Aloe Vera Used for the Protection of Earthworm
Rajesh Kumar Singh*
Department of Chemistry, Jagdam College, J P University, Chapra, India
Abstract
Earthworm is life of soils. It regulates soils productivity, humidity and concentration of water, oxygen, minerals, temperature and pH. The life of earthworm is getting destroyed by application of artificial insecticides, pesticides, herbicides, fertilizers, various types of wastes and acid rain. These killer substances change physical, chemical and biological properties of soils and generate corrosive atmosphere for earthworm. These pollutants change the concentration of natural minerals which are present in soils by increasing the acidic character of soils. The acidic soils develop microbiological corrosion cell with earthworm and it oxidize carbohydrate, protein and fat into CO2, H2O, NH3, glycerol and organic acid. Corrosion reaction increases the temperatures of soils and finally kill earthworm. Heavy metals and gaseous pollutants come into soil by different medium which produce corrosive effect for earthworm. Pyrite ores of industrial area enter into soil to form acid that acid is corroding earthworm. These foreign materials contaminate soil and reduce the fertility of soil. Aloe Vera juice is applied to control the pH values of soils and protect the life of earthworm. The experimental work shows that Aloe Vera has capability to control the physical, chemical and biological parameters of soils and it also enhances the fertility and productivity of soil and protects the life of earthworm by corrosive pollutants. For this work soil samples are taken from the coal area of Jhariya district (in Dhanbad), steel plant area Chas in Bokaro district, urban area of Chak Beyriya in district of Patna and village area of Fulwariy-Tajpur in district of Chapra.
Keywords
Soils, Earthworm, Biological Corrosion Cell, Pollutants, pH, Aloe Vera
Received: February 16, 2015
Accepted: March 7, 2015
Published online: March 12, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
Earthworms are very important components of soils because they can regulate fertility of soils [1], its humidity [2] and pH [3]. But these organisms are destroyed [4] by interaction of industrial‘s pollutants, effluents, flues, hazardous wastes, municipal wastes, households wastes, hospital wastes, artificial fertilizers, pesticides, insecticides, herbicides, rodenticides, particulates, corrosive gases, acid rain and global warming. These substances are mostly acidic and basic in character and they can form a microbiological corrosion cell [5] with earthworms’ thus biological corrosion reaction starts with these species and in this way their morphology can be changed which leads to the destruction of these organism in soils [6]. These harmful substances change physical, chemical and biological properties of soils. They can disturb pH values and minerals composition of soils [7].
The major sources of corrosive pollutants, effluents, flues and hazardous wastes are chemical industry, acid manufacture, sugar, coal mines, washery, coke manufacture, distillery, electroplating, paint manufacture, petroleum refinery, plastic manufacture, pulp and paper industry, steel industry, tannery, textile processing, electronic equipment, city waste, fertilizer industry, pesticides and herbicides industry, mining and ore processing, metallurgy, chemical industry, alloys, leather, electrical power plant, nuclear reactor, soap and detergent industry, synthetic rubber, medicines, cosmetics, adhesives, explosives, salts, food processing, automobile industry, bricks making industry, rice and flour mills, glass and ceramic industry, cement industry which are contaminating soils.
The above mentioned industries release directly and indirectly harmful hazardous wastes into ponds, lakes, canals, rivers, sea and ocean and these pollutants can contaminate soils [8], atmosphere [9] and water sources [10]. Ponds, canals, rivers and ground water are used for irrigation of soils and this polluted water sources create corrosive environment for earthworms [11].
The inorganic pollutants are oxide of carbon, oxide of nitrogen, oxide of sulphur, oxide of halogen, hydride of sulphur, hydride of halogen, ammonia and organic pollutants are aldehyde (formaldehyde, acetaldehyde) and ketone (acetone), carboxylic acid (formic acid and acetic acid), pyridine, alcohol (methyl alcohol, ethyl alcohol), thiol (methylthiol, ethylthiol), methyl isocyanide, amines (methylamine and ethylamine) released by various industrial and nonindustrial sources. The concentrations of these pollutants are increased into atmosphere and they absorb moisture to convert into acids [12]. These acids convert into cloud and come into soils by rain. These acids produce not only bad effects on soils and earthworms but also change the pH value of soils.
2. Objective
This work focused on protection of earthworm and maintained the fertility of soil and their important components. The soil toxicity has controlled by application of Aloe Vera. It can also save the life of earthworm.
