Hypoglycemic Antioxidant Potential of Herbal Extract Studied in High Fat Fed and Low Dose Streptozotocin Induced Type 2 Diabetic Rats
R. Maheswari*, M. Vennarasi
Department of Biochemistry, K. M. G College of Arts and Science, Gudiyattam, India
Abstract
In our study the use of plants for medicinal purpose used locally in the treatment of various diseases and we examined for their antioxidant activity. Therefore, the study deals with the evaluation of the beneficial effects of Tribulus terrestris (TT) extract on hyperglycemia, lipid profile, renal damage markers and oxidative stress in the liver and pancreas of type 2 diabetes mellitus (T2DM) in rats. T2DM was induced by feeding rats with high-fat diet (HFD, 40%) for two weeks followed by a single dose of Streptozotocin (STZ, 40mg/kg, bw, ip). An aqueous leaves extract of Tribulus terrestris (TT) was evaluated for its protective effect (antioxidant effect) against High Fat Fed/Diet (HFD), Streptozotocin (STZ) Type 2 Diabetes induced in male albino rats. An Aqueous leaves extract of Tribulus terrestris (TT), a source of several phytochemicals, is a potent antioxidant. Adult male wistar rats were divided into four different groups with 2 rats in each group. Group I Served as vehicle treated normal saline (Control), rats were fed standard diet (12% calories as fat) throughout the experiment. Group II Rats received (Control+TT) rats were fed standard diet throughout the experiment and given TT (50mg/kg body weight orally) for 4 weeks. Group III received (HFD/STZ group) rats were fed HFD (40% fat, 18% protein and 41% carbohydrate, as a percentage of total kcal) for 2 weeks and then injected with STZ (40 mg/kg bw, Ip, in citrate buffer; pH 4.5). Group IV Rats received (HFD/STZ+TT) rats were fed HFD for 2 weeks and then injected with STZ and then supplemented with TT for 4 weeks. After TT treatment, blood was drawn and rats were then sacrificed, and their liver and pancreas were dissected out for biochemical assays. The level of fasting blood glucose (FBG), glycated hemoglobin (HbA1C), total cholesterol (TC), triglycerides (TG), low density lipoprotein-cholesterol (LDL-C) and very low density lipoprotein-cholesterol (VLDL-C) significantly (P<0.05) increased while high density lipoprotein cholesterol (HDL-C) and hepatic glycogen decreased in the HFD/STZ group. TT treatment augmented these effects in the HFD/STZ+TT group. The HFD/STZ group showed elevated renal injury markers in serum, including blood urea nitrogen (BUN), serum creatinine (Scr) and alkaline phosphatase (ALP), which were decreased significantly (P<0.05) by TT treatment. Moreover, treatment with TT significantly (P<0.05) ameliorated thiobarbituric reactive substances (TBARS), malonaldehyde (MDA) and protein carbonyl (PC), and glutathione (GSH), glutathione-s-transferase (GST) and catalase (CAT) in liver and pancreas of HFD/STZ group. The study suggests that TT is effective in reducing hyperglycemia.
Keywords
Tribulus terrestris, Type 2 Diabetes Mellitus, Oxidative Stress, Hyperglycemia
Received: June 27, 2015
Accepted: July 4, 2015
Published online: July 20, 2015
@ 2015 The Authors. Published by American Institute of Science. This Open Access article is under the CC BY-NC license. http://creativecommons.org/licenses/by-nc/4.0/
1. Introduction
A disturbance in homeostatic phenomenon between generation of free radicals and the rate at which they are scavenged by enzymatic and nonenzymatic antioxidants is referred to as oxidative stress [1]. Type 2 diabetes mellitus is a heterogeneous disorder characterized by a progressive decline in insulin action (insulin resistance), followed by the inability of beta cells to compensate for insulin resistance (pancreatic beta cell dysfunction). Insulin resistance is a characteristic metabolic defect that proceeds over beta cell dysfunction and is primarily associated with resistance to insulin-mediated glucose disposal at the periphery and compensatory hyperinsulinemia.
