Ortho-Diphenol Content, Iron Chelating and Hydrogen Peroxide Scavenging Properties of Algerian Virgin Olive Oils
Hayette Louaileche1, *, Ouassila Zegane1, Sonia Keciri2
1Laboratory of Applied Biochemistry, Faculty of Life and Natural Sciences, University of Bejaia, Bejaia, Algeria
2Technical Institute of Arboriculture of Fruits and Vine (TIAFV), Bejaia, Algeria
Abstract
In the current study, ortho-diphenol content and antioxidant potential of virgin olive oil from two Algerian cultivars (Chemlal and Frantoio) were investigated. The antioxidant activity was evaluated using two spectrophotometric methods: ferrous iron chelating ability and hydrogen peroxide (H2O2) scavenging power. Our results revealed that in addition to the cultivar, both ortho-diphenol content and antioxidant activity of olive oils were significantly influenced by the geographical origin of the olive fruit. Oil extracts of Chemlal cultivar from Biskra had the highest level of ortho-diphenols and exhibited the strongest antioxidant potential, whereas the lowest ortho-diphenol content and the weakest antioxidant activity were recorded for oil of Frantoio cultivar from Biskra. The investigated oils are a good source of bioactive ortho-diphenols confirming the interest of olive oil consumption to prevent oxidative stress and diseases where oxygen reactive species are involved.
Keywords
Virgin Olive Oils, Antioxidant Activity, Ortho-Diphenols, Cultivar
Received: August 5, 2015
Accepted: August 19, 2015
Published online: September 10, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Olive oil became far more important throughout Mediterranean and non-Mediterranean areas, not only as a staple food, but also as a source of energy and a pharmaceutical (Boskou, 2006). Virgin olive oil is the pillar fat of Mediterranean diet. It is made from olive fruits and obtained by squeezing olives without any extraction with solvent (Vitaglione et al., 2015). A part of the beneficial effects of the Mediterranean diet may be due to the presence of "minor components", such as olive oil-derived ortho-diphenols, that exhibit biologically relevant activities (Visioli and Galli, 1998).
According to Visioli and Galli (2001) and Cicerale et al. (2009), experimental data obtained from human and animal studies demonstrate that olive oil phenolics displayed biological properties which may be involved in the reduction in risk and severity of many chronic and degenerative disorders based on inflammatory pathways, cardiovascular-cerebral diseases, breast and colon cancers.
Among olive phenolics, ortho-diphenols including hydroxyl-tyrosol and oleuropein are recognized as the most important in relation to their antioxidant activity which can be related to hydrogen donation, i.e., their ability to improve radical stability by forming an intra-molecular hydrogen bond between the hydrogen of their hydroxyl group and their phenoxyl radicals (Visioli and Galli, 1998).
Algeria has approximately 300,000 ha of olive trees of which 80 % is for oil extraction. Oil production ranges from approximately 50,000 to 70,000 t.
The purpose of the current study is to investigate and to compare the ortho-diphenol content, as well as the anti-oxidant activity of virgin olive oils of two cultivars (Chemlal and Frantoio) from Biskra, Algeria. The influence of the geographical origin on the investigated parameters was also evaluated on Chemlal which is the most representative olive cultivar in Algeria.
2. Materials and Methods
2.1. Samples
The virgin olive oils of two cultivars (Frantoio and Chemlal) were used in the present study. Frantoio cultivar was harvested from Biskra whereas Chemlal cultivar was from three cultivation areas (Biskra, Skikda and Bouira). The oil samples were stored in amber glass bottles at 4°C in the dark, until analysis.
2.2. Extraction Procedure
The phenolic extracts were obtained following the procedure of Mraicha et al. (2010) with slight modifications. Oils (10g) were dissolved in 10 ml of methanol/water 60% (v/v) and 5 ml of n-hexane. After stirring, the mixtures were centrifuged for 15min at 5000 rpm. The hydroalcoholic phase is recovered while the hexanic phase will undergo a twice extraction with 10ml of methanol 60%. The fractions were combined, and washed with n-hexane.
2.3. Determination of O-Diphenol Content
O-diphenol content was determined as reported by Bendini et al. (2003). Olive oil extracts (4 ml) were mixed with 1 ml of sodium molybdate. After 15 min, the absorbance was measured at 370 nm. O-diphenol content calculated from the calibration curve of caffeic acid (y= 20.56x+0.0124; R2 = 0.99) was expressed as mg caffeic acid equivalents per kg of oil (mg CAE/Kg).
