Eclosion Rhythm of Plodia interpunctella Under Non-24 h Thermocycles
Shigeru Kikukawa*, Yuta Kakihara, Yuki Okano, Renpei Shindou, Naoyuki Sugino, Jinnai Tsunekawa, Akihiro Yasui, Kazuhito Yoneda
Biological Institute, Faculty of Science, University of Toyama, Toyama, Japan
Abstract
The present study focuses on the adult eclosion rhythm of the Indian meal moth, Plodia interpunctella Hübner (Lepidoptera: Pyralidae), under two non-24 h thermocycles of 30°C/20°C and 25.5°C/24.5°C in constant darkness (DD). The thermocycles were provided as two combinations: one with a constant 12-h thermophase (T) alternated with a cryophase (C) of varying durations (6-18 h) and the other with a constant 12-h C alternated with varying durations (6-18 h) of the T. For 30°C/20°C thermocycle with a constant 12-h T, the temporal position of the adult eclosion peak is observed to advance as the cycle length extends. The peak occurs within the T and appears at about 17 h after the temperature-fall. However, when the duration of the C is constant for 12 h, the average peak time of adult eclsion is Zt 5.0 after the temperature rises. Under the 25.5°C/24.5°C thermocycle, the temporal position of the adult eclosion peak is a function of the cycle length. As the insects do not experience non-24 h thermocycles in nature, our results provide clues to the basic nature of the time-keeping mechanism of P. interpunctella.
Keywords
Adult Eclosion Rhythm, Indian Meal Moth, Plodia interpunctella, Non-24 h Thermoperiod
Received: July 16, 2015
Accepted: July 30, 2015
Published online: August 26, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
The Indian meal moth, Plodia interpunctella Hübner (Lepidoptera: Pyralidae), shows rhythmic patterns in adult eclosion under various thermocycles (1). The temporal position of the peak eclosion (φE) is dependent on the level and amplitude of the temperature cycle. In a thermocycle of 30°C/20°C in continuous darkness (DD) with Thermophase: Cryophase (TC) 12:12 h, φE is zeitgeber time (Zt) 4.7 ± 1.7 (mean ± standard deviation) after the rise in temperature. The time at which the temperature rises during the thermocycle is designated as Zt 0. This peak advances in time as the amplitude of the thermocycle decreases. For TC 12:12 h (25.5°C/24.5°C), φE is Zt 21.9 ± 3.2 in late cryophase. These observations are recorded under a "24-h" thermocycle.
Generally, a time-keeping mechanism underlying a circadian rhythm does not oscillate exactly with a 24-h period (see 2, 3, 4, 5, for review). Most insects, if not all, exhibit "circa" 24-h free-running rhythms under constant conditions. The time-keeping oscillator is calibrated every day by an external time cue referred to as Zeitgeber such as the photoperiod and/or thermocycle (1, 6).
The present study focuses on whether the adult eclosion rhythm of P. interpunctella is modified by thermocycles of 30°C/20°C and 25.5°C/24.5°C under DD. To analyze the entrainability, the insects are maintained under non-24 h thermocycles and their eclosions as adults are observed. Such a non-24 h treatment has not been studied extensively amongst insect groups. Earlier, Arai studied the adult eclosion rhythm of Dacus dorsalis (7) and the egg hatching rhythm of Eobiana engelhardti subtropica (8) and concluded that, under non-24 h thermocycles, these rhythms synchronize with thermoperiod of approximately 24 h duration. The present study explores the basic physiological mechanism of the adult eclosion clock in P. interpunctella in relation to circadian organization.
2. Materials and Methods
A laboratory culture of P. interpunctella was established from larvae collected at Toyama in 1998 and maintained as described earlier (6). These insects enter diapause at the 5th larval instar in response to environmental conditions such as short photoperiods and/or thermoperiods (9, 10). In the present study, to prevent the insects from entering diapause, eggs deposited within 24 h in constant darkness (DD) were maintained at 30°C for 14 days after oviposition. This treatment prevented diapause induction completely. The larvae were then exposed to different lengths (non-24 h varying from 18 to 30 h) of thermocycles in DD. The thermocycles used were 30°C/20°C and 25.5°C/24.5°C in which the thermophase (T) and the cryophase (C) were alternately fixed at 12 h, and accordingly the time duration of the other phase (C or T) was either shortened or prolonged. Thus, TC 12:8 h was a thermocycle of 12-h thermophase and 8-h cryophase. After exposure of the larvae to the thermocycle, the number of adults that emerged (n = 97 – 321) was counted within a few minutes at 1-h intervals under a dim red light of <100μW/cm2 (see also 3). The temporal position of the peak (φE) of the adult eclosion rhythm was represented by the mean ± standard deviation (SD). The recordings of adult eclosion during the entire observation period for each non-24 h temperature cycle were compiled to analyze the peak of the adult eclosion rhythm.