Tribulus terrestris (L) (Family: Zygophyllaceae) is popularly known as Gokhshura in Sanskrit. The Sanskrit literature on Ayurveda describes many uses of the plant. Dwivedi [2] in Bhavaprakash nighantu in shloka No. 43 & 44, invigorating useful in the treatment of urinary affection, madhura, gastric stimulant, aphrodisiac, nutritive, used in the treatment of urinary calculi, polyuria, dyspnea, cough, piles dysuria, heart disease, pacifies deranged vata. Our ancient Rishi’s have described various medicinal properties of the herb. An attempt has been made, to study beneficial effects of the herb on human system and establish relationship between medicinal properties and chemistry of the plant.
2. Materials and Methods
2.1. Chemicals and Reagents
Tribulus terrestris (TT) plants leaves powder was obtained as a gift from Siddhamaruthuva Salai, Vellore. 5,5’-Dithiobis-(2-Nitrobenzoic acid) (DTNB), Glutathione reduced (GSH), Thiobarbituric acid (TBA), and 2,4-Dinitrophenyhydrazine (DNPH), Streptozotocin (STZ) were purchased from Sigma-Aldrich, Chemicals Pvt. Ltd, India. All other chemicals used were of good quality and analytical grade.
2.2. Tribulus terrestris (TT) Leaves Extract Preparation
Tribulus terrestris (TT) aqueous leaves extract was prepared by weighing accurately 10grams of dried leaves powder dissolved in 100ml of double distilled water (1/10 w/v) and centrifuged at 4°C for 20 min at 4000g the solution was stirred on a magnetic stirrer for one hour. It is then centrifuged and the supernatant was stored at -20°C until the end of the experiment. We selected an aqueous extract because most of the antioxidant components of Tribulus terrestris (TT) are extracted in water. During the experience, the aqueous Tribulus terrestris (TT) leaves extract was daily prepared and administrated to rats. 10% Tribulus terrestris (TT) leaves extract was prepared by weighing 1g of leaves powder in 10ml of double distilled water.
2.3. Animal Model
Male albino rats of Wistar strain (Rattus novergcus) (200±10g) procured from Tamil Nadu University for Veterinary and Animal Sciences, (TANUVAS) Chennai, India were used for the study. Animals were fed with commercially available standard rat pelleted feed (M/s Pranav Agro Industries Ltd., India) under the trade name Amrut rat/mice feed and water was provided ad libitum. The rats were housed under conditions of controlled temperature (30±2°C) and acclimatized to 12-h light, 12-h dark cycle. Animal experiments were conducted according to the guidelines of institutional animal ethical committee. Animals were randomly divided into four groups of 2 animals each.
2.4. Experimental Design
SEGGREGATION OF GROUPS: Experimental animals were divided into four groups of two rats in each group as follows.
Group I: Served as vehicle treated normal saline (Control). (12% Calories as fat)
Group II: Rats received standard diet throughout the experiment along with aqueous leaves extract of TT (50mg/kg body weight orally) for 4 weeks.
Group III: Rats received (HFD/STZ group) HFD (40% fat, 18% protein and 41% carbohydrate, as a percentage of total kcal) for 2 weeks and then injected with STZ (40 mg/kg body weight Intra peritoneal (IP), in citrate buffer, pH 4.5), (Table 1).
Group IV: Rats received (HFD/STZ+TT) rats were fed HFD for 2 weeks and then injected with STZ and then supplemented with TT for 4 weeks.
The development of hyperglycemia in rats was confirmed by fasting blood glucose (FBG) estimation after 6 days of STZ injection. The animals that maintained fasting blood glucose higher than 140mg/dl were considered diabetic and selected for studies. The TT treatment was started after diabetes was confirmed, and dose was determined from previous studies and literature survey.
