Effect of Plant Hormone on Micropropagation and Callogenesis of Medicinal Plant (Curculigo orchioides)
Samaneh Zokae1, Tahmineh Esfandiyari Sabzevar2, Raheleh Ahmadzadeh Ghavidel3, *
1Samaneh Zokae, MSC of Plant Pathology, College of Agriculture, Ferdowsi University of Mashhad, Iran
2Tahmineh Esfandiyari Sabzevar, PhD Scholare of Medicenal Plant, College of Agriculture, Islamic Azad University of Shirvan, Iran
3Raheleh Ahmadzadeh Ghavidel, Phd of Agroecology, College of Agriculture, Ferdowsi University of Mashhad, Iran
Abstract
Curculigo orchioides is a perennial herb of the family Hypoxidaceae. It is an endangered plant species of medicinal importance. In this study a simple and efficient protocol for in vitro micropropagation of Curculigo orchioides. Through shoot tip and nodal portion explants and callogenesis of Curculigo orchioides through leaf, node and explants was used. A completely randomized design was followed for the experiment multiple shoots were obtained on all concentrations of BAP and NAA, but BAP (1 mg/l) showed the best response (90 and 80%) from both explants. Similarly, the best callus response was observed on MS medium supplemented with 2, 4- D + Kin (1+1 mg/l) in all explants. Then plantlets were transferred to different potting media in green house for acclimatization at the end of a three-month period, an average of 125 plants were obtained from a single meristem.
Keywords
Curculigo orchioides, Micropropagaton, Plant hormones, Tissue Culture
Received: May 30, 2016
Accepted: June 12, 2016
Published online: November 2, 2016
@ 2016 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY license. http://creativecommons.org/licenses/by/4.0/
1. Introduction
Curculigo orchioides (family - Hypoxidaceae), is a monocot with tuberous root stocks. The species is a stemless perennial herb of medicinal importance and a native of India. It is an herb with prominently nerved and plicate leaves. The flowers are bright yellow and distichous. It is normally propagated vegetatively through suckers. This is one of the first plant species that appears in the forest after the first shower of rains and also the last to disappear on completion of monsoon. Curculigo orchioides is reported to have hypoglycaemic, spasmolytic and anticancer principles (Faheem et al., 2011). Tissue culturing of medicinal plants is widely used to produce active compounds for herbal and pharmaceutical industries (Jasrai et al., 2000). In vitro propagation of plants holds a tremendous potential for producing high quality plant based medicines.
Micropropagation begins with the selection of plant material to be propagated. The plant tissues are removed from an intact plant in a sterile condition. Clean stock materials that are free of viruses and fungi are important in the production of the healthiest plants. Once the plant material is chosen for culture, the collection of explant(s) begins and is dependent on the type of tissue to be used; including stem tips, anthers, petals, pollen and others plant tissues. The explant material is then surface sterilized, usually in multiple courses of bleach and alcohol washes, and finally rinsed in sterilized water. This small portion of plant tissue, sometimes only a single cell, is placed on a growth medium, typically containing sucrose as an energy source and one or more plant growth regulators (plant hormones). Usually the medium is thickened with agar to create a gel which supports the explant during growth. Some plants are easily grown on simple media, but others require more complicated media for successful growth; the plant tissue grows and differentiates into new tissues depending on the medium. For example, media containing cytokinin are used to create branched shoots from plant buds (Kirtikar et al., 1935).
Various in vitro techniques as micropropagation from existing and adventitious meristems or organ, tissue and cell cultures provide a large amount of Curculigo orchioides. A simple, efficient and reproducible regeneration system for in vitro propagation of Curculigo orchioides via nodal explants cultured on MS medium supplemented with different concentrations of BAP, NAA and IBA was also reported (Murashige and Skoog 1962) Keeping the above facts in mind, namely the gradual decline of this endangered species, the present study was undertaken to develop a suitable protocol for its rapid multiplication.
2. Materials and Methods
The tubers were collected from fields, the materials were washed thoroughly under running tap water (1 h) and teepol. There was a serious problem of contamination with almost 70% of the explants, being affected even after a prolonged surface sterilization treatment. Keeping this in view, a long (15 - 18 h) pre-treatment of a more stringent nature was followed. Shoot apices were excised and stored in a solution containing chloramphenicol (0.1%), bavistin (0.1%), polyvinylpyrrolidone (0.1%), activated charcoal (0.1%) and citric acid (0.1%). In the laboratory, the excised apices were kept for pre-treatment (15 - 18 hr) on gyrotary shaker (150 rpm). This pretreatment solution consisted of chloramphenicol (0.5%) and bavistin (0.25%). The treated apices were washed thoroughly (three - four times) with distilled water. These explants were then surface sterilized with HgCl2 (0.1%; w/v). After repeated rinsing (five times) with distilled water, two - three leaves were removed and meristem tips (10 - 15 mm size) were inoculated on MS medium (Murch et al., 2000).