3. Results
3.1. Temperature Cycle of 30°C/20°C
A peak of adult eclosion rhythm was observed at Zt 4.7 ± 1.7 (mean ± SD) after the rise in temperature in the thermocycle of TC 12:12 h (24-h cycle) (30°C/20°C) in constant darkness (DD) (1). The entrainability of this circadian oscillator was further investigated under non-24 h thermocycles in DD. In the first series of experiments, the larvae were exposed to thermocycles consisting of 12-h thermophase (30°C) and various lengths (6-18 h) of the cryophase (20°C). When TC was 12:6 h, larvae eclosed as adults at random and no peak was observed (data not shown). However, the rhythm of adult eclosion appeared to be bimodal in TC 12:7 h (Fig. 1A). Adult eclosion, however, occurred continuously for a long period, hence, the peak could not be specified and circadian rhythm was not detected. For TC 12:8 h (Fig. 1B), TC 12:9 h (Fig. 1C), TC 12:10 h (Fig. 1D) and TC 12:11 h (Fig. 1E), the temporal position of the eclosion peak was Zt 9.1 ± 1.8, Zt 8.7 ± 2.1, Zt 7.2 ± 1.7 and Zt 6.4 ± 2.2, respectively, after the increase in temperature. Thus, the peak advanced and appeared earlier, as the duration of the cryophase increased (see also Fig.5). In these series of experiments, the larvae were exposed to 9-17 thermocycles. When the cycle length of the thermoperiod was >24 h, the temporal position of the peak was further advanced and appeared earlier (see also Fig. 5). The observations for TC 12:13 h (Fig. 1F), TC 12:14 h (Fig. 1G) and TC 12:15 h (Fig. 1H) showed the peaks at Zt 4.0 ± 2.1, Zt 3.5 ± 3.4 and Zt 2.8 ± 2.9, respectively. Under these conditions, larvae experienced 10-16 thermocycles. For TC 12:14 h and TC 12:15 h, many larvae eclosed as adults within 1 h after the temperature rose. Based on these observations, it can be inferred that adult eclosion may be triggered directly by the shock of the temperature-rise. According to our observations for TC 12:16 h, TC 12:17 h and TC 12:18 h, > 60 % of larvae eclosed within 4 h after the temperature rose (data not shown), and no adult eclosions were observed during the cryophase. Thus, the time-keeping mechanism of this insect appears to be entrained by thermocycles ranging from TC 12:8 h to TC 12:15 h.
In the second series of experiments, the duration of the thermophase (30°C) was varied and the cryophase (20°C) was kept constant at 12 h. The observations showed that for TC 6:12 h and TC 7:12 h, >55 % of larvae eclosed as adults within 1 h after the rise in temperature (data not shown). Thus, adult eclosion, appeared to be stimulated directly by temperature change from 20°C to 30°C. It was observed that, for TC 8:12 h (Fig. 2A), the adult eclosion rhythm was bimodal: about 30 % of the larvae eclosed as adults within 3 h after the temperature rose, whereas eclosion of the remaining 43 % of the larvae was observed between Zt 7.0 and Zt 9.0 after the temperature rose. It can be inferred that this second peak, in fact, represents the true peak of the rhythm. In the series of experiments for TC 9:12 h (Fig. 2B), TC 10:12 h (Fig. 2C) and TC 11:12 h (Fig. 2D), the peaks of adult eclosion rhythm were observed at Zt 4.6 ± 1.4, Zt 6.1 ± 2.7 and Zt 6.0 ± 2.1, respectively. Under these conditions, larvae experienced 10-17 thermocycles. For TC 13:12 h (Fig. 2E) and TC 14:12 h (Fig. 2F), the peaks were at Zt 4.2 ± 2.7 and Zt 4.6 ± 3.6, respectively (see also Fig. 5). Under these conditions, larvae were exposed to 8-15 thermocycles. According to our observations of the larvae with TC 15:12 h to TC 18:12 h, c. 60 % of larvae eclosed as adults within 1 h after the temperature rose (data not shown). Based on these observations, it can be inferred that adult eclosion pattern was apparently triggered by the temperature change from 20°C to 30°C, and eclosion occurred mostly during the thermophase, with only a few exceptions of adult eclosion during the cryophase. The results of these experiments indicate that the time-keeping mechanism was entrained by thermocycles ranging from TC 9:12 h to TC 14:12 h, and the average peak time of adult eclosion was Zt 5.0 after the temperature rose. Though the regression analysis (line b in Fig. 5B) showed r = 0.4639 (P = 0.3541) which is insignificant, it can be concluded that, when the duration of cryophase was constant 12 h, the peak adult eclosion occurred around Zt 5.0 after the increase in temperature.
