Root and Shoot Growth of Dragon Fruit (Hylocereus undatus) Stem Cutting as Influenced by Indole Butytic Acid
Thayamini H. Seran*, A. Thiresh
Department of Crop Science, Faculty of Agriculture. Eastern University, Chenkalady, Sri Lanka
Abstract
This study was done to determine the most suitable concentration of IBA for better establishment of stem cuttings of dragon fruit (Hylocereus undatus). Therefore, the stem cuttings were dipped in various concentrations of IBA and then planted in the polythene bags containing sand: top soil: cow dung at a rate of 2:1:1. This experiment was designed in Completely Randomized Design with three replicates. The treatments included 2000, 4000, 6000, 8000 ppm concentrations of IBA and a control. The results revealed that cuttings treated with 8000 ppm IBA gave the best root growth and it was not significantly different from the higher concentrations of IBA (6000 ppm). At 60 days after planting of cuttings, shoot length (9.5 cm), shoot fresh (10.25 g) and dry (0.59 g) weights were recorded higher in cuttings dipped in 6000 ppm concentration of IBA. The results suggest that IBA at 6000 ppm concentration is better for the establishment of the dragon fruit cuttings.
Keywords
Dragon Fruit, IBA, Rooting, Stem Cutting, Shoot Growth, Vegetative Propagation
Received: February 28, 2015
Accepted: March 13, 2015
Published online: March 20, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Dragon fruit is a member of the family Cactaceae and perennial, climbing cactus with triangular green stem. Aerial roots grow from the base of the stems, providing anchorage for the plants (Zee et al., 2004). The commonly cultivated vine cactus is Hylocereus undatus (Haw.) and it is a fruit crop as well as an ornamental plant. Dragon fruit is valuable for treating different types of diseases and also has significant economic value (Suryono, 2006). It is usually propagated by seeds or cuttings. Seed propagation method is very simple but seeds are not true to type due to cross pollination and also seeds are to be stored for about 28 days without losing viability (Andrade et al., 2005). Therefore large number of plantlets with healthy shoot and root system can be produced to meet the demand of increasing commercial cultivation through vegetative propagation methods.
Dragon fruit is propagated by stem cuttings which may be planted directly in the field or in pots (Zee et al., 2004). Cuttings are dipped in rooting hormone before planting to boost root formation. In many plant species, vegetative propagation is normally practiced using stem cuttings (Abdullah et al., 2005; Henrique et al., 2006). In some species, rooting occurs in cuttings without auxin treatment but in others cuttings do not root easily (Hartmann et al., 2002; Blythe et al., 2004). There is a possibility for expansion of dragon fruit cultivation in Sri Lanka. This experiment was carried out to study the effect of indole butytic acid (IBA) on establishment of stem cuttings and to select the optimum concentration of IBA for better growth of stem cuttings of dragon fruit.
2. Materials and Methods
This study was conducted in a net house at the Eastern University of Sri Lanka and this was set in a Complete Randomized Design with three replicates for each treatment. The treatments were different concentrations (0-8000 ppm) of IBA. Stem cuttings (10 cm long) were collected early in the morning and the basal part (about 1 inch) of the cuttings was dipped into different concentrations of IBA for 15 sec. In control treatment, cuttings were immersed in distilled water. Top soil, sand, cow dung at the rate of 1:2:1 were mixed thoroughly and soil mixture was filled in each polythene bag (12 cm long and 6 cm width) upto three fourth of its length. After soaking the cuttings, they were placed into soil mixture and kept in a net house. Water was sprayed over the cuttings daily morning and evening. This experiment was repeated twice.
Growth of cuttings is usually measured in terms of increase in dry weight and height. Such measurement indicates that growth rate varies with the age of the cuttings. Number and length of roots as well as fresh and dry weights of root and shoot were recorded at 60 days after planting of cuttings. The collected data were analyzed using Statistical Analyzing System (SAS). The treatment means were compared using Duncan’s Multiple Range Test (DMRT) at 5% significant level.