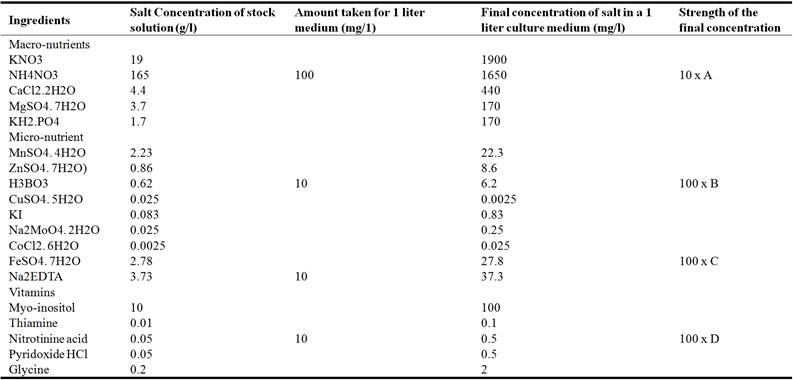
A modified Murashige and Skoog (1962) medium containing 3% Sucrose solidified with 0.8% Agar was used as the basal medium (Table 1).
Table 1. The composition a modified Murashige and Skoog medium.
Preparation and sterilization of growing medium (when not provided pre-poured) These steps will make 1 L of growth medium which is enough to prepare about 65 growing tubes. Dissolve the MS mixture in about 800 ml of distilled water. Stir the water continuously while adding the salt mixture. Add 30 g sugar and stir to dissolve. Adjust pH to 5.8 using 1M NaOH or 1M HCl as necessary while gently stirring. Add distilled water to make the total volume up to 1 L. 2. Weigh out 10 grams of agar and add it to the MS solution. Heat the solution gently while stirring until all the agar has dissolved. 3. Pour the still warm medium into the polycarbonate tubes to a depth of about 4 cm which will use about 15ml of media per tube. 4. Place the tubes (with lids sitting on the tubes but not tightened) in a pressure cooker and sterilize for 20 minutes. Cool the pressure cooker, then remove the tubes and tighten the lids. Alternatively, the tubes can be placed in boiling water for 30 minutes, but make sure that none of the water is able to enter the tubes ((Rout et al., 2000)
If you wish to use plants other than cauliflower you need to prepare two different media which contain plant hormones necessary to stimulate development of differentiated tissues. The first one should contain a cytokinin such as BAP which promotes shoot formation and the second one a rooting hormone such as NAA or store bought rooting hormone. To do this, prepare the mixture up until the end of step 2. Keeping the mixture warm so that it does not solidify, divide it equally into two pre-warmed containers. Each container can be used to prepare 30 or so tubes as above. The first container should have BAP added at the rate of 2.0mg/l. The second container should have the NAA hormone added at the rate of 0.1 mg/L. To do this it is necessary to make concentrated solutions of both BAP (2.0mg/ml) and NAA (1.0mg/ml) and filter sterilize them. Add 1ml of the concentrated BAP stock or 100μl of the NAA concentrated stock to each 1 liter of media that you prepare. If you use rooting hormone that is purchased from your local hardware or nursery supply store instead of NAA then just follow the directions before adding to your media (Ramawat et al., 2004).
supplemented with various concentrations ranging from 1.0-3.0 mg/l of BAP and NAA for micropropagation and 2,4-D alone ranging from 1.5-3.0 mg/l and also in combination with IBA (1+2 and 2+1mg/l). The pH of each medium was adjusted to 5.5+0.2 before adding phytagel and medium was autoclaved a 15 lb/inch2 for 20 minutes at 121°C. Cultures were incubated at 22+2°C with a photoperiod of 16 hours light and 8 hours dark at 2000-3000 lux light intensity of cool white fluorescent light. Shoot tips and nodal explants were used for shoot initiation in media supplemented with either various concentrations of BAP (1, 2 and 3 mg/l) or BAP+NAA (1+1, 1+2 and 2+1 mg/l). MS basal medium without growth regulators served as the control.
3. Results
To our knowledge there are no published reports on multiplication of Curculigo orchioides from shoot tips. The plant generation from meristems is considered to be one of the most promising ways for multiplying a selected variety true to its type. Such individuals are genetically similar showing the same agronomic characteristics. C. orchioides in its natural conditions is propagated through suckers, which are highly prone to viral infections (Narayanswamy, 1997). The response of shoot initiation from shoot tip in medium supplemented with BAP alone at all three concentrations was 90, 70 and 60 percent, respectively (Table 2). On other hand, medium supplemented with different combinations of BAP+NAA (1+1, 1+2 mg/l and 2+1 mg/l) showed delayed response with value of 70, 70 and 50 percent, respectively.
Table 2. Effect of BAP and combination with NAA on shoot induction from shoot tip explants of Curculigo orchioides.