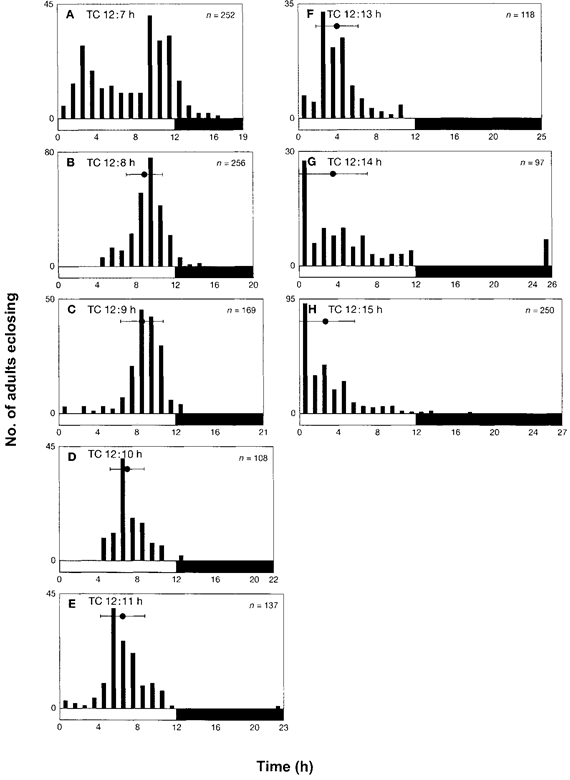
Figure 1. Adult eclosion of the Indian meal moth Plodia interpunctella under various non-24 h temperature cycles in constant darkness (DD). (A) TC 12:7 h, (B) TC 12:8 h, (C) TC 12:9 h, (D) TC 12:10 h, (E) TC 12:11 h, (F) TC 12:13 h, (G) TC 12:14 h and (H) TC 12:15 h where T represents a thermophase of 30°C and C represents a cryophase of 20°C. The number (n) of adults that demonstrate eclose was observed at 1-h intervals. Horizontal white and black bars show the thermophase and cryophase of each thermocycle, respectively. ● indicates the mean eclosion time with standard deviation (bar). Note that the thermophase was held constant at 12 h.
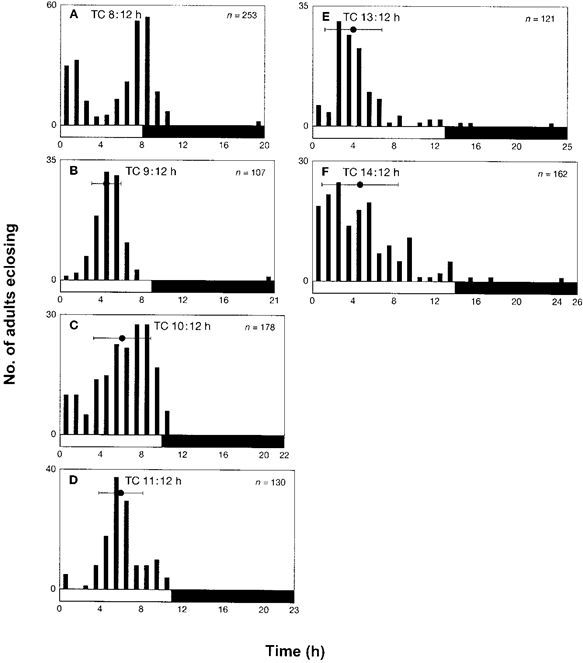
Figure 2. Adult eclosion of Plodia interpunctella under various non-24 h thermocycles of 30°C/20°C in DD. (A) TC 8:12 h, (B) TC 9:12 h, (C) TC 10:12 h, (D) TC 11:12 h, (E) TC 13:12 h and (F) TC 14:12 h (refer Fig. 1). Note that the cryophase was held constant at 12 h.
