Use of Fruits and Raffia Palm Sap (Raffia hookeri) in Chilled Bull Semen Extenders in Cameroon
P. H. Bayemi1, *, N. R. Banla2, I. Leinyuy1, A. T. Niba2, V. M. Nsongka1, G. Mario3, M. Shamshuddin3
1Institute of Agricultural Research for Development, Yaounde, Cameroon
2University of Dschang, Faculty of Agronomy and Agricultural Sciences, Department of Animal Production, Dschang, Cameroon
3Animal Production and Health Section, International Atomic Energy Agency (IAEA), Vienna, Austria
A study was carried out to investigate the effect of substituting egg yolk in egg-yolk-citrate (EYC) extenders with extracts from Tomato and Pawpaw fruits as well as Coconut milk and Raffia palm sap on the motility of bull spermatozoa. Semen collected from Holstein Friesian bulls by the artificial vagina was extended with the proportions of egg yolk in egg-yolk-citrate extenders substituted at 25% (75% egg yolk: 25% Substituent), 50% (50% egg yolk:50% Substituent), 75% (25% egg yolk:75% Substituent) and 100% (no egg yolk in extender) and stored under refrigeration conditions (4-8oC). Results show that coconut milk (43.4%) and pawpaw juice (41.25%) led to the highest number of progressively motile sperms (P< 0.05) without evidence of difference between them (P > 0.05). They were followed by tomato Juice (40.35%), fresh raffia palm sap (40.40) and Trehalose (34.68%). Substitution of egg yolk at 75% had highest individual motility while total substitution (100%) had the lowest (P<0.0001). Meanwhile 50% and 75% substitution led to similar sperm progressive motility (P>0.05). Sperms retained 70% motility up to 15 days with coconut milk, 14 days with pawpaw juice, 14 days with fresh raffia sap, 13 days with tomato juice and 10 days with Trehalose. In the egg yolk treatment control, the drop below 70% was after the 4th day. Room temperature semen could be used only within 24H in all treatments. Fruits extenders performed better than egg yolk extenders, thus showing the beneficial effects of fruits in improving sperm motility. This is the first time such a result is reported. More studies need to be carried out on avoiding the sharp fall in motility 2 weeks after extension, on chemical concentrations of used fruits and in fertility of sperms with these extenders. In Cameroon where eggs are relatively more expensive, extenders may adequately be made with tropical fruits.
Keywords
Bull Semen, Egg Yolk, Fruits Extenders, Sperm Motility
Received: April 1, 2015
Accepted: May 2, 2015
Published online: May 28, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
There has been an ever increasing gap between the demand and supply of meat and milk in Cameroon despite a high number of animals standing at 5-6 million heads (MINEPIA, 2005; Pingpoh et al, 2007; MINRESI, 2007; Ndambi and Bayemi, 2008). Poor genetics of indigenous animals were identified as an important factor that limits productivity (Ebangi et al, 2004). Genetic improvement programmes by using artificial insemination (AI) are being implemented but their extensions are limited by high cost of imported semen and liquid nitrogen and short half-life of locally produced fresh semen (Bayemi et al, 2005 a,b). Also maintaining a cool chain with conventional fresh semen techniques has been challenging since one or two days are needed only to transport semen from the laboratory to the inseminators. There is therefore need to search for methods of increasing the productivity of indigenous cattle in meat and milk. This has led to the establishment of cattle production research units at Bambui and Wakwa centres of the Institute of Agricultural Research for Development (IRAD). Some of the interventions used to improve cattle production in Cameroon include crossbreeding of local females with exotic breeds and control of crossbreds to reduce the effects of undesirable traits (Ngwa, 2010). The introduction of Artificial Insemination (AI), which has been widely accepted as a technology that can bring about rapid genetic improvement in cattle and buffalos (IAEA, 2007), has been a major step ahead in attaining the objective of improving cattle production in Cameroon. It is imperative that farmers who will eventually use semen in AI be supplied with that of excellent quality as optimum conception rates will only be achieved if the quality of semen used is good.