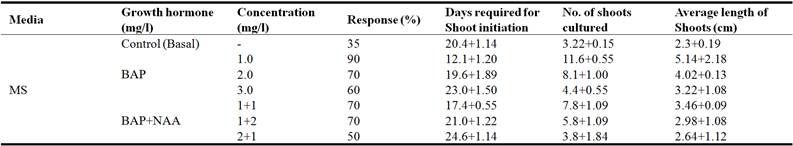
Table 3. Effect of various concentrations of BA on shoot growth from meristem-tip.

Various concentrations of BA (Table 3) were helpful in inducing the growth of meristem tip. However, the best and rapid growth was observed on MS medium with lowest concentrations of BA (0.44 μM). In contrast, a genus of a related family (Hypoxidaceae), Curculigo orchioides, required a very high concentration of BA (22.2 μM) for shoot regeneration (Nadgauda et al., 1978).
4. Discussion
Plant tissue culture is the technique of maintaining and growing plant cells, tissues or organs especially on artificial medium in suitable containers under controlled environmental conditions Multiple shoots were separated and transferred to liquid MS (half strength), containing different concentrations of NAA for root induction. Transfer to agar based semi-solid medium was not suitable for root induction. However, according to (Purohit et al., 1994), transfer of in vitro multiplied shoots from solid- to liquid medium with filter paper platform was necessary to ensure a healthy root system in turmeric. Emergence of roots occurred within a period of 15 - 20 days. Further incubation of one week led to a very vigorous root growth The maximum root growth was recorded on MS with 0.53 μM of NAA as a supplement (Table 4).
Table 4. Effect of various concentrations of NAA on root induction from in vitro raised shoots of Curculigo.

Half strength of MS medium, prior to their transfer to the soil. To check fungal growth, plants were sprayed with a 0.1% bavistin once a week. Humidity was maintained by covering it with rigid plastic cover and frequently spraying of water. Similar process of maintaining humidity was practiced for hardening of banana (Rolf et al., 1995) and Alpinia (Sidhu, 2010). The hardened plants were transferred to the field after 45 days of good growth Almost 96% of the regenerated plants survived and showed a vigorous growth of rhizome and roots without any morphological variations.
References
- Faheem, M., S. Singh, B. S. Tanwer, M. Khan and A. Shahzad. (2011). In vitro regeneration of multiplication shoots in Catharanthus roseus- An important medicinal plant. Adv. Appl. Sci. Res. 2 (1): 208-213.
- Jasrai, Y. T., Wala, B. B., (2000). Curculigo orchioides Gaertn. (Kali musli): An endangered medicinal herb, In: Role of Biotechnology in Medicinal and Aromatic Plants, Vol. IV, Khan IA and Khanum A (Eds.), Ukaaz Publications, Hyderabad, India, pp. 89-95.
- Kirtikar, K. R., Basu, B. D., (1935). Indian Medicinal Plants, Vol. IV, Blatter E, Caius JF and Mhaekar KS (Eds.), Bishen Singh Mahendra Pal Singh, Delhi, India, pp. 2469-2470.
- Murashige, T., F. Skoog. (1962). A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol. Plant. 15: 473-497.
- Murch, S. J., R. S., Krishna, P. K., Saxena. (2000). Tryptophan as a precursor for melatonin and serotonin biosynthesis in in-vitro regenerated St. John's ort (Hypericum perforatum. cv. Anthos) plants. Plant Cell Rep. 19: 698-704.
- Nadgauda, R. S., Mascarenhas, A. F., Hendre, R. A., Jagannathan, V. (1978). Rapid miltiplication of turmeric (Curcuma longa L.). Indian J. Exp. Biol., 16: 120-122.
- Narayanswamy, S., (1997). Plant Cell and Tissue Culture, Tata McGraw-Hill Publishing Co. Ltd., New Delhi, India, pp. 116-118.
- Purohit, S. D, Dave, A., Gotam, K. (1994). Micropropagation of safed musli (Chlorophytum borivilianum), a rare Indian medicinal herb. Plant Cell Tiss. Org. Cult., 39: 93-96.
- Ramawat, K. G., Sonie, K. C., Sharma, M. C. (2004). Therapeutic potential of medicinal plants: An introduction. In: Biotechnology of Medicinal Plants: Vitalizer and Therapeutics (Ramawat, K. G. Eds.), Sci. Pub. Inc., Enfield (NH), USA, p 1-18.
- Rolf, D. I., Ricardo, T. F. (1995). Micropropagation of Alpinia purpurata from inflorescence buds. Plant Cell Tiss. Org. Cult., 40: 183-185.
- Rout, G. R., Samantray, S., Das, P. (2000). In vitro manipulation and propagation of medicinal plants. Biotechnology Advances 18: 91-120.
- Sidhu, Y. (2010). In vitro micropropagation of medicinal plants by tissue culture. The Plymouth Student Scientist. 4 (1): 432-449.