Table 1. Composition of HFD (High Fat Diet) (Srinivasan K et al., 2004).
| Ingredients | Diet (g/kg) |
| Powdered NPD (Normal Pellet Diet) | 365 |
| Lard | 310 |
| Casein | 250 |
| Cholesterol | 10 |
| Vitamin and mineral mix | 60 |
| DL-Methionine | 03 |
| Yeast Powder | 01 |
| Sodium Chloride | 01 |
2.5. Tissue Preparation
Rats were anesthetized by ether inhalation and blood was collected from the dorsal aorta. Approximately 0.2 ml of whole blood was taken in EDTA containing microtubes from the total blood of each animal, and immediately preserved in the refrigerator for subsequent analysis of glycated hemoglobin (HbA1c). Serum was separated by centrifugation at 1200Xg for 10 min and stored at -80°C before analysis. Rats were then sacrificed, and their liver and pancreas tissues were excised immediately and perfused with ice-cold saline. For biochemical estimations, liver and pancreas tissues were homogenized at 4°C with 10times w/v 0.1M phosphate-buffer (PB), pH7.4 containing protease inhibitor. The homogenate was centrifuged at 800Xg for 5 min at 4°C to separate the nuclear debris and was used for estimation of Thiobarbituric reactive substances (TBARS) and Malonaldehyde (MDA). The supernatant was further centrifuged at 10,000Xg for 20min at 4°C to get the post-mitochondrial supernatant (PMS), which was used for various biochemical assays.
2.6. Statistical Analysis
Results are expressed as mean ± S.E.M. (n=2). Statistical analysis of the data was obtained via analysis of variance (ANOVA), followed by Tukey’s test. P<0.05 was considered statistically significant.
3. Results
Table 2. Effect of TT treatment on FBG, hepatic glycogen and HbA1c in the HFD/STZ induced rat model of diabetes.
| Groups/Parameters | Control (Ctrl) | Control + Tribulus Terrestris | HFD/STZ | HFD/STZ+Tribulus Terrestris | |
| Fasting Blood Glucose (mg/dl) | 105.74±5.0 | 107.30±4.5 | 326.85±6.5a | 223.80±7.8b | |
| Hepatic Glycogen (µg) | 363.25±4.5 | 365.02±3.41 | 167.84±1.29a | 228.04±2.4b | |
| HbA1c (%) | 5.53±0.27 | 5.80±0.90 | 11.16±0.65a | 8.79±0.75b | |
Values are expressed as mean ± S.E.M. (n=2).
Table 3. Effect of TT treatment on serum lipid profile.
| Groups/Parameters | Control (Ctrl) | Control + Tribulus Terrestris | HFD/STZ | HFD/STZ+Tribulus Terrestris |
| TC mg/dl | 152.24±2.3 | 154.91±2.4 | 297.42±3.8a | 203.42±2.1b |
| TG mg/dl | 104.24±2.7 | 105.31±2.9 | 254.55±2.2a | 185.74±3.3b |
| HDL-C mg/dl | 47.1±0.90 | 46.09±0.8 | 18.96±0.85a | 26.89±0.77b |
| LDL-C mg/dl | 81.65±1.1 | 83.63±1.4 | 225.11±0.92a | 136.96±0.80b |
| VLDL-C mg/dl | 20.23±0.8 | 20.00±0.96 | 48.89±1.3a | 36.23±0.86b |
Table 4. Effect of TT treatment on renal function markers (BUN, Scr and ALP) in serum of the HFD/STZ induced rat model of diabetes.
| Groups/Parameters | Control (Ctrl) | Control + Tribulus Terrestris | HFD/STZ | HFD/STZ+Tribulus Terrestris |
| BUN mg/dl | 17.00 ±1.30 | 18.12 ±3.9 | 49.00±1.3a | 38.52 ±0.77b |
| Scr mg/dl | 1.07 ±0.02 | 1.14 ±0.04 | 2.63 ±0.01a | 1.89 ±0.06b |
| ALP (units/dl | 24.30±0.67 | 25.10 ±0.72 | 42.63±0.81a | 30.85±0.54b |
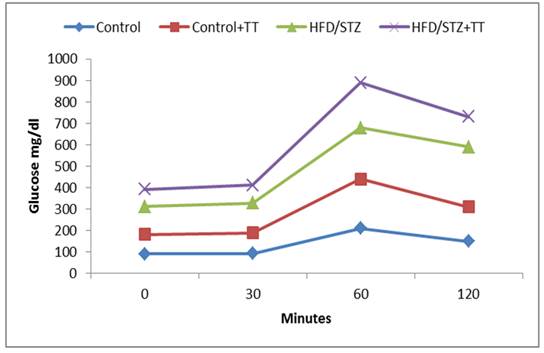
Graph 1. Effect of TT treatment on oral glucose tolerance.