The success of artificial insemination largely depends on the quality of the semen and proper AI practices (IAEA, 2007; Bayemi et al, 2010). However, this AI has been up to recently, dependent on the use of frozen semen in Cameroon, imported from developed countries, which has limited the extension of this technique to small scale farmers because of the very high cost of liquid nitrogen (about $US 20 per litre). This has led to the development of chilled semen techniques, using extenders like phosphate buffers, yolk phosphate extenders, citrate buffers, egg-yolk-citrate extenders, Tris buffers, buffered-yolk extenders (Ngwa, 2010). The chilled semen used at the IRAD research centre Bambui lasts for 5 to 7 days (Bayemi et al, 2010). This has gone a long way to reduce the cost involved in AI. However the length of sperm viability in storage is a big hindrance to the use of chilled semen technology. There is therefore the need to look for locally available, low cost technology that can be used to extend chilled semen for a longer duration.
The main objective of this study was to evaluate the substitution of egg yolk with fresh raffia palm sap, coconut milk, tomato juice, pawpaw juice, and Trehalose in citrate extenders of bull semen and their effect on sperm motility.
2. Materials and Methods
2.1. Procedure for the Preparation of Extenders
The extender for each treatment was prepared using two fresh eggs from indigenous hens. The egg yolk was separated from the albumen and cleaned by using a filter paper to remove the albumen. The egg yolk was then distributed into five test tubes for each treatment representing replacements as follows: 0% (1.250ml); 25% (0.9375ml); 50% (0.625); 75% (0.3125ml) and 100% (0.000ml) egg yolk substitution giving a total of 3.125ml egg yolk used. Citrate included in the five cited egg yolk replacements was as follows. 0% (0.294g); 25% (0.147g); 50% (0.147g); 75% (0.0.147g) and 100% (0.147g). The other constituents were similar for all treatments: Penicellin (0.00375g), streptomycin (0.00625), distilled water (5ml). Components in the Trehalose treatment were as follows: citrate (0.147g), Penicillin (0.00375g), Streptomycin (0.00625g), egg yolk (1.25ml), Trehalose (0.1875) and distilled water (5ml).The trisodium citrate dihydrate, Penicillin and streptomycin sulphate salts were each weighed using an electronic balanced and dissolved in 5ml of distilled water. Lincomycin HCl powder (antibiotic/fungicide; 375 mg) was dissolved in 50 ml of distilled water and homogenized. One millilitre of the prepared solution was added to each of the test tubes. This was in order to control yeast cells present especially in coconut milk and fresh raffia palm sap.
a. Coconut milk
Coconut milk was obtained from green premature Coconut fruits (Cocos nuciferal.) fruit. The water (termed milk) was extracted and stored in the freezer. Fifteen ml of the coconut milk was centrifuged at 3000 rpm for 15 minutes in order to sediment the debris in suspension. The centrifuged coconut milk was heated at 92 - 95°C for 10 minutes, then cooled to room temperature (18-22°C). Proportions of the coconut milk were distributed into test tubes containing egg yolk and the buffer solution in their appropriate proportions.
b. Fresh Raffia Palm sap
The raffia palm sap is the fluid obtained by tapping the inflorescence or succulent part of stem of raffia palm (Raffia hookeri). The primary purpose of tapping the sap of raffia palm is to obtain a whitish fluid which when left to stand for a few hours. Fresh palm sap is generally described as a 10 to 14% neutral sucrose solution (Okafor, 1978; Megwa, 1984) containing various nutrients usually expected in typical plant sap.
One litre of fresh raffia palm sap was collected from a wine taper every morning when the extender was to be prepared, and rushed to the laboratory (within 30 minutes). It was centrifuged at 1000 rpm for 5 minutes to sediment solid particles. The centrifuged fresh palm wine was then boiled for 10 minutes, cooled to room temperature (18-22 °C) and distributed into test tubes containing egg yolk and the buffer solution in their appropriate proportions.
c. Tomato Juice
Two ripped and healthy tomato fruits were washed with running tap water, wiped with alcohol, cut, mashed with a spoon in a dish and the juice was extracted. The juice was collected into centrifuge tubes and centrifuged at 3000 rpm for 15 minutes. The supernatant from the first centrifugation was centrifuged again at 3000 rpm for 15 minutes. The second supernatant was then collected into a 15 ml test tube and distributed into test tubes containing egg yolk and the buffer solution.