2.4. Determination of Antioxidant Activity
2.4.1. Ferrous Iron Chelating (FIC) Assay
The determination of ability of the extracts to chelate iron (II) was carried out according to Decker and Welch (1990). A volume of 250 µl of extract was added 25 µl of FeCl2 (2 mM) and 800 µl of distilled water. After 5 min, 50 µl of 5.0 mM ferrozine were added to the mixture and the absorbance measured at 562 nm after 5 min of reaction. The chelating activity was calculated using the following formula:
Ferrous iron chelating (%) = [1-(As-Ab)/Ac] x100
where Ac is the absorbance of the control, As is the absorbance of the mixture containing the extract and ferrozine and Ab is the absorbance of the mixture containing the extract without ferrozine.
2.4.2. Scavenging Capacity on Hydrogen Peroxide
The scavenging capacity of the oil extracts on hydrogen peroxide was determined according to Ruch et al. (1989). One hundred and fifty microlitres of extracts were mixed with 1 ml of hydrogen peroxide (40 mM) in phosphate buffer (0.1M; pH 7.4) and 1350 µl of phosphate buffer solution (0.1 M, pH 7.4). After 10 min of incubation, the absorbance was recorded at 230 nm. The hydrogen peroxide scavenging activity was calculated using the following formula:
scavenging effect (%) = [(Ac–(Ae-Ab)/Ac]*100
where Ac is the absorbance of the control, Ae is the absorbance of the extract and Ab is the absorbance of the blank.
2.5. Statistical Analysis
The data reported were subjected to analysis of variance and were expressed as mean ± standard deviation (SD) of three independent measurements. Significant differences between values of all parameters were determined at p≤0.05 according to the least significant difference (LSD) test using the Statistica 5.5 software.
3. Results and Discussions
3.1. Ortho-Diphenol Content
As shown in figure 1, the O-diphenol content of the analyzed oils varies significantly (p≤0.05) among the different cultivars. Oil extract of Chemlal from Biskra has the highest O-diphenol content (30.99 mg CAE/Kg) while the lowest content is noted for the oil of Frantoio from Biskra (12.31 mg CAE/Kg) and Chemlal from Skikda (13.60 mg CAE /Kg).
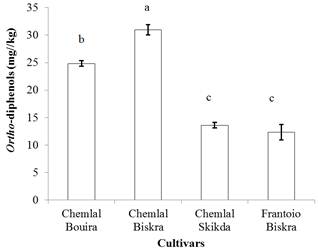
Fig. 1. Ortho-diphenol contents of olive oil samples. Different
letters indicate statistically significant differences (p < 0.05).
O-diphenol contents recorded in the present study are in agreement with those obtained by Bubola et al. (2014) for Croatian virgin olive oils, Haddam et al. (2014) for olive oil from Morocco and Sousa et al. (2014) for oils of one of the main olive Portuguese cultivar (Cobrançosa). However, the amounts of O-diphenols detected for the samples examined are lower than those reported by Gutierrez et al. (2002) for Spanish varieties (Picual, Arbequina and Hojiblanca) and Guerfel et al. (2009) for Tunisian varieties (26.91 to 261.24 mg/kg). The variations noted could be attributed to several parameters, including the olive cultivar, the degree of olive ripening, and environmental factors (climatic conditions, geographical origin, etc.).
3.2. Antioxidant Activity
In view of the complexity of diverse oxidation process and the nature of antioxidants, there is no universal method that can measured in a precise way, the antioxidant activity. In the present work, two assays (ferrous iron chelating ability and hydrogen peroxide scavenging power) were carried out to evaluate the antioxidant capacity of the olive oil extracts.
3.2.1. Hydrogen Peroxide Scavenging Power
Hydrogen peroxide itself is not very reactive. However, it can be toxic for cells because the super-active hydroxyl radical (OH.) may be formed from the Fenton reaction in the presence of metal ions. Also, excess of hydrogen peroxide produced in the pathogenesis of brain injuries and neurodegenerative diseases may damage cells through direct peroxidation of lipids, and oxidation of proteins.
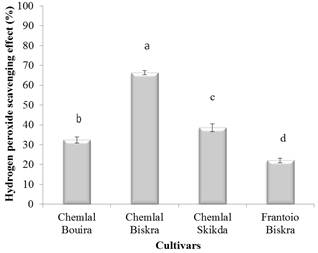
Fig. 2. Hydrogen peroxide scavenging activity of olive oil extracts.
Different letters indicate statistically significant differences (p < 0.05).