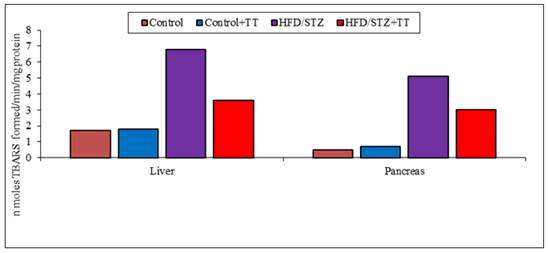
Graph 2 (A). Effect of TT treatment on TBARS levels.
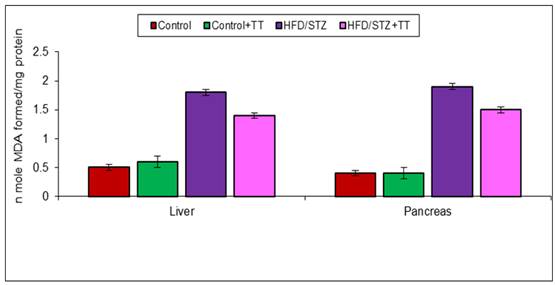
Graph 2 (B). Effect of TT treatment on MDA levels.
Graph 3 (A). Effect of TT treatment on PC content.
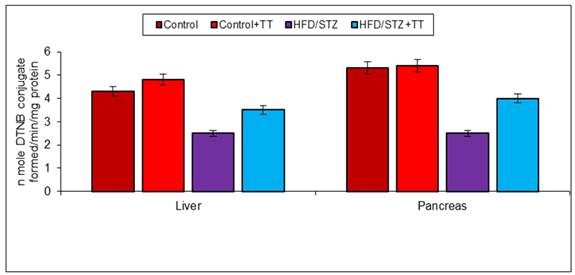
Graph 3 (B). Effect of TT treatment on GSH content.
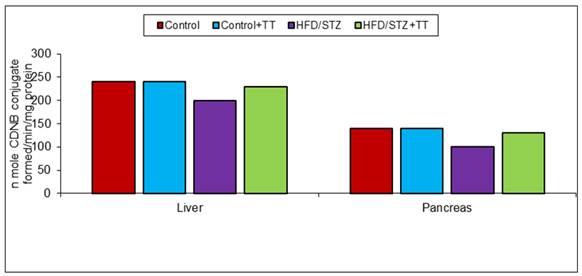
Graph 4 (A). Effect of TT treatment on GST activity.
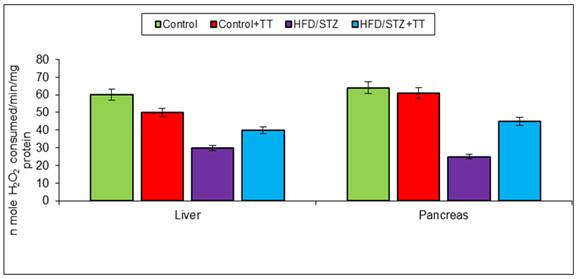
Graph 4 (B). Effect of TT treatment on CAT activity.

Fig. 1. Effect of High-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced Diabetes and TT on the SDS PAGE pattern of the hepatic tissue of control and experimental groups.
Protein fragmentation analysis by SDS PAGE electrophoresis in rat liver tissue homogenate.