3. Results and Discussion
3.1. Number of Roots
Table 1. Effect of IBA concentrations on number of roots and length of longest root & shoot per cutting at 60 days after planting.
| IBA (ppm) | Root numbers per cutting | Longest root length(cm) | Shoot length(cm) |
| 0 (control) | 7.0 ± 1.0c | 11.0 ± 1.5c | 4.3 ± 1.0c |
| 2000 | 9.7 ± 2.7bc | 14.3 ± 0.9bc | 4.6 ± 1.4c |
| 4000 | 11.0 ± 2.5bc | 16.0 ± 1.1b | 7.3 ± 1.5ab |
| 6000 | 15.3 ± 1.4ab | 21.3 ± 2.0a | 9.5 ± 1.3a |
| 8000 | 19.3 ± 3.3a | 22.0 ± 0.6a | 6.8 ± 1.2b |
| F test | * | * | * |
Values are means± standard error of three replicates. Means followed by the same letter are not significantly different from each other at 5%significant level according to the Duncan’s Multiple Range Test. F test: *: P <0.05.
The result showed that different treatments had remarkable effect (P<0.05) on the average number of roots per cutting (Table 1). IBA concentration at 8000 ppm significantly differed from other treatments except 6000 ppm IBA. The degree of root formation per cutting steadily increases with increase concentration of IBA among tested treatments. It may be due to the presence of exogenous auxin treated cuttings. Fathi and Ismailpor (2000) stated that there is a correlation between primordia division in root initiation and endogenous or exogenous auxin. The roots are vital organ for anchorage and absorption of water and minerals from the soil for growth of cuttings. This finding is supported by Fathi and Ismailpor (2000) who reported that auxin enhances the root formation in cuttings. In Ginkgo biloba, the highest percentage of rooted cuttings was noted in 4000 ppm IBA (Barzegaret al., 2004).
The maximum average number of roots (19.3) per cutting was observed in 8000 ppm IBA and minimum average number of roots (7.0) was in absence of IBA i.e. control. Application of the different concentrations of IBA may direct to vary in number of roots per cutting. Hashemabadi and Sedaghathoor (2005) indicated that 4000 ppm IBA was the most suitable concentration to boost rooting of cuttings in Camellia japonica while Reddy et al. (2005) mentioned that maximum number of roots was recorded with 2000 ppm IBA in cuttings of scented geranium. The results obtained with IBA were in conformity with several researchers but effective concentration of auxin for increased rooting varies with plants.
3.2. Longest Root Length
The average length of longest roots per cutting was significantly (P<0.05) varied among the treatments (Table 1). The maximum average length of longest root was attained in 8000 ppm IBA without statistical difference with the 6000 ppm IBA while the shortest average length of longest root was in the control (Table 1 and Plate 1). The average value of longest root length ranged from 11.0 cm to 22.0 cm. It is supported by Reddy et al. (2005) who minimum Length of longest root was observed with control in scented geranium cuttings and maximum with IBA at 2000 ppm.

Plate 1. Ex vitro rooting of dragon fruit cuttings at 60 days after planting.
[1 - Control treatment; 2 - Cutting treated in 2000 ppm; 3 - Cutting treated in 4000 ppm; 4 - Cutting treated in 6000 ppm; 3 - Cutting treated in 8000 ppm]
The average length of longest roots increased gradually with increase in IBA concentration. Difference in length of longest roots between treatments may be due to application of IBA that may initiate hydrolysis of nutritional reserves in cuttings to increase root length. Bajwaet al. (1977) found treatment with IBA at a concentration of 100 ppm not only induced more roots per cutting but also the length of the roots. Sharma et al. (2002) observed the highest length of root per cutting in Gradenia lucida when treated with 2000 ppm IBA.

Plate 2. Shoot length of dragon fruit cuttings at 60 days after planting.
[A- Shoot length of cuttings in control (1) and 6000 ppm (2); B- Shoot length of cuttings in control (1) and 8000 ppm (2)]
3.3. Shoot Length
There was a significant difference (P<0.05) in average length of shoot among the treatments (Table 1). Highest length (9.5 cm) of shoots was recorded in cuttings dipped in 6000 ppm concentration of IBA and lowest length (4.3 cm) was in the absence of IBA i.e. control (Table 1 and Plate 2).All IBA treatments gave better shoot growth compared to control. Reddy et al. (2005) stated that auxin cause increased linear growth of stem due to cell elongation and maximum shoot length was recorded with the application of auxin (IBA, NAA) at 2000-2500 ppm in scented geranium cuttings.
3.4. Fresh Weight of Shoots
The fresh weight of shoots showed remarkable variations (P<0.05) between the treatments (Figure 1). The mean fresh weight of shoots ranged from 2.68 g to 10.25 g. Highest fresh weight (10.25 g) of shoots was recorded in cuttings dipped in 6000 ppm concentration of IBA which was significantly differed from other treatments except 4000 ppm concentration.
