategy for metal antitumor agents as exemplified by Gold (III) complexes", Metal-Based Drugs., 6, 291-300.
- Silver, S., Gupta, A., Matsui, K., and Lo, J., (1999). "Resistance to silver(I) cations in bacteria: Environments, genes and proteins", Metal-Based Drugs., 6, 315-320.
- Thunus, L., and Lejeune, R., (1999) "Overview of transition metal and lanthanide complexes as diagnostic tools" Coord. Chem. Rev., 184, 125-155.
- Ronconi, L., Marzano, C., Russo, U., Sitran, S., Graziani, R., and Fregona, D., (2003). "Organotin(IV) complexes of ethylsarcosine hydrochloride: synthesis, characterization and in vitro cytotoxic activity", Appl. Org. Met. Chem., 17(1), 9-16.
- Talati, J. D., and Gandhi, K. K., (1983). "N-Heterocyclic compounds as corrosion inhibitors for aluminium-copper alloy in hydrochloric acid", Corr. Sci., 23, 1315-1332.
- Rysakova, L.A., and Loshkarev, M.A., (1984). "Absorption and inhibitor properties of nitrogen were containing six membered heterocyeles (quinoline) in a neutral medium", Elektrokhimiya., 20, 1102-1109.
- Konno, H., Nagayaama, M., Leidheiser, Jr. H., and Granata, R.D., (1984). "The composition and properties of a protective layer formed on a steel by anodizing with 8-hydroxyquinoline", Kinzoku-Hyomer Gijutsu., 35, 402-408.
- Doadrio, A., Craciunescu, D., Sosa, B., and Fruma, A., (1979). "Relations between the structure and antitumor activity of complex platinate and cuprate salts", An. R. Acad. Farm., 45, 497-506.
- Heinish, L., Fleck, W. F., and Jacob, H. E., (1980). "Copper(II) complexes with N-heterocyclic formyl-isothiosemicarbazone with antibacterial andb-lactamase inhibiting effect", Z. Allg. Mikrobiol., 20, 619-619.
- Doadrio, L.A., Lozano, F.R., and Doadrio, V.A.L., (1980). "A new dimeric complex of Mo(VI) and 8-hydroxyquinoline", Ann. Quim. B., 76, 193-197.
- Aly, M.M., and Shalaby, A.M., (1980). "Coordination of zwitterionic-8-quinolinol (oxine) to mixed oxalates-carboxylate complexes of divalent nickel, manganese and magnesium", Monatsh Chem., 111(4), 935-943.
- Gomez-Vaamonde, C., Alverez-valdes, A., Navarro-Ranningar, M.C., and Masagure, J.R., (1984). "Synthesis and characterization of complexes of Pd(II) with 2-aminopyidine", Transition Met. Chem., (9)2, 52-54.
- Wenclawiak, B., and Flemming, M., (1987). "Preparation and characterization of all platinum metal 8-hydroxyquinolinates", Fresenius Z. Anal. Chem., 326, 551-557.
- Swamy, B., Donnel, M., and Frederick, M., (1998). "Synthesis of free Ruthenium coordinated 5,6-diamino-1,10-phenanthroline", Tetrahedron Lett., 38, 8159-8160.
- Claudio, P., and Fabio, M.H., (2000). "Novel bis (acylparazolonato) cadmium(II) derivatives and their reactivity towards aromatic and aliphatic N2 donor ligand", Dalton., 5, 831-836.
- M.M.Haque, Md.Kudrat-E-Zahan Laila Arjuman Banu, Md.Shariful Islam and M.S.Islam,(2015) "Synthesis and Characterization with Antineoplastic, Bio-Chemical, Cytotoxic and Antimicrobial studies of Schiff Base Cu(II) ion Complexes", Bioinorg. Chem. Appl., http://dx.doi.org/10.1155/2015/923087
- Md.Kudrat-E-Zahan, M. S. Islam and Md.Abul Bashar. (2015) "Synthesis, Characterization and antimicrobial activity of Some Metal Complexes of Mn(II), Fe(III) Co(II), Ni(II), Cu(II) and Sb(III) Containing Bidentate Schiff base of SMDTC", Rus. J. gen Chem., 85 (3), 667-672.
- Md.Kudrat-E-Zahan and M. S. Islam. (2015) "Synthesis, Characterization and Antimicrobial activity of Some Metal Complexes of Cu(II), Ni(II), Zn(II), Pb(II),Co(II), Mn(II) and U(VI) Containing Bidentate Schiff base of [S-methyl-3-(4-methoxybenzylidine)dithiocarbazate]", Rus. J. gen Chem., 85 (4), 979-983.
- N.V.Thakkar and R.M.Patil. (2010) "Synthesis of Mononuclear Metal Complexes with Some Tetradentate Schiff Base Ligands", Synt. React. Inorg. Metal-Org. Chem., 30(6), 1159-1174.