d. Pawpaw Juice (Carica papaya)
Pawpaw (Caricaceaes family) juice was obtained from mature ripped fruits. Ripped pawpaw was washed with running tap water and wiped with alcohol. A sterilized knife was used to dissect the pawpaw. The pawpaw seeds were removed and a clean sterile spoon was used to gently grate the soft tissue of the pawpaw fruit into a clean dish. The pulp was then transferred into test tubes and centrifuged at 3000 rpm for 15 minutes. The supernatant from the first centrifugation was centrifuged again at 3000 rpm for 15 minutes. The supernatant resulting from the second centrifugation was collected into a clean 15 ml tube and distributed into test tubes containing egg yolk and the buffer solution.
e. The control was the standard extender used at IRAD Bambui (Bayemi et al, 2010).
f. Extender with Trehalose
An extender was prepared just as the control and then to it, a proportion of Trehalose was added as a function of its volume. This addition was on the basis of 3g Trehalose in 80ml of distilled water (Hu et al, 2010).
2.2. Bull Preparation and Semen Collection
Bull preparation, semen collection and evaluation were done as described by Bayemi et al (2010). There were five ejaculates as replicates for each treatment. Each ejaculate was used in all treatments.
2.3. Post Extension Analysis
The extended semen for each treatment was divided into two equal volumes. One part was stored at Room temperature (18-25 oC) and in the dark and the other part was stored at refrigeration temperatures (4-8oC). Evaluation in storage continued for 26 days when the individual and progressive motilities (Bayemi et al, 2010) died down to zero. Semen was not thawed after extension before evaluation.
2.4. Statistical Analyses
The effects of treatments, level of replacement of egg yolk, replicate and time (days) were evaluated using the general linear model (GLM) procedure of Statistical Analysis Systems (SAS, 2002). The data was first adjusted using an angular transformation into arcsine (Ibrahim, 1998; Yadolah, 2008). Untransformed data have been reported in the results for clarity of interpretation. The Duncan Multiple Range test was used to separate means and level of significance was considered significant at P < 0.05.
3. Results
3.1. Chilled Semen
There was an overwhelming evidence that individual and progressive motilities of spermatozoa in Pawpaw juice were highest (P<0.0001). Meanwhile sperm motility in tomato juice, coconut milk, fresh palm sap were higher than trehalose treated semen (P<0.0001), which in turn had a better sperm motility than the control, that is semen stored in extender with egg yolk not substituted (Table 1: P<0.0001).
Substitution of egg yolk at 75% had highest individual motility while total substitution (100%) had the lowest (P<0.0001). Meanwhile 50% and 75% substitution led to similar sperm progressive motility (Table 2: P>0.05). This was observed in all fruits treatments (Tables 2 to 6).
Table 1. Effect of Tomato Juice, Coconut milk, Fresh Raffia Palm Sap, Pawpaw Juice and Trehalose on the individual and progressive motility of sperms.
| Treatment | Individual Motility | Progressive Motility |
| LS means (%) | LS means (%) | |
| Pawpaw Juice | 45a | 43a |
| Coconut Milk | 42b | 41ab |
| Tomato Juice | 42b | 40b |
| Fresh Raffia Palm Sap | 42b | 40b |
| Trehalose | 36c | 34c |
a,b,c: least square means with the same superscript in the same column do not significantly differ (p>0.0001).
Table 2. Substitution of Egg Yolk with Tomato Juice, Coconut Milk, Fresh Raffia Palm Sap, Pawpaw Juice and Trehalose.
| Level of substitution of egg yolk in the extender | LS means (%) | |
| Individual motility | Progressive motility | |
| 0% | 29d | 27c |
| 25% | 53c | 51b |
| 50% | 55b | 54a |
| 75% | 58a | 55a |
| 100% | 14e | 13d |
a,b,c,d: least square means with the same superscript in the same column do not significantly differ (p>0.0001).