Acid rain can play important role for destruction of soils’ organisms [13] and adversely effect pH of soils [14]. Soils possess natural minerals like Na, K, Fe, Cu, Zn, P, N2, O2 and the composition of these minerals are reduced by acid rain. The concentration of CO2, CH4 and water vapor are increased in atmosphere so the earth’s temperature is raised and it creates problems for the survival of soils organisms.
Particulates come into atmosphere due to large scale industrialization, urbanization, deforestation, agriculturalization, infrastructure development works, mining and minerals processing works, stones breaking works, constructions works, making of railway tracks etc. These particulates contain heavy metals Fe, Cr, Co, Ni, Cu, Zn, Mo, As, Pb, Bi, and soot’s of carbon, sulphur and fly ash. These metals are deposited on the surface of different components of environment and enter into soils directly and indirectly through various sources. These heavy metals contaminate soil and alter soils physical, chemical and biological properties. They also develop hostile environment for soil organisms [15] and change its pH values.
3. Methodology
Collecting soils of different region like mining area (Jhariya in dist. of Dhanbad), industrial area (Chas in dist. of Bokaro), urban area (Chak Beyriya in dist of Patna) and village area (Fulwariy-Tajpur in dist of Chapra) and examine their pH values, concentration of minerals and oxygen and temperature. Toxicity of soils studied above mention areas. Earthworms are dispersed in different types of soils in the absence and presence of Aloe vera and their physical, chemical and biological activities were observed at different concentrations of Aloe Vera, temperatures and times. The pH values of soils are recorded in different areas absence and presence of Aloe Vera at intervals of time. Earthworm’s biological corrosion activities are calculated at 24hrs. 48hrs, 72hrs, 96hrs and 120hrs.
4. Results
Analysis of pH values of soils in different regions and their values are mentioned in Table1. It is observed that mining area soils are more acidic with respect of other areas. The number of earthworms aided in various types of pH values of soils are counted at different intervals of time and the results are depicted in table1 and figure2. Results of table1 and figure2 indicated that mining area soil killed more earthworms. The acidic soils developed bio-electrochemical cell with earthworms and oxidized its amino acids and carbohydrates into ammonia, water and carbon dioxide. Fat dissociated in form glycerol and organic acids in presences of acids which oxidized into alkane and carbon dioxide. The bio-electrochemical cell occurs with earthworm and its mechanism as it is shown in figure1.
Anodic reaction with amnioacids
NH2CH2COOH + 2 H2O/H+ → 2CO2 + NH3 + 6H+ + 4e
Cathodic reaction
2H+ + 2e → H2
Anodic reaction with carbohydrate
C6H12O6 + 6H2O/H+ → 6CO2 + 24H+ + 24e
Cathodic reaction
2H+ + 2e → H2
Anodic reaction with Fats
C3H5(OCOR)3 + H2O/H+→ C3H5(OH)3 + 3RCOOH
Fat Glycerol Fatty acid
RCOOH → RCOO- + H+
2RCOO- → R-R + 2CO2 + 2e
Cathodic reaction
2H+ + 2e → H2
Bioelectrochemical reaction mechanism for earthworms indicated that they are corroded in acidic medium. Aloe Vera is used to control corrosive nature of soils. The solution of Aloe Vera was aided into different pH values of soils at variation of times and it results were recorded in table2 and figure3. The results of table 2 indicated that pH values of soils increased after addition of Aloe Vera and it minimized biological corrosion reaction. It is observed that number of earthworms increased after addition of Aloe Vera. It suppressed the concentration of H+ ions and controlled formation of biological corrosion cell.