3.5. Fresh Weight of Roots
Statistical analysis of data on fresh weight of roots indicated significant differences (P<0.05) between the treatments (Figure 1). The mean fresh weight of roots ranged from 0.55 g to 2.96 g. The root weight was not significantly differed by treating cuttings with 2000, 4000 and 6000 ppm IBA. The increase in root weight is related to number of roots. Higher fresh weight (2.96 g) of roots was recorded in cuttings dipped in 8000 ppm concentration of IBA which was remarkably varied from other treatments. All IBA treatments gave better results in root formation compared to control. IBA enhanced the number of roots as a result the roots absorbed more nutrients from soil consequently it leads to better root growth. These results were confirmed by Sharma et al. (2002) who recorded the highest fresh weight of roots per cuttings of Gardenia lucida when treated with auxin. IBA stimulates root growth (Shofiana et al., 2013).
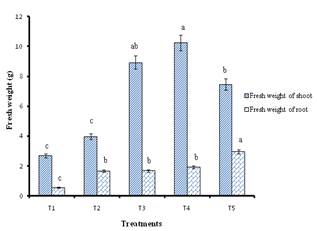
Figure 1. Fresh weight of shoot and root of cutting in different treatments at 60 days after planting of cuttings.
3.6.Dry Weight of Shoots
The result recorded on dry weight of shoots clearly showed significant variations (P<0.05) among the treatments (Table 2). Mean dry weight of shoots ranged from 0.15 g to 0.59 g. IBA concentration at 6000 ppm significantly differed from other treatment expect 4000 ppm concentration. The maximum dry weight of shoots (0.59 g) was recorded in cutting dipped in 6000 ppm concentration of IBA and minimum dry weight of shoots (0.15 g) was observed in the absence of IBA i.e. control. Auxin (NAA) enhanced development of shoot initials and their further development in rose cutting (Rahmat et al., 2007).
Table 2. Effect of IBA concentrations on dry weight of shoots per cutting at 60 days after planting
| Treatments | Dry weight of shoot (g) | Dry weight of roots (g) |
| 0 | 0.15± 0.00c | 0.14 ± 0.01d |
| 2000 | 0.17± 0.03c | 0.29 ± 0.02c |
| 4000 | 0.47± 0.02ab | 0.35 ± 0.01c |
| 6000 | 0.59± 0.05a | 0.52 ± 0.02b |
| 8000 | 0.36± 0.01b | 0.90 ± 0.01a |
| F value | ** | ** |
Values are means± standard error of three replicates. Means followed by the same letter are not significantly different from each other at 5% significant level according to the Duncan’s Multiple Range Test. F test: **: P <0.01.
3.7.Dry Weight of Roots
The dry weight of roots clearly indicated significant variation (P<0.05) between the treatments (Table 2). The maximum mean dry weight (0.90 g) of roots was recorded in cuttings dipped in 8000 ppm concentration of IBA which was significantly higher than the other treatments. Control treatment gave the lowest mean dry weight (0.14 g) per rooted cutting. Dry weight of root increased with increase of concentration of auxin. This result is supported by Reddy et al. (2005) who recorded highest dry weight of root with the application of auxin. Cutting treated with IBA had better rooting of Paeonia cutting (Xian et al., 2008) and application of 2500 ppm IAA improve the root growth of lemon stem cutting (Seran and Umadevi, 2011).
4. Conclusion
In this study, root formation in dragon fruit stem cuttings was significantly influenced by the application of IBA. IBA treated cuttings gave better root system compared to control in recorded agronomic parameters. Cuttings treated with 8000 ppm IBA produced the best results in root formation without being significantly differed from cuttings treated with 6000 ppm IBA. However, the highest shoot formation was obtained by 6000 ppm IBA treatment but it was remarkably varied from other treatments except with 4000 ppm IBA. In most cases, cuttings treated with 6000 ppm IBA gave the best results or produced results which are not significantly different from the higher concentrations of IBA. As a result, 6000 ppm IBA concentration would be better in rooting and shoot formation of the cuttings of dragon fruit plants for better plant growth in vegetative propagation.