Table 3. Effect of substitution of Egg Yolk with Tomato Juice in EYC extenders on IM and PM sperm cells.
| Level of replacement of Egg Yolk with Tomato Juice | LS means (%) | |
| Individual motility | Progressive motility | |
| Control,(EYC extender with 20%egg yolk) 0% tomato juice | 28.b | 27b |
| 25% tomato juice | 53a | 51a |
| 50% tomato juice | 55a | 53a |
| 75% tomato juice | 55a | 52a |
| 100% tomato juice | 20c | 19c |
Table 4. Effect of levels of replacement of Egg Yolk with Coconut Milk in EYC extenders on IM and PM sperm cells.
| Level replacement of egg yolk with coconut milk | LS means (%) | |
| Individual motility | Progressive motility | |
| Control, EYC extender with 20%egg yolk. (0% CM: 100 egg yolk) | 29c | 27b |
| 25% CM: 75% egg yolk | 57ab | 54a |
| 50% CM: 50% egg yolk | 55b | 54a |
| 75% CM: 25% egg yolk | 61a | 59a |
| 100% CM: 0% egg yolk | 14d | 12c |
Table 5. Effect of levels of replacement of Egg Yolk with Fresh Raffia Palm Sap in EYC extenders on IM and PM sperm cells.
| Level | LS means (%) | |
| individual motility | Progressive motility | |
| Control, EYC extender with 20%egg yolk. (0% FRPW: 100 egg yolk). | 29c | 27c |
| 25% FRPW: 75% egg yolk | 54b | 52b |
| 50% FRPW: 50% egg yolk | 57ab | 56ab |
| 75% FRPW: 25% egg yolk | 59a | 57a |
| 100% FRPW: 0% egg yolk | 12d | 11d |
All Means have the same standard error of ± 17.992 for individual motility and 18.01506 for progressive motilities
Table 6. Effect of levels of replacement of Egg Yolk with Pawpaw Juice in EYC extenders on IM and PM sperm cells.
| Level | LS means (%) | |
| individual motility | progressive motility | |
| Control EYC extender with 20%egg yolk. (0% PJ: 100 egg yolk) | 29b | 27b |
| 25% PJ: 75% egg yolk | 58a | 56a |
| 50% PJ: 50% egg yolk | 61a | 58a |
| 75% PJ: 25% egg yolk | 62a | 59a |
| 100% PJ: 0% egg yolk | 17c | 16c |
All Means have the same standard error of ± 18.080 for individual motility and 17.884 for progressive motilities.
Table 7. Effect of supplemented EYC extenders with Trehalose on IM and PM sperm cells.
| Level | LS means (%) | |
| Individual motility | Progressive motility | |
| Control, EYC extender with 20%egg yolk. | 29b | 27b |
| 0.187g of Trehalose added to EYC extender | 42a | 40a |
Generally individual and progressive motility decrease with time from the day of extension down to the end of the experiment. It was observed that individual and progressive motility were closely related and so progressive motility was considered because it is more important in artificial insemination (Figures 1 to 5).
Scores of motility with time shows that sperms retained 70% motility up to 13 days with tomato juice, 15 days with coconut milk, 14 days with fresh raffia sap, 14 days with pawpaw juice and 10 days with Trehalose. The egg yolk treatment control at 70% was at 2-4 days after extension.
3.2. Room Temperature Semen
Individual and progressive motility was found to rapidly fall in semen extended in EYC extenders kept at room temperature irrespective of the level of replacement of egg yolk proportions in the extender. However, semen extended in extenders in which egg yolk was completely replaced by either tomato juice, coconut milk, Fresh raffia palm sap, and pawpaw juice was still potentially fertile on the second day of extension (PM=40%), but motility died down very fast afterwards (Figure 6).