3.2. Temperature Cycle of 25.5°C/24.5°C
Noting that the 10°C increase in temperature stimulated adult eclosion in some thermoperiodic regimes as described above, we further investigated eclosion in a temperature cycle of 25.5°C/24.5°C to eliminate the effect of temperature shock. For TC 12:12 h, we observed that adult eclosion occurred at Zt 21.9 ± 3.2 after the rise in temperature (1). First, the larvae were maintained in a 12-h thermophase with various lengths of the cryophase (6-18 h). On exposure of the larvae to TC 12:6 h (data not shown) and TC 12:7 h (Fig. 3A), adult eclosion was arrhythmic and there was no single peak of adult eclosion. However, on exposure to TC 12:8 h (Fig. 3B), TC 12:9 h (Fig. 3C), TC 12:10 h (Fig. 3D) and TC 12:11 h (Fig. 3E), the peaks of adult eclosion occurred at Zt 10.9 ± 2.9, Zt 8.7 ± 3.0, Zt 4.3 ± 3.0 and Zt 1.9 ± 2.6, respectively. Thus, the peak advanced and appeared earlier as the cryophase prolonged (see also Fig. 5). When the cycle length became longer than 24 h, the peak advanced even further. For TC 12:13 h (Fig. 3F) and TC 12:14 h (Fig. 3G), the temporal positions of the adult eclosion peak were Zt 16.9 ± 4.1 and Zt 13.9 ± 5.9, respectively. However, the value of SD was relatively large and, in TC 12:15 h (Fig. 3H), no recognizable peak was observed. For TC 12:16 h, TC 12:17 h and TC 12:18 h, the adult eclosion was arrhythmic (data not shown). Thus, based on the above observation, it can be inferred that the time-keeping mechanism was entrained by thermocycles ranging from TC 12:8 h to TC 12:14 h. On exposure to temperature cycles that were <24 h, larvae experienced 13-24 thermocycles, whereas, on exposure to temperature cycles that were >24 h, larvae experienced to 12-18 thermocycles.
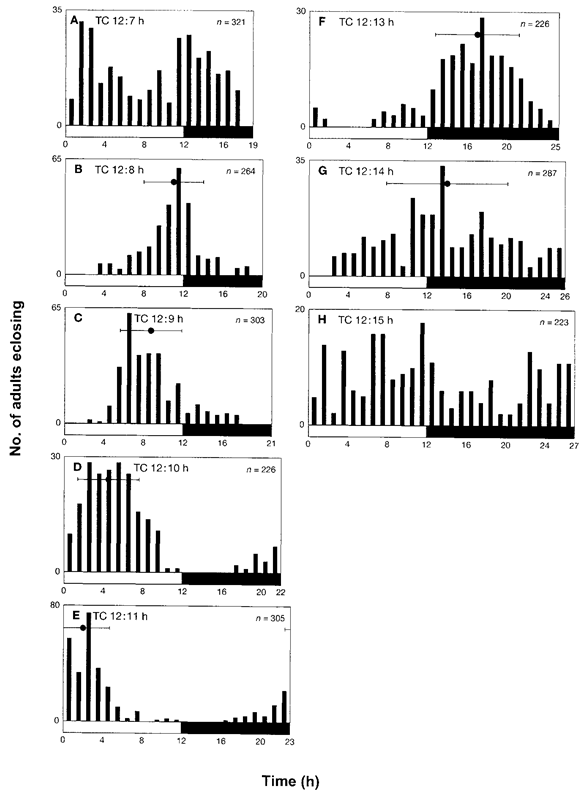
Figure 3. Adult eclosion of Plodia interpunctella under various non-24 h thermocycles of 25.5°C/24.5°C in DD. (A) TC 12:7 h, (B) TC 12:8 h, (C) TC 12:9 h, (D) TC 12:10h, (E) TC 12:11 h, (F) TC 12:13 h, (G) TC 12:14 h and (H) TC 12:15 h (refer Fig. 1). Note that the thermophase was held constant at 12 h.