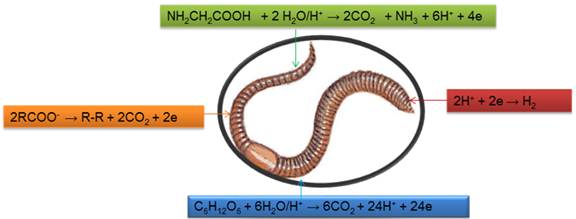
Figure 1. Bioelectrochemical corrosion reaction with earthworm
Table 1. pH values of soils without Aloe Vera in presence of earthworm
| Soils of different regions (150g) | pH | Earthworm | 24hrs | 48hrs | 72hr | 96hrs | 120hrs |
| Mining area (Jhariya in Dhanbad) | 5.6 | 25 | 18 | 15 | 11 | 6 | 1 |
| Industrial area (Chas,Bokaro) | 6.2 | 25 | 20 | 17 | 14 | 12 | 9 |
| Urban area (Chak Beriya) | 6.4 | 25 | 22 | 19 | 16 | 13 | 10 |
| Village area(Fulwariya, Chapra) | 6.7 | 25 | 24 | 23 | 20 | 18 | 15 |

Figure 2. Numbers of earthworm in different pH of soils Vs. time (hrs)
Table 2. pH values of soils with Aloe Vera in presence of earthworm
| Soils of different regions (150g) | pH | No. of Earthworms | 24hrs | 48hrs | 72hr | 96hrs | 120hrs |
| Mining area (Jhariya in Dhanbad) | 6.0 | 25 | 24 | 27 | 29 | 32 | 35 |
| Industrial area (Chas,Bokaro) | 6.5 | 25 | 25 | 30 | 34 | 37 | 45 |
| Urban area (Chak Beriya) | 6.8 | 25 | 27 | 33 | 38 | 47 | 51 |
| Village area( Fulwariya, Chapra) | 6.9 | 25 | 28 | 37 | 42 | 50 | 65 |
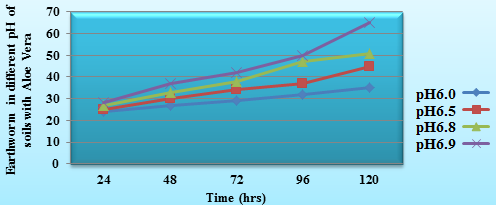
Figure 3. Earthworm in different pH of soils with Aloe Vera Vs. thime (hrs)
Earthworm’s weight was measured in absence and presence of Aloe Vera into different pH values of soils and its results are depicted in table 3 and figure4. It is noticed that its weight varies in different nature of soils. The weight of earthworm’s increased in Aloe Vera mixed soils with respect of unaided soils. The figure 5 indicated that after addition of Aloe Vera earthworms improved their physical and biological properties.
Heavy metals entered into soils by different sources and its compositions are recorded in Table4. They reacted with soils to acidic and basic compounds which generated corrosive atmosphere for earthworms and increased temperature of soils reducing humidity, concentration of oxygen, water and others natural minerals. Heavy metals concentrations were analyzed after addition of Aloe Vera; it is observed that their concentrations were decreased after addition of Aloe Vera.
Table 3. Earthworm position in different soils without and with Aloe Vera
| Soils | Mining area | Industrial area | Urban area | Village area |
| pH | 5.6 | 6.2 | 6.4 | 6.7 |
| Wt. of earthworm(mg) without Aloe Vera | 5.103 | 5.103 | 5.103 | 5.103 |
| Wt. of earthworm (mg) without Aloe Vera after 24hrs | 3.124 | 4.231 | 4.534 | 4.786 |
| Wt. of earthworm (mg) with Aloe Vera after 24hrs | 3.956 | 4.645 | 4.812 | 5.367 |
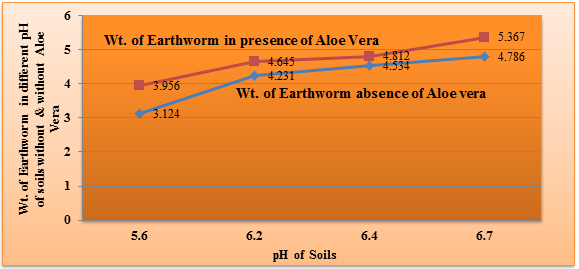
Figure 4. Weight of earthworm absence & presence of Aloe Vera Vs. pH

Figure 5. Earthworm kept in Aloe Vera aided soil
Table 4. Concentration of external minerals into Soils
| Minerals (in PPM) | Mining area soils | Industrial area soils | Urban area soils | Village area soils |
| Fe | 632 | 328 | 125 | 59 |
| Ni | 423 | 231 | 101 | 23 |
| Cr | 124 | 89 | 25 | 12 |
| Pb | 78 | 67 | 21 | 5 |
| As | 142 | 134 | 57 | 10 |
| Zn | 76 | 55 | 32 | 8 |
| Cu | 112 | 99 | 34 | 10 |
| Mo | 78 | 64 | 7 | 2 |
| Bi | 34 | 23 | 00 | 00 |
| Aloe Vera (100ml) 120hrs | Mining area soils(250g) | Industrial area soils(250g) | Urban area soils (250g) | Village area soils (250g) |
| Fe | 341 | 211 | 75 | 29 |
| Ni | 159 | 131 | 47 | 10 |
| Cr | 77 | 43 | 5 | 2 |
| Pb | 30 | 18 | 7 | 4 |
| As | 99 | 105 | 22 | 6 |
| Zn | 44 | 13 | 14 | 00 |
| Cu | 69 | 49 | 11 | 00 |
| Mo | 19 | 25 | 1 | 00 |
| Bi | 9 | 8 | 00 | 00 |
5. Conclusion
Earthworms are life of soils. Soil’s physical, chemical, biological and fertility properties depend on availability of earthworms. Their availabilities are reducing by toxic substances which are entered into soils by natural and artificial sources. Toxic substances were altered the pH values of soils and produced question mark on the survival of earthworm. Aloe Vera used as remedy for soils to check corrosive effect of soils. The experimental results observed that Aloe Vera had capability to control pH of soils, temperature, humidity, concentration of oxygen and composition of minerals and it was created ecofriendly atmosphere for soils and increasing the number of earthworms as well as their productivity.