Figure 2 indicates that all of the analyzed olive oil extracts have the ability to scavenge the hydrogen peroxide and their ability varied significantly (p<0.05). Chemlal cultivar from Biskra exhibited the highest hydrogen peroxide scavenging effect (66.32%), while the weakest activity (22.07 ± 1.10%) is marked for the extract of Frantoio cultivar from Biskra. Limited information is so far available on the hydrogen peroxide scavenging activity of olive oil extracts. Data obtained by Franco et al. (2014) showed slight variations in the hydrogen peroxide scavenging capacity (69.9–76.8%) of the Spanish virgin olive oil methanolic extracts.
3.2.2. Ferrous Iron Chelating (FIC) Ability
Transition metals have a major role in the generation of oxygen free radicals in living organisms. The production of these reactive oxygen species contribute to the damage of biological molecules (proteins and lipids).
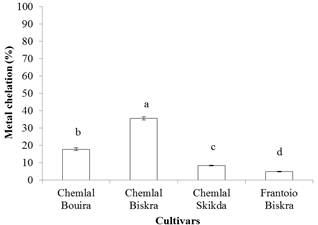
Fig. 3. Ferrous iron chelating ability of olive oil extracts. Different
letters indicate statistically significant differences (p < 0.05).
Iron exists in two distinct oxidation states (ferric and ferrous ions). The ferric ion (Fe3+) is the relatively biologically inactive form of iron. However, it can be reduced to the active ferrous ion (Fe2+), and oxidized back through Fenton type reactions, with production of hydroxyl radicals. Consequently, the chelating agents of transition metals such as iron may have an essential role for antioxidant defense in the biological systems and food matrices.
Concerning the ferrous iron chelating capacity, its values varied significantly (p<0.05) between the investigated oil samples. The results of iron chelating capacity by extracts of different samples are shown in Fig. 3. For the analyzed oils, the highest chelating activity is recorded for Chemlal variety from Biskra (35.60±0.83%) whereas Frantoio cultivar from Biskra exhibited the lowest activity (5.00 ± 0.24%). The differences in the chelating ability between samples could be due to the differences in phytochemical content, under the effect of varietal and geographical factors. Data on ferrous chelating potential of extract of olive oils are not available in the literature.
3.3. Correlation Between O-Diphenols Content and Antioxidant Power
Relationships were obtained between the O-diphenol content and the antioxidant capacities assessed by hydrogen peroxide scavenging potential (figure 4) and metal chelating ability (figure 5), revealed from their good correlation (R2= 0.96 and R2= 0.88, respectively). The antioxidant properties of the O-diphenols are associated with their ability to form intra-molecular hydrogen bonds between the hydroxyl group and the phenoxylic radicals (Visioli and Galli, 1998). On the other hand, Carrasco-Pancarbo et al. (2006) indicated that the presence of O-diphenols enhances the ability of the phenolic compounds to act as antioxidants. This relationship suggested that the O-diphenols of olive oil extracts might be the major contributors to the antioxidant activities assessed by these methods.
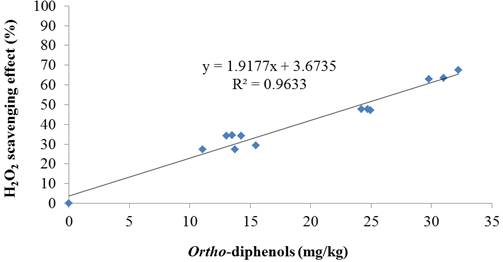
Fig. 4. Correlation between ortho-diphenol content and hydrogen peroxide (H2O2) scavenging activity of olive oil extracts.
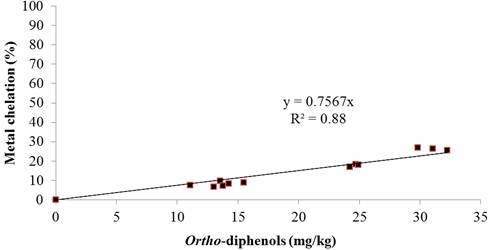
Fig. 5. Correlation between ortho-diphenol content and iron chelating effect of olive oil extracts.
4. Conclusion
Several studies have been carried out to investigate the free radical scavenging potential of olive oils but to our knowledge, the literature focused on the data on hydrogen peroxide scavenging effect and metal chelating ability is scarce. Results of this study showed a different in ortho-diphenols content, a varietal distinction. Chemlal variety from Biskra contains the highest amount of O-diphenols.
Antioxidant activity evaluated in vitro was conducted by two tests, including ferrous metal chelating potential and hydrogen peroxide scavenging effect. The antioxidant activity of methanol extracts of olive oils varied greatly depending on the variety and growing area. This study provides basic information on the presence of bioactive compounds and antioxidant capacity in olive oil. The results obtained confirm that these oils contribute in the preventing diseases where reactive oxygen species are involved. It will be interesting to carry out research on the identification and structural characterization of different phenolic compounds of these oil extracts.