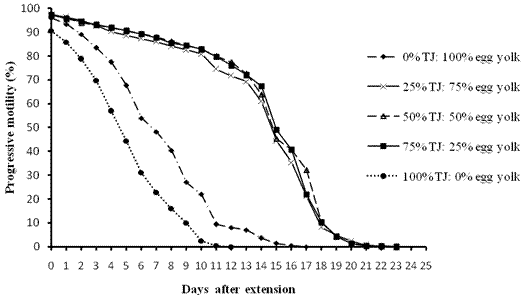
Figure 1. Replacement of Egg Yolk with Tomato Juice
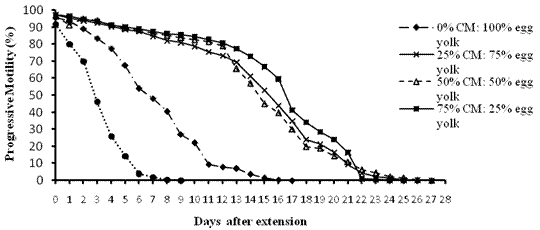
Figure 2. Replacement of Egg Yolk with Coconut Milk
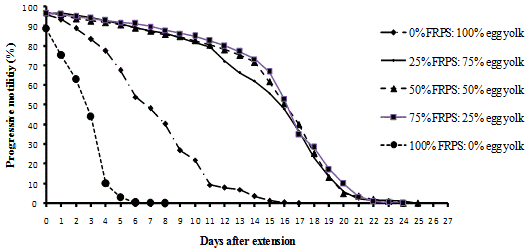
Figure 3. Replacement of Egg Yolk with Fresh Raffia Palm Sap
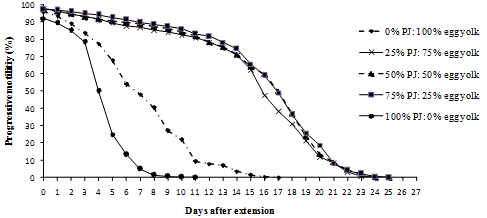
Figure 4. Replacement of Egg Yolk with Paw Paw Juice
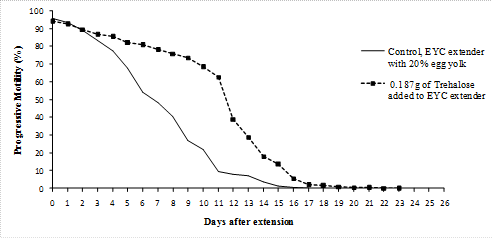
Figure 5. Addition of Trehalose to Egg-Yolk-Citrate extenders.
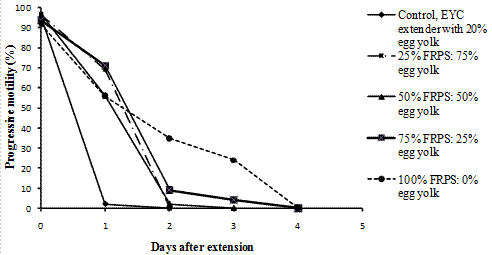
Figure 6. Extended semen with Fresh Raphia Palm Sap at room temperature.
4. Discussion
Room temperature semen only lasted here for 24 hours. Bouchard et al (1990) reported that canine semen could last for 4 days at room temperature. In Cameroonian conditions where cooling is rare in remote areas, room temperature semen could still be used a day after extension.
Results show that sperms still showed motility of 70% 15 days after extension in coconut milk. This semen could still be used 17 days after extension if the target of 50% motility is retained. Shamsuddin et al (1987) reported that the quality of chilled semen decreased with time, and remained suitable for use up to Day 2 as judged by motility and morphology. For Verstegen et al (2005), adding egg yolk to Tris-glucose extenders improved chilled canine semen to last for 7 days. They concluded that chilled canine spermatozoa could be preserved for 12 days, arguing that chilled dog semen could be preserved up to 21 days though in their case, the extender had to be exchanged when motility was below 50% but although an increase due to that exchange of extender occurred, a sharp fall was still noticed 2 days after, bringing motility to nearly the original level. If their opinion were considered, it could be said that motile sperms in this experiment lived over 21 days. It would still be however good to verify fertility of chilled semen used at 15 and 21 days after extension.
Munsi et al (2007) found that chilled bull semen could be preserved for 5 days and retained 50% progressive motility and that within that period sperm motility declined by 4% a day. Within the same period in this study sperm motility only declined by 1% per day. It is only 14 days after extension that it declined by 4%. They followed a sharp fall leading to the death of all sperm within the next 5 days (19 to 23days) depending on treatments. In this study, use of fruits extenders improved viability of potentially fertile sperms by up to 10 days. Extenders are supposed to protect spermatozoa, conserving motility and fertility over time by stabilizing the plasmalema, providing energy substrates and preventing deleterious effects of changes in pH and osmolarity (Brown, 1992; Goodman and Cain, 1993). The results of this study suggest that fruits can successfully be used as extenders in chilled bull semen with pawpaw juice and coconut milk leading to the best average motility during the 26-day experimental period.