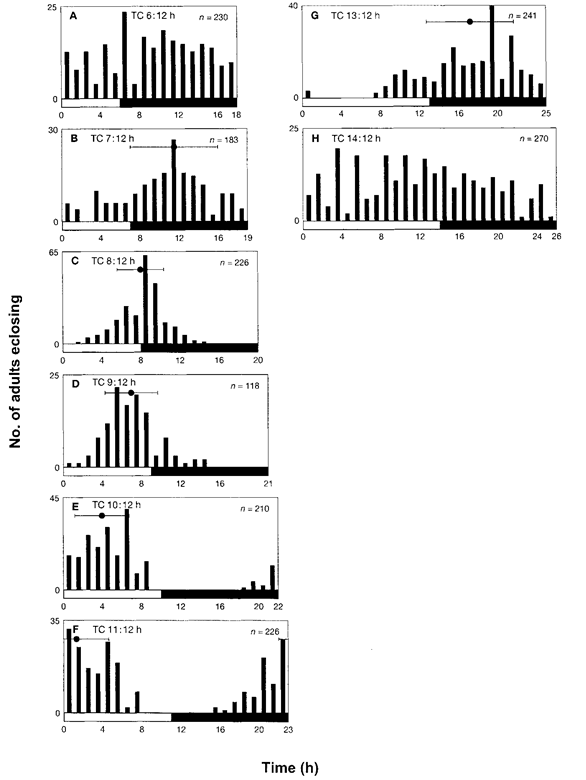
Figure 4. Adult eclosion of Plodia interpunctella under various non-24 h thermocycles of 25.5°C/24.5°C in DD. (A) TC 6:12 h, (B) TC 7:12, (C) TC 8:12 h, (D) TC 9:12 h, (E) TC 10:12 h, (F) TC 11:12 h, (G) TC 13:12 h and (H) TC 14:12 h (refer Fig. 1). Note that the cryophase was held constant at 12 h.
In the next series of experiments, the larvae were maintained under thermocycles consisting of various lengths of the thermophase and a constant 12-h cryophase. For TC 6:12 h (Fig. 4A), adults eclosed at random. For TC 7:12 h (Fig. 4B), TC 8:12 h (Fig. 4C), TC 9:12 h (Fig. 4D), TC 10:12 h (Fig. 4E) and TC 11:12 h (Fig. 4F), the average eclosion peaks were observed at Zt 11.5 ± 4.3, Zt 8.2 ± 2.3, Zt 6.9 ± 2.6, Zt 3.9 ± 2.7 and Zt 1.2 ± 3.2, respectively, after the rise in temperature. For TC 13:12 h (Fig. 4G), the peak occurred at Zt 17.3 ± 4.3, whereas for TC 14:12 h (Fig. 4H), no peak was observed. Moreover, in thermocycles with exposure longer than TC 15:12 h, there was no observable eclosion rhythm (data not shown). Thus, the time-keeping mechanism appeared to be entrained by thermocycles ranging from TC 7:12 h to TC 13:12 h, and the phase shift occurred in a linear fashion (see also Fig. 5B). The larvae experienced 17-25 cycles in thermocycles that were <24 h, and 15-19 cycles in thermocycles that were >24 h.
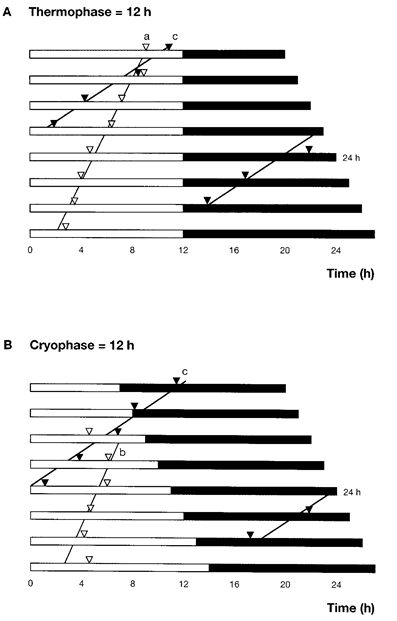
Figure 5. Temporal relationship between adult eclosion peak of Plodia interpunctella and non-24 h thermocycles. (A) A constant 12-h thermophase (white bar) was combined with various lengths of the cryophase (black bar). (B) A constant 12-h cryophase was combined with various length of the thermophase. ▽represents the adult eclosion peak for the 30°C/20°C thermocycle and ▼ represents the adult eclosion peak for the 25.5°C/24.5°C thermocycle. The correlation coefficients (r) of lines a, b and c were 0.9885**, 0.4639 and 0.9803**, respectively. Note that line c in (A) and (B) was exactly the same. The temporal position of the adult eclosion peak under the 24-h thermocycle was obtained from Kikukawa et al (2013).