Recommendation
Author is thankful to the UGC, New Delhi for providing financial support. Author is also thankful to the department of chemistry, Jagdam College, J P, University, Chapra for providing laboratory facilities.
References
- Bloomfield JP, Williams RJ, Gooddy DC, Cape JC, Guha P (2006) Impact of climate change on the fate and behavior of pesticides in surface and groundwater, Science of the Total Environment 369, 163-177.
- Baxter RA, Gilbert RE, Lidgett RA, Mainprize JH and Vodden HA (1975) The degradation of PCBs by microorganisms, Sci. Toatal Environ. 4, 53-61.
- Balthazor TM and Hallas LE (1985) Glyphosate –degrading microorganisms from industrial activated sludge Applied and Environmental Microbiology, Bol. 51, 432-434.
- Arias-Estevez M, Lopez-Periago E, Martinez-Carballo E (2008) (Review), The mobility and degradation of pesticides in soils and the pollution of groundwater resources. Agriculture, Ecosystems and Environment 123, 247-260. Eurasian Soil Science 39, 1271-1283.
- Chen G, Abichou T, Subramaniam PK (2007), Impact on surface charge density on colloid deposition in unsaturated porous media, Colloids and Surfaces A: Physicochem. Eng. Aspects 302, 342-348.
- Celis R, Hermosin MC, Carrizosa MJ, Cornejo J (2002) Inorganic and organic clays as carriers for controlled release of the herbicide hexazione, J. Agric. Food Chem. 50, 2324-2330.
- Dekker LW, CJRitsema and K Oostindie (2004) Dry Spots in Golf Courses: Occurrence, Amelioration and Prevention, Acta Horticulture 661, 99-104.
- Eggleton J and Thomas KV (2004) A review of factors affecting the release and bioavailability of contaminants during sediment disturbance events, Environment International 30, 973-980.
- Eriksson E, Baun A, Mikkelesen PS and Ledin A (2007) Risk assessment of xenobiotics in stromwater discharged to Harrestrup A, Denmark Desalination 215, 187-197.
- Feng JC, Thompson DG and Reynolds PE (1990) Fate of Glyphosate in a Canadian Forest Watershed, 1. Aquatic Residues and Off-Target Deposit Assessment, J. Agric. Food Chem. 38, 1110-1118.
- Feng X, Simpson A, Simpson M (2006) Investigation the role of mineral-bound humic acid in phenanthrene sorption, Environmental Science and Technology 40, 3260-3266.
- Ghanem A, Bados P, Estaum AR and Mougin C (2007) Concentrations and specific loads of glyphosate, diuron, atrazin, nonylphenol and metabolites thereof in French urban sewage sludge, Chemosphere 69, 1368-1373.
- Gurses A, Karaca S, Yalcin M (2004) Determination of adsorptive properties of clay|water system:methylene blue sorption, J. Colloid Interface Sci. 269, 310-314.
- Lombardi BM, Torres Sanchez RM, Eloy P, Genet M (2006) Interaction of thiabendazole and benzimidazole with montmorillonite, Appl. Caly Sci. 33, 59-65.
- Wick LY, Shi L, Harms H (2007) Electro-bioremediation of hydrophobic organic soil contaminants: A review of fundamental interactions, Electrochemica Acta 52, 3441-3448.