It was surprising that fruits extenders performed better than egg yolk extenders, showing thus the beneficial effects of fruits in improving sperm motility. This is the first time such a result is reported. Crumley (2009) showed that many fruits contained gluthation (GSH), an antioxidant which extends sperm viability and motility (Munsi et al, 2007). GSH helps maintaining the integrity of normal acrosomes (Sinha et al, 1996). Moreover, GSH may stabilize the plasmolemma of spermatozoa and so increase motility. Foote et al (2002) suggested that spermatozoa are weak in antioxidants and they can readily undergo lipoperoxidation, particularly in the presence of oxygen. Addition of antioxidants might have beneficial effects on sperm. It remains to be proven that fruits provide these antioxidants.
Verstegen et al (2005) also found that glucose improved semen quality and viability compared to fructose. The mechanisms by which the prolongation of motility characteristics is obtained are probably related to glucose supplementation. Fruits are also rich in glucose and this may have contributed to their good effect. These authors also stated that, egg yolk had a beneficial effect on sperm motility because of the energy substrate that it provides as well as the addition of phospholipids. But in this study, fruits extenders performed better. This suggests that supply of energy in chilled bull semen extenders might well be more important than addition of phospholipids. Though McGinnis et al (2005) improved preservation of desiccated mouse sperm using Trehalose, it did not improve sperm motility in the present study compared to fruits extenders. This comes further to stress on the need of readily available sugars in preserving sperm motility. During the recent years, the use of chilled semen appeared as an interesting alternative to freezing for short- to medium-term semen conservation, particularly where the high costs of materials (e.g., nitrogen containers) and complex custom/shipment procedures can be avoided (Iguer-Ouada and Verstegen, 2001).
5. Conclusion
In this study, fruits extenders performed better with regards to motility than egg yolk extenders, thus showing the beneficial effects of fruits in improving sperm motility. This is the first time such a result is reported. In Cameroon where eggs are relatively more expensive, extenders may adequately be made with tropical fruits. More studies need to be carried out on avoiding the sharp fall in motility 2 weeks after extension and chemical concentrations of used fruits. However, fertility studies will need to be done for extenders with tropical fruits.
Acknowledgements
This work was carried out thanks to the financial support of the International Atomic Energy Agency (IAEA).
References
- Bayemi, P.H., I. Leinyuy V.M. Nsongka, E.C. Webb & A.L. Ebangi, 2010. Viability of cattle sperm under different storage conditions in Cameroon. Journal of Tropical Animal Health Production. 42:1779–1783
- Bayemi, P.H., Bryant, M.J., Perera, B.M.A.O., Mbanya, N.J., Cavestany, D., Webb, E.C., 2005a. Milk production in Cameroon: a review. Livestock research for rural development 17, 6.www.cipav.org.co/lrrd/lrrd17/6/cont1706.
- Bayemi, P.H., Bryant,M.J., Pingpoh, D., Imele, H., Mbanya, J., Tanya,V., Cavestany, D., Awoh, J., Ngoucheme, A., Sali, D., Ekoue, F., Njakoi, H.,Webb, E.C., 2005b. Participatory rural appraisal of dairy farms in the northwest province of Cameroon. Livestock research for rural development 17, 6.www.cipav.org.co/lrrd/lrrd17/6/cont1706.
- Bouchard G.F., Morris J.K., Sikes J.D., Youngquist R.S., 1990. Effects of storage temperature, cooling rates and two different semen extenders on canine spermatozoal motility. Theriogenology; 34:147–57.
- Brown R.M., 1992. An update of artificial insemination with fresh, chilled, and frozen semen. Probl Vet Med; 4:445–52.
- Crumley H., 2009. What Foods Increase Glutathione Levels? The Journal of Nutrition, May 2004, 134, 1114-1119.http://www.livestrong.com/article/32331-foods-increase-glutathione-levels/
- Ebangi, A.L.,Tawah, C.L., Mbah, D.A., Tanya, V.N.,Ndi, C., Ekue,F.N., Messine, O., Ottou, J.B.B., Enoh., M.B., 2004. Mortalities amongst different genotypes of crossbred dairy calves in a tropical environment. Journal of the Cameroon Academy of Science vol. 1, No. 2: 117–124.