4. Discussion
Figure 5 illustrates the phase relationships between the adult eclosion peaks of P. interpunctella under different thermoperiodic conditions. The line a in Figure 5A shows a phase shift of an adult eclosion peak under the thermocycle of 30°C/20°C with a constant 12-h thermophase. It is observed that as the cycle length extends, the peak advances, and the correlation coefficient (r) is 0.9885**. The peak occurs within the thermophase at about 17 h after the temperature-fall from 30°C to 20°C. When the cryophase (20°C) is a constant 12 h (Fig. 5B), the peak shown by line b occurs within the thermophase (30°C) but fluctuates around an average Zt 5.0 after the temperature-rise. The r of line b is 0.4639 which is not statistically significant. Thus, under these regimes, insects seem to use the temperature-rise as a time cue.
The temperature cycle of 25.5°C/24.5°C produces quite different results. Line c shown in Figure 5A and B is a phase shift of the peak, and r is 0.9803**. The peak is significantly advanced as the length of the temperature cycle is prolonged, regardless of the duration of the thermophase or cryophase. Thus, the temporal position of the peak seems to be a function of the cycle length. To use the cycle length as a cue, the insects must be able to measure either the time between the rise in temperature of a given cycle and the subsequent cycle or the time between the fall in temperature of two such cycles. In the thermocycle of 30°C/20°C, the peak occurs within the thermophase only. However, in the 25.5°C/24.5°C thermocycle, the peak occurs during the cryophase as well. Apparently, the amplitude of the temperature cycle is the key for determining the temporal position of the adult eclosion peak in P. interpunctella. As the insects never experience non-24 h thermocycles under natural conditions, the above phenomenon illustrates the basic physiological nature of the time-keeping system of P. interpunctella.
5. Conclusion
We revealed the entrainability of the adult eclosion rhythm of Plodia interpunctella under the non-24 h thermoperiodic conditions. Temperature-rise or –fall was observed to serve as a time cue (Zeitgeber). In the temperature cycles with 1°C difference, the temporal position of φE was determined as a function of the cycle length. Apparently, the time-keeping system functioned in a different manner, when considering the amplitude of the temperature cycle, which is a new finding especially in Plodia interpunctella.
References
- Kikukawa, S., Hashizume, R., Honda, M., Inoue, Y., Maekawa, T., Sakata, R., Takahashi, N., Tanaka, K. & Uchida, Y., 2013 Adult eclosion rhythm of the Indian meal moth Plodia interpunctella: response to various thermocycles with different means and amplitudes. Physiological Entomology, 38: 253-259.
- Aschoff, J., 1965 Circadian Clocks, North-Holland, Amsterdam.
- Beck, S.D., 1980 Insect Photoperiodism, 2nd edn. Academic Press, New York.
- Saunders, D.S., 2002 Insect Clocks, 3rd edn. Elsevier, The Netherlands.
- Refinetti, R., 2006 Circadian Physiology, 2nd edn. Taylor & Francis, London.
- Kikukawa, S., Hashizume, R., Honda, M., Inoue, Y., Maekawa, T., Sakata, R., Takahashi, N., Tanaka, K. & Uchida, Y., 2012 Effects of photoperiod and temperature on the rhythm and free-running of adult eclosion in the Indian meal moth Plodia interpunctella. Physiological Entomology, 37: 258-265.
- Arai, T., 1976 Effects of temperature and light-dark cycles on the diel rhythm of emergence in the Oriental fruit fly, Dacus dorsalis Hendel (Diptera: Trypetidae). Japanese Journal of Applied Entomology and Zoology, 20: 69-76. (In Japanese).
- Arai, T., 1998 Egg hatching rhythms of Eobiana engelhardti subtrolpica Bey-Bienko (Orthoptera: Tettigoniidae) under non-24 h thermoperiods. Entomological Science, 1: 495-501.
- Masaki, S. & Kikukawa, S., 1981 The diapause clock in a moth: response to temperature signals. Biological Clocks in Seasonal Reproductive Cycles (ed. by B.K. Follett), pp. 101-112. J. Wright & Sons, U.K.
- Kikukawa, S. & Masaki, S., 1984 Interacting effects of photophase and scotophase on the diapause response of the Indian meal moth, Plodia interpunctella. Journal of Insect Physiology, 30: 919-925.