Lane 1: control, Lane 2: High-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced Diabetes, Lane 3: HFD/STZ+ TT, Lane 4: TT alone.
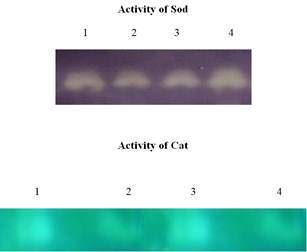
Fig. 2. Effect of Effect of High-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced and TT on the Native gel electrophoresis (SOD & CAT) pattern of the blood control and experimental groups.
4. Discussion
In the present study we demonstrated that HFD/STZ-induced diabetes in rats causes hyperglycemia, hyperlipidemia, renal function deficits and oxidative damage in liver and pancreas. Moreover, treatment with TT, by virtue of its anti-oxidant potential, significantly ameliorated HFD/STZ-induced alterations. This shows that TT possess a phytochemical reservoir of heuristic therapeutic value [3] and exhibits hypoglycemic and high anti-oxidant potential.
Elevated level of free radicals causes imbalance in homeostatic phenomena between oxidants and antioxidants in the body. The increase in free radicals in diabetic animals may be due to the auto-oxidation of glucose. Antioxidants, which can scavenge free radicals against damage and decay, have an important role in biological system and may be helpful in the prevention of these diseases. In the present study, during OGTT, blood glucose level were found to be increased in HD/STZ group with time and were maintained until 120 min in diabetic rats. TT treatment significantly improved glucose tolerance, as indicated by a reduction in peak blood glucose level at 60 and 120 min in the HFD/STZ+TT group. Moreover, the HFD/STZ-induced diabetic group exhibited increased FBG concentration while TT treatment to HFD/STZ+TT group reduced its concentration thereby showing its antihyperglycemic activity. Anti-hyperglycemic effects of TT are reporting due to increased peripheral glucose utilization. In our study, HbA1c level was also found to be significantly increased in HFD/STZ group compared to control rats while TT treatment decreased the level of HbA1c. This beneficial effect of TA may occur due to its free radical scavenging property inhibit oxidative reactions associated with protein glycation [4]. A decrease in blood glucose level might also contribute to decreased level of glycated hemoglobin in TT treated HFD/STZ group. It is clearly described that glycogen deposition from glucose is impaired in diabetic animals [5]. In our study, the hepatic glycogen content in HFD/STZ group decreased when compared to HFD/STZ+TT treated and control rats. These results were corroborated with previous studies in diabetic animals [6]. TT might enhance glucose utilization by peripheral tissues and increase the glycogen stores in the liver.
It has been demonstrated that during DM a variety of derangements in metabolic and regulatory processes occurs, which in turn leads to hyperlipidemic condition in diabetic people [7]. In our study, the HFD/STZ-model of diabetes exhibited abnormalities in lipid metabolism as evidenced from the significant elevation of serum TC, TG, LDL-C, VLDL-C and reduction of HDL-C levels. Treatment with TA for four weeks significantly reduced the TC, TG, LDL-C, VLDL-C level and significantly increased HDL-C levels in HFD/STZ rats. These results are in accordance with previous report [8,9].
In addition, recent evidence suggests that diabetic condition is associated with changes in morphology and eventually function alteration in kidney [10]. Previous research has reported increased activity of these biomarkers during renal damage [11]. Our work clearly show that increased levels of kidney functional markers in serum (BUN, Scr, and ALP) of HFD/STZ group. In contrast, the HFD/STZ+ TT treated rats showed significant reduction in these markers, thus showing its ability to protect against diabetes-induced kidney damage.
LPO (lipid peroxidation), is often used as an index of oxidative tissue damage which causes free radical damage to membrane components of the cell and resulting cell necrosis and inflammation [12]. The increase oxidative stress might have induced the peroxidation of polyunsaturated fatty acids and lead to the formation of TBARS and MDA, as byproducts of LPO. Increased TBARS and MDA production played an important role in the progression of diabetes [12]. Free radicals can also lead to the formation of carbonyl groups which are the end products of protein oxidation. Their levels in tissues serve as relatively stable markers of oxidative damage [13]. In the present study, TBARS, MDA and PC formation increased significantly in the liver and pancreas of HFD/STZ group. Treatment with TT significantly modulated these parameters.