- Foote R.H., Brockett C.C., Kaproth M.T., 2002. Motility and fertility of bull sperm in whole milk extender containing antioxidants. Animal Reproduction Science 71: 13–23.
- Goodman M.F., Cain J.L., 1993. Retrospective evaluation of artificial insemination with chilled extended semen in the dog. Journal of Reproduction Fertility; 47(Suppl.):554.
- Hu, J.-H., Zan L.S., Zhao X.L., Li Q.W., Jiang Z.L., Li Y.K., and Li X. 2010. Effects of trehalose supplementation on semen quality and oxidative stress variables in frozen-thawed bovine semen. Journal of Animal Science, 88:1657-1662.
- IAEA, 2007. Improving the Reproductive Management of Dairy Cattle Subjected to Artificial Insemination. Vienna, Austria: IAEA. 148pp.
- Ibrahim H. 1998. Small Ruminant Production Techniques. ILRI Manual 3. ILRI (International Livestock Research Institute), Nairobi, Kenya. 207 pp.
- Iguer-Ouada M., Verstegen J.P., 2001. Long-term preservation of chilled canine semen: effect of commercial and laboratory prepared extenders. Theriogenology, 14: 671-684.
- McGinnis L.K., Zhu L., Lawitts J.A., Bhowmick S., Toner M., Biggers J.D., 2005. Mouse sperm desiccated and stored in trehalose 3 medium without freezing. Biological Reproduction. 73(4):627-33
- Megwa S. 1984. Aspects of the preparation of formulated palm wine. M.Sc. Dissertation. University of Nigeria, Nsukka.
- Minepia, Ministre de l'Elevage, des Pêches et des Industries Animales, 2005. Evolution et Situation Actuelle de l’Elevage au Cameroun. Rapport de synthèse.41pp
- Minresi, Ministère de la recherché Scientifique et de l’Innovation, 2007. Journées d’excellence de la recherche Scientifique et de l’Innovation au Cameroun (JERSIC).Prix spécial du président de la république, (2007) 3-5 juillet.
- Munsi, M.N., Bhuiyan M.M.U., Majumder S., and Alam M.G.S., 2007. Effects of Exogenous Glutathione on the Quality of Chilled Bull Semen. Reprod Dom Anim 42: 358–362.
- Ndambi, A. and Bayemi, P.H., 2008. Cameroon-dairy sector and chain profile. In. Hemme et al. IFCN Dairy Report 2008. International Farm Comparison Network, IFCN Dairy Research Center, Kiel, Germany
- Ngwa, Tarla Divine, 2010. Effect of egg yolk concentrations, cooling methods and ph on chilled bovine semen characteristics. Dissertation, FASA, University of Dschang, Cameroon.
- Okafor N., 1978. Microbiology and biochemistry of oil palm wine. NAIFOR Newsletter 12: 8- 6.
- Pingpoh, D.P., Mbanya, J., Ntam, F., Malaa, D.K., 2007. Some risk management practiced among the beef cattle farmers of the North West Province of Cameroon: Effect on technology dissemination. 8pp.
- SAS/STAT, 2002, Statistical Analyses System (8th ed., Volume 2). Cary NC: SAS Institute Inc.
- Shamsuddin M., Ahmed J.U., Alam MGS, Modak P.C., 1987: Effect of age of semen on conception rate in cattle under farm condition. Bang Veterinary Journal 21: 51–58.
- Sinha M.P., Sinha A.K., Prasad R.L., 1996: The effect of glutathione on the motility, enzyme leakage and fertility of frozen goat semen. Animal Reproduction Science 41: 237–243.
- Verstegen, J.P., Onclin K., Iguer-Ouada, M., 2005. Long-term motility and fertility conservation of chilled canine semen using egg yolk added Tris–glucose extender: in vitro and in vivo studies. Theriogenology 64: 720–733.
- Yadolah Dodge, 2008. The Concise Encyclopedia of Statistics. University of Neuchâtel, Switzerland. Springer reference, 612pp.