References
- Abdullah, A.T.M., Hossain, M.A. and Bhuiyan, M.K. (2005). Propagation of Laktan (Baccaurea sapidaMuel.Arg.) by mature stem cutting. Research Journal of Agriculture and Biological Science 1(2): 129–134.
- Andrade, R.A.de., Oliveira, I.V.de.M. and Martins, A.B.G. (2005). Influence of condition and storage period in germination of red pitaya seeds. Revista-Brasileira-de-Fruticultura 27(1): 168-170.
- Bajwa, G.S., Gurcharan singh, Sandhu, A.S and khajurai, H.N. (1977). Rooting of sweet lime (Citrus limettiodes Tanaka) cutting as affected by the type of cut and IBA concentration. Haryana Journal of Horticultural Science 6: 115-116.
- Barzegar Torghabe, L., Hamidaughli, Y., Hatamzadeh, A. and Haddad, A. (2004). Effect of media and different concentration of IBA on rooting of Ginkgo Biloba cuttings. MS thesis. University of Guilan, Gilan, Iran.
- Blythe, E. K.; J. L. Sibley; J. M. Ruter and K. M. Tilt (2004). Cutting propagation of foliage crops using a foliar application of auxin. Scientia Horticulturea 103(1): 31-37.
- Fathi, G. and Ismailpor, B. (2000). Plant growth regulators. Jihad-e-Daneshgahi Publication, Mashhad, Iran.
- Hartmann, H.T., Hudson, T., Kester, D.E., Dale E. K., Davies Jr., F.T. and Geneve, R. L. (2002). Hartmann and Kester’s plant propagation: principles and practices. 7th ed. Prentice-Hall. London.
- Hashemabadi, D. and Sedaghathoor, S. (2005). Effects of synthetic auxin (IBA and NAA) on rooting cuttings of ornamental Camellia. 4th Iranian Horticultural Science Congress, Iran.
- Henrique, A., Campinhos, E.N., Ono, E.O. and Pinho, Z. (2006). Effect of plant growth regulators in the rooting of Pinus cuttings. Brazilian Archives of Biology and Technology 49(2): 189–196.
- Rahmat, U. K., Sohail Khan, M., Rashid, A., and Farooq., M.A., (2007). Effect of Exogenous Indole-3-acetic Acid and Naphthalene Acetic Acid on Regeneration of Damask Rose Cuttings in Three Growing Media. Pakistan Journal of Biological Sciences 10: 3626-3631.
- Reddy, C.H., Chandra sekhar, R., Reddy, Y.N. and Rajkumar, M. (2005). Effect of growth regulators on rooting of Scented geranium (Pelargonium graveolens (L.) cuttings. Journal of Research ANGARU 33(4): 114-116.
- Sen, P.K., Sen, S. and Basu, R.N. (1965). Effect of auxin organic nutrients on rooting of cuttings of Justicia gendarussa L taken from stock plants growth under various levels of nitrogen. Indian Journal of Plant Physiology 836-849.
- Sharma, A.K., Ashutoshmishra., Trivedi, O.N. and Shukla, P. K., (2002). Effect of IAA and IBA on gardenia cuttings. Journal of Ornamental Horticulture New Series 5: 71.
- Shofiana, A., Sri Rahayu, Y. and Budipramana, L.B. (2013).Pengaruh Pemberian Berbagai Konsentrasi Hormon IBA (Indole Butyric Acid) terhadap Pertumbuhan Akar pada Stek Batang Tanaman Buah Naga (Hylocereus undatus). LenteraBio 2(1):101–105.
- Suryono, J. (2006). Consuming dragon fruit to treat various diseases. Indonesia Sinar Tani 15-21.
- Thayamini H. Seran and Umadevi T. (2011). Influence of indole acetic acid (IAA) on the establishment of stem cuttings in Lemon (Citrus limon L.). Journal of Agricultural Research 49(4): 517-524.
- Xian, F.G., Xiling, F., Dekui, Z. and Yan, M. (2008). Effect of auxin treatments, cuttings collection date and initial characteristics on Paeonia ‘Yang Fei Chu Yu’ cutting propagation. Scientia Horticulturea 119: 177-181.
- Zee, F., Yen, C.R. and Nishina, M. (2004). Pitaya (Dragon fruit, Strawberry pear). Fruit and Nuts 9. Cooperative Extension Service, College of Tropical Agriculture and Human Resources, University of Hawaii at Manoa, Honolulu, Hawaii.