In body endogenous system glutathione, acts as an antioxidant by scavenging free radicals [14]. It has been described that low glutathione levels in diabetes is an indicator of increased oxidative stress [15]. Decreased level of GSH in diabetic rats may increase susceptibility to tissue oxidative damage. In the present study, we also observed a significant decrease in GSH content in the liver and pancreas of HFD/STZ. TT treatment increased GSH content, is possibly due to less production of free radicals or its scavenging property. The decreased level of GSH is also contributed to lower activity of GST antioxidant enzyme, because GSH is required as a substrate for GST activity. Catalase is also another important antioxidant enzyme that involved in the removal of free radicals by the detoxification of H2O2. It breaks H2O2, a toxic compound, into water and oxygen. In the present study, we observed antioxidant enzymes (GST and CAT) activity decreased significantly in HFD/STZ group. Treatment with TT significantly improved these antioxidant enzymes activities in HFD/STZ+TT group.
It is thought that the flavonoids and OPCs provide free radicals scavenging activity and enhance the antioxidant status in body. Antidiabetic activity of TT may be due to the stimulation of the β- cells of the pancreatic islets.
In conclusion, our results showed that TT is a potent antidiabetic agent and beneficial in the control of diabetes related abnormalities in serum lipid profile, renal markers and oxidative damage in liver and pancreas of HFD/STZ-induced rat model of T2DM. Further studies regarding the mechanism underlying the protective action against oxidation, the lipid-lowering action, and the hypoglycemic action of TT extract need to be defined whether the TT extract can offer an alternate treatment for the patients of T2DM.
5. Conclusion
In view of these findings, it is possible to conclude that high-fat diet (HFD, 40%) for two weeks followed by a single dose of Streptozotocin administration results in pronounced oxidative stress and hepatic, renal damage. TT treatment would protect hepato and renal toxicity against high-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced toxicity through decreasing ROS, hydroxyl radicals, lipid peroxidation, preventing protein degradation (Fig 1), because TT are instrumental for the bioactive properties of leaves extract, above results may be important mechanisms underlying the protective effects of TT leaves extract observed in high-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced hepato and renal toxicity. Thereby, we provide the mechanistic basis of TT clinical application to protect patients from high-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced hepato and renal toxicity.
High-fat diet (HFD, 40%) followed by a single dose of Streptozotocin induced increased the hepato, renal and serum levels of oxidative stress markers such as lipid peroxidation and protein carbonyls. However, TT treatment significantly inhibited the formation of lipid peroxides and protein carbonyls in high-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced exposed rats. This could be due to potential antioxidant effect of TT.
Exposure to high-fat diet (HFD,40%) followed by a single dose of Streptozotocin induced significantly decreased the serum, hepatic and renal levels of protein thiol, reduced glutathione and the activities of superoxide dismutase, catalase, glutathione peroxidase, glutathione-S- transferases & glutathione reductase. TT treatment improved the antioxidant status by increasing the activities/levels of these enzymic and non-enzymic antioxidants.
Exposure to high-fat diet (HFD, 40%) followed by a single dose of Streptozotocin induced showed significant increase in the activities of hepatic and renal injury marker enzymes in serum and a subsequent decrease in these enzyme activities in kidney. Supplementation of TT reverted these hepatic and renal (kidney) injury markers enzyme activities to near normal.
The electrophoretic pattern of proteins by SDS PAGE also showed the protective role of TT
The native gel electrophoretic pattern of SOD and CAT also showed the protective efficacy of TT on high-fat diet (HFD, 40%) followed by a single dose of Streptozotocin induced hepatic and renal toxicity.
Along with biochemical and molecular alterations we have also observed histological changes in hepatic and renal tissues. Histopathological studies revealed alterations of the hepatic and renal tissues caused by high-fat diet (HFD, 40%) followed by a single dose of Streptozotocin induced exposure were prevented by TT administration.