Acknowledgments
Thanks are due to all persons who have provided the olive samples of this study.
References
- Bendini A., Bonoli M., Cerretani L., Bigguzi B., Lercker G.andToschi, T.G. 2003.Liquid and solid-phase extractions of phenols from virgin olives oil and their separation by chromatographic and electrophoretic methods. Journal of Chromatography, 985: 425–433.
- Bengana M., Bakhouche A., Sanchez- Lozano J., Amir Y.,andYouyou A. 2013. Influence of olive ripeness on chemical properties and phenolic composition of Chemlal extra-virgin olive oil. Food Research International, 54: 1868–1875.
- Boskou D. 2006.Characteristics of the Olive Tree and Olive Fruit inOlive Oil: Chemistry and Technology.Editor, Boskou, D. 2nd edition.
- Bubola K.B., Koprivnjak O., Sladonja B.andBelobrajic I. 2014. Influence of storage temperature on quality parameters, phenols and volatile compounds of Croatian virgin olive oils. Grasas Y Aceites, doi: http://dx.doi.org/10.3989/gya.022214.
- Carrasco-Pancorbo A., Gomez-Caravaca A.M., Cerretani L., Bendini A., Segura-CarreteroA.and Fernandez-Gutierrez A. 2006. Rapid quantification of the phenolic fraction of Spanish virgin olive oils by capillary electrophoresis with UV detection. Journal of Agricultural and Food Chemistry, 54(21): 7984-7991.
- Cicerale S., Conlan X.A., Sinclair A. J. and Keast R.S.J. 2009. Chemistry and health of olive oil phenolics. Critical Reviews in Food Science and Nutrition, 49: 218-236.
- Criado M.N., Morello J.R., Motilva M.J. and Romero M.P. 2004. Effect of growing area on pigment and phenolic fractions of virgin olive oils of the Arbequina variety in Spain. Journal of American Oil Chemists Society, 81: 633-640.
- Decker E.A.andWelch B. 1990. Role of ferritin as a lipid oxidation catalyst in muscle food. Journal of Agricultural Food Chemistry, 56: 674-677.
- Franco M.N., Galeano-Díaz T., Loppez O., Fernández-Bolaños J.G., Sánchez J., De Miguel C., Gil V. and Martín-Vertedor D. 2014. Phenolic compounds and antioxidant capacity of virgin olive oil. Food Chemistry, 163: 289–298
- Guerfel M., Ouni Y., Taamalli A., Boujnah D., Stefanoudaki E. and Zarrouk M.2009. Effect of location on virgin olive oils of the two main Tunisian olive cultivars. European Journal of Lipid Science and Technology, 111: 926–932.
- Gutiérrez F., Villafranca M.J. and Castellano J.M.2002.Changes in the main components and quality indices of virgin olive oil during oxidation. Journal of the American Oil Chemists' Society, 79(7): 669-676.
- Haddam,M., Chimi H., El-Antari A., Zahouily M., Mouhibi R., Zaz A., Ibrahimi M. and Amine A.2012.Caractérisation physico-chimique et stabilité oxydative des huiles d’olive des variétés Picholine marocaine, Haouzia, Koroneiki et Arbéquine de la région oléicole centrale du Maroc (Chaouia-Ouardigha). Olivae,119: 23-35.
- Mraicha F., Ksantini M., Zouch O.,Ayadi M., Sayadi S. and Bouaziz M.2010. Effect of olive fruit fly infestation on the quality of olive oil from Chemlali cultivar during ripening. Food and Chemical Toxicology, 48: 3235–3241.
- Ruch R.J., Cheng S.J. and Klaunig J.E. 1989. Prevention of cytotoxicity and inhibition of intercellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogen, 10:1003–1008.
- Serteser A., Kargıolu M., Gök V., Özcan M.and Arslan D. 2009. Antioxidant properties of some plants growing wild in Turkey. Grasas Y Aceites, 60 (2): 147-154.
- Vitaglione P., Savarese M., Paduano A., Scalfi L., Fogliano V., and Sacchi R. 2015. Healthy virgin olive oil: a matter of bitterness. Critical Reviews in Food Science and Nutrition, 55:1808–1818.
- Visioli F.andGalli C. 1998. The effect of minor constituents of olive oil on cardiovascular disease: new findings. Nutrition Reviews, 56: 142–147.
- Visioli F. and Galli C. 2001. Antiatherogenic components of olive oil. Current Atherosclerosis Reports, 3: 64–67.