Epidemiological evidence links high antioxidant status with low risk of degenerative disease including tumor promotion and cancer in humans. The increased consumption of fresh vegetables and fruits is usually associated with the decreased use of fish, meats and fats. Furthermore, supplementation of bioavailable and safe antioxidants are essential because we do not get enough antioxidant vitamins and minerals from foods and beverages we consume daily. These research studies demonstrate TT as a safe, novel, highly potent and bioavailable free radical scavenger and antioxidant possessing a broad spectrum of health benefits. TT functions at the genetic level and promotes therapeutic efficacy. Further mechanistic and clinical studies are in progress to unveil the mechanism of this novel natural antioxidant.
Acknowledgement
We are highly thankful to the faculty members of the PG & Research Department of Biochemistry for their appreciativeness and making the necessary facilities available for the research. The authors would like to thank the Secretary & Correspondent, Principal of K.M.G college Gudiyattam for their encouragement, providing the necessary facilities and support in carrying out the work.
References
- Papas MA. (1996). Determinants of antioxidant status in humans. Lipids. 31: 77-82.
- Dwivedi, V. (1977) "Bhavaprakash Nigantu", Motilal Banarasidas Delhi, 157. 2. Chatterjee, A. and Pakrashi, S., "The treatise of Indian Medicinal Plants", PID, Delhi,3, 128,(1994).
- Hartwell JL. (1982). In: Plants Used Against Cancer. A Survey, Lawrence MA: Quarterman, 89-93.
- Elgawish A, Glomb M, Freelander M, Monnier VM. (1996). Involvement of hydrogen peroxide in collagen cross-linking by high glucose in vitro and in vivo. J Biol Chem. 271: 12964–12971.
- Bollen M, Keppens S, Stalmans W. (1998). Specific features of glycogen metabolism in the liver. Biochem J. 336: 19–31.
- Tan BK, Tan CH, Pushparaj PN. (2005). Anti–diabetic activity of the semi–purified fractions of Averrhoa bilimbi in high fat diet fed–streptozotocin–induced diabetic rats. Life Sci. 76: 2827-2839.
- Goldberg RB. (1981).Lipid disorders in diabetes, Diabetes Care. 4: 561-572.
- Sangeeta, D., Sidhu, H., Thind, S.K., Nath, R., Vaidyanathan, S. (1993).Therapeutic response of T. terrestris aqueous extract on hyperodaluria in male adults rats. Phytotherapy research 7(2), 116-119.
- Vaidyaratnam PS. (1994).Varier’s Indian Medicinal Plants, A Compendium of 500 Species. Orient Longman: Arya Vaidya Sala Kottakal. 3: 423.
- Stumvoll M, Meyer C, Mitrakou A, Nadkarni V, Gerich JE. (1997). Renal glucose production and utilization: new aspects in humans. Diabetologia. 40: 749-757.
- Parveen K, Khan MR, Siddiqui WA. (2007). Pycnogenol® prevents potassium dichromate (K2Cr2O7)-induced oxidative damage and nephrotoxicity in rats. Chem Biol Interact. 181: 343-350.
- Ilhan N, Halifeoglu I, Ozercan HI. (2001). Tissue malondialdehyde and adenosine triphosphatase level after experimental liver ischaemia-reperfusion damage. Cell Biochem Funct. 19: 207-212.
- Chevion M, Berenshtein E, Stadtman ER. (2000). Human studies related to protein oxidation:protein carbonyl content as a marker of damage. Free Radic Res.33: S99–S108.
- Baynes JW, Thorpe SR. (1999). Role of oxidative stress in diabetic complications. A new perpective on an old paradigm. Diabetes. 48: 1-9.
- McLennan SV, Heffernan S, Wright L, Rae C, Fisher E, Yue DK, Turtle JR. (1991). Changes in hepatic glutathione metabolism in diabetes. Diabetes. 40: 344-348.



